Scalable Synthesis of Phosphorylated Indole-Isoquinolinone Derivatives via Manganese Promotion
Introduction to Advanced Heterocyclic Synthesis
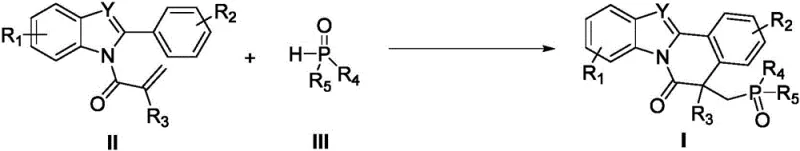
The development of efficient synthetic routes for complex fused heterocycles remains a cornerstone of modern medicinal chemistry, particularly for constructing scaffolds found in bioactive molecules and functional materials. Patent CN110467640B discloses a groundbreaking preparation method for indole or benzimidazole-isoquinolinone condensed heterocyclic derivatives, specifically targeting the incorporation of phosphorus moieties into these rigid frameworks. This innovation addresses the critical need for diverse, phosphorylated heterocycles which serve as pivotal intermediates in the design of next-generation pharmaceuticals and agrochemicals. By leveraging a manganese-promoted radical cyclization strategy, this technology enables the construction of quaternary carbon centers with high atom economy and exceptional regioselectivity. The methodology represents a significant leap forward in synthetic efficiency, offering a robust platform for generating libraries of novel compounds that were previously difficult or prohibitively expensive to access using traditional transition metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art strategies for synthesizing indolo-isoquinoline or benzimidazolo-isoquinoline skeletons often relied heavily on precious metal catalysis or energy-intensive photochemical processes. For instance, previous reports utilized silver-catalyzed decarboxylative radical cyclization or photocatalytic systems involving iridium or ruthenium complexes to drive the formation of these fused rings. While effective on a small laboratory scale, these conventional approaches suffer from severe economic and operational drawbacks when translated to industrial manufacturing. The reliance on expensive silver salts or specialized photocatalysts drastically inflates the raw material costs, making the final API intermediates financially unviable for mass production. Furthermore, these methods frequently exhibit narrow substrate scope, failing to tolerate sensitive functional groups or requiring stringent anhydrous conditions that complicate process engineering and increase safety risks in large-scale reactors.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes economical manganese(III) acetate dihydrate (Mn(OAc)₃·2H₂O) as a single-electron oxidant and promoter, eliminating the need for precious metals entirely. This manganese-mediated pathway operates under remarkably mild thermal conditions, typically around 60°C, using acetic acid as a benign and inexpensive solvent. The reaction demonstrates exceptional versatility, successfully coupling N-methacryloyl-2-phenylindoles with a wide array of P-H bond-containing compounds, including diarylphosphine oxides and dialkyl phosphites. This shift from noble metals to base metals not only reduces the direct cost of goods sold (COGS) but also simplifies the downstream purification process by removing the burden of trace heavy metal removal, a critical requirement for pharmaceutical grade materials. The result is a streamlined, cost-effective protocol that delivers high yields of phosphorylated products with excellent functional group tolerance.
Mechanistic Insights into Mn-Promoted Radical Cyclization
The core of this transformative synthesis lies in the unique ability of manganese(III) to initiate a free radical cascade cyclization. Mechanistically, the Mn(III) species acts as a one-electron oxidant that activates the P-H bond of the phosphorus source, generating a phosphorus-centered radical species. This highly reactive radical subsequently undergoes intermolecular addition to the electron-deficient alkene moiety of the N-methacryloyl group on the indole substrate. Following this initial addition, the resulting carbon-centered radical undergoes an intramolecular cyclization onto the adjacent aromatic ring (either the indole or benzimidazole system), effectively closing the isoquinolinone ring. The final oxidation and deprotonation steps restore aromaticity and yield the stable, fused heterocyclic product. This radical manifold is distinct from ionic pathways, allowing for the formation of sterically congested quaternary centers that are often challenging to construct via traditional nucleophilic substitutions.
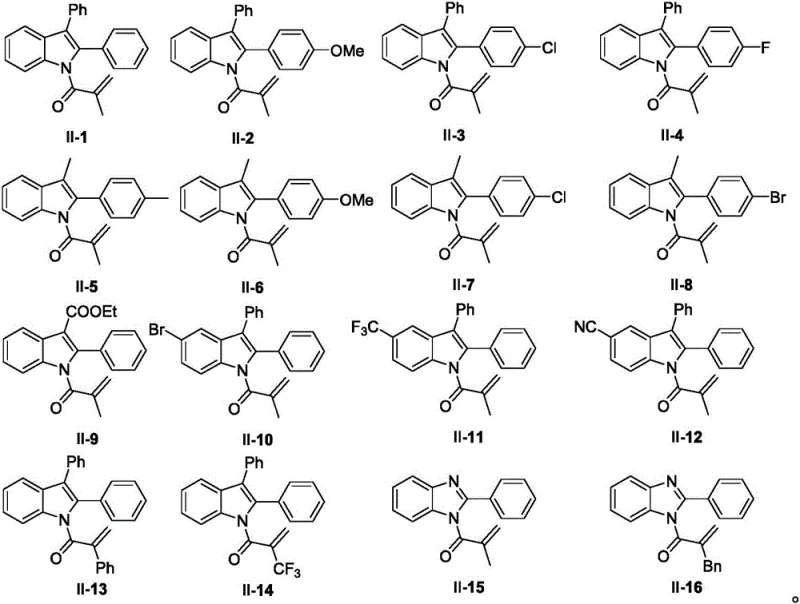
Furthermore, the mechanistic robustness of this system is evidenced by its remarkable tolerance to diverse electronic and steric environments. The reaction proceeds efficiently regardless of whether the indole backbone bears electron-withdrawing groups like halogens and nitro groups or electron-donating groups like methoxy and alkyl chains. This broad compatibility suggests that the radical intermediates are sufficiently long-lived to navigate the cyclization pathway without being quenched by common impurities or side reactions. For R&D teams, this implies a high degree of predictability when designing analogs; modifications to the core scaffold are unlikely to derail the synthesis, thereby accelerating the lead optimization phase in drug discovery programs. The ability to incorporate phosphorus directly into the scaffold also opens avenues for modifying the physicochemical properties of the final drug candidate, such as solubility and metabolic stability.
How to Synthesize Phosphorylated Indole-Isoquinolinone Efficiently
Executing this synthesis requires careful attention to stoichiometry and atmospheric control to maximize the efficiency of the radical propagation cycle. The standard protocol involves charging a reaction vessel with the indole substrate, the phosphorus source, and three equivalents of manganese(III) acetate dihydrate in acetic acid. The detailed standardized synthesis steps are provided in the guide below.
- Charge a Schlenk reactor with N-methacryloyl-2-phenylindole substrate, P-H containing compound, and Mn(OAc)3·2H2O promoter.
- Add acetic acid (HOAc) as the solvent and heat the mixture to 60°C under an inert argon atmosphere for 8-12 hours.
- Concentrate the reaction mixture under vacuum and purify the residue via silica gel column chromatography using n-hexane/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this manganese-promoted methodology offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with silver or palladium markets. Additionally, the use of commodity chemicals like acetic acid and manganese salts ensures a stable, multi-vendor supply chain for raw materials, mitigating the risk of shortages that often plague specialty reagents. The simplicity of the workup procedure, which relies on standard silica gel chromatography rather than complex scavenging resins for metal removal, further reduces processing time and waste generation, contributing to a leaner, more sustainable operation.
- Cost Reduction in Manufacturing: The substitution of expensive silver or photocatalytic systems with inexpensive manganese acetate results in a drastic reduction in catalyst costs per kilogram of product. Since manganese salts are bulk commodities, their pricing is stable and predictable, allowing for accurate long-term budgeting. Moreover, the absence of heavy metals simplifies the purification workflow, reducing the consumption of specialized scavengers and lowering the overall environmental compliance costs associated with heavy metal waste disposal.
- Enhanced Supply Chain Reliability: The starting materials, specifically N-methacryloyl-2-phenylindoles and various phosphine oxides, are structurally simple and can be sourced from multiple global suppliers or synthesized in-house using established protocols. This redundancy in the supply base ensures continuity of supply even during market disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the rate of batch failures and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures (60°C) and uses acetic acid, a solvent with a well-understood safety profile and established recycling infrastructure. This makes the process inherently safer and easier to scale from pilot plant to commercial production compared to photochemical reactions which require specialized flow reactors or large surface area illumination. The reduced toxicity profile of the reagents aligns with green chemistry principles, facilitating easier regulatory approval and minimizing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-promoted synthesis route. These answers are derived directly from the experimental data and scope analysis presented in the patent literature, providing a reliable foundation for process evaluation.
Q: What are the advantages of using Mn(OAc)3 over silver or photocatalysts?
A: Manganese(III) acetate is significantly more economical than silver salts or specialized photocatalysts. It operates under mild thermal conditions (60°C) without the need for expensive light sources, reducing both capital expenditure and operational costs for large-scale manufacturing.
Q: What is the substrate scope for this phosphorylation reaction?
A: The method exhibits broad universality, tolerating various substituents on the indole/benzimidazole ring including halogens, cyano, nitro, and alkoxy groups. It is compatible with diverse P-H sources such as aryl phosphine oxides, alkyl phosphine oxides, and phosphites.
Q: Is this process suitable for industrial scale-up?
A: Yes, the process utilizes simple equipment (Schlenk tubes or standard reactors), common solvents like acetic acid, and avoids hazardous reagents. The high yields and straightforward purification via column chromatography make it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorylated Indole-Isoquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of novel therapeutics. Our technical team has thoroughly analyzed the manganese-promoted pathway described in CN110467640B and is fully prepared to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phosphorylated indole-isoquinolinone derivative meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this cost-effective technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can accelerate your timeline and reduce your overall development costs.
