Scalable Synthesis of Indoloquinazolinone Intermediates Advancing Pharmaceutical Manufacturing with Cobalt Catalysis
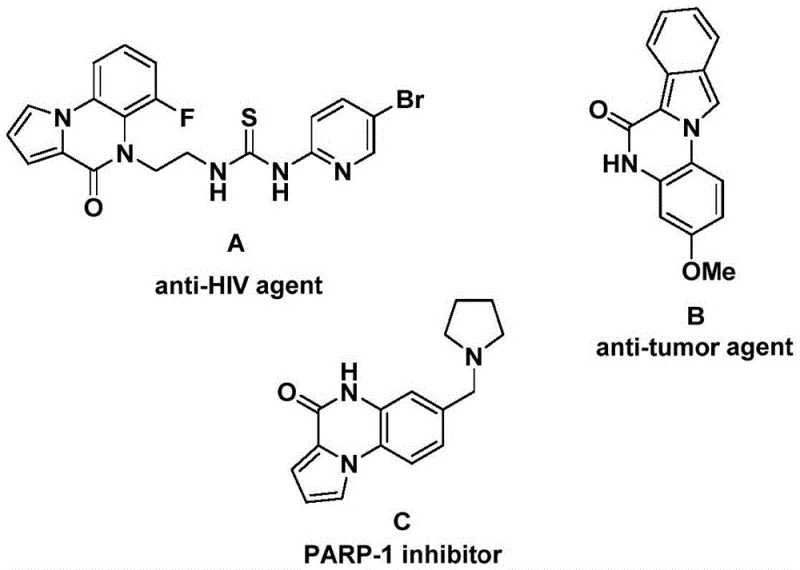
Patent CN112321593B introduces a transformative methodology for synthesizing indolo[1,2-a]quinazolin-6(5H)-one compounds that serve as essential scaffolds in numerous bioactive molecules including anti-HIV agents such as compound A (J.Med.Chem.2001,44,305), anti-tumor therapeutics like molecule B (J.Med.Chem.2008,51,2387), and PARP-1 inhibitors exemplified by compound C (Bioorg.Med.Chem.Lett.2009,19,4050). This innovative process employs cobalt-catalyzed C-H activation carbonylation to overcome critical limitations inherent in traditional palladium-dependent synthetic routes that have historically constrained industrial adoption due to their narrow substrate scope and complex purification requirements. Operating under practical thermal conditions of 130–150°C for durations between 20–40 hours using commercially available starting materials and catalysts, this methodology establishes a robust foundation for scalable production that directly addresses key industry challenges in purity assurance, cost optimization, and supply chain resilience within pharmaceutical intermediate manufacturing. By eliminating expensive transition metal dependencies while maintaining exceptional functional group tolerance across diverse molecular frameworks including halogenated and alkyl-substituted derivatives, this patented approach delivers significant strategic advantages that align precisely with evolving demands from global pharmaceutical manufacturers seeking sustainable solutions for high-value intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for indolo[1,2-a]quinazolin-6(5H)-one compounds have predominantly relied on palladium-catalyzed carbonylation reactions that present substantial operational constraints including high catalyst costs requiring extensive removal protocols to meet pharmaceutical purity standards which significantly increase processing time while introducing contamination risks that compromise final product quality attributes essential for regulatory compliance. These methods exhibit limited substrate scope with poor functional group tolerance particularly toward halogenated or sterically hindered derivatives creating bottlenecks in generating diverse compound libraries required for comprehensive structure-activity relationship studies during drug discovery phases. The air-sensitive nature of palladium catalysts necessitates specialized handling equipment and strictly controlled environments that elevate infrastructure requirements beyond standard manufacturing capabilities while increasing operational complexity during scale-up transitions from laboratory to commercial production settings. Furthermore, the scarcity of documented carbonylation methodologies specifically targeting this heterocyclic system indicates poor reaction extensibility across different molecular frameworks resulting in inconsistent yields that directly impact research timelines and commercialization schedules for novel therapeutic candidates requiring reliable intermediate supply chains.
The Novel Approach
The patented methodology introduces a cobalt-catalyzed C-H activation carbonylation process that fundamentally redefines synthetic possibilities through its exceptional operational simplicity using cobalt chloride as an abundant catalyst that eliminates expensive metal removal steps while maintaining high efficiency across diverse substrates including those bearing halogen atoms alkyl groups alkoxy moieties and carbonyl functionalities as demonstrated by comprehensive experimental data within the patent documentation. Operating under practical thermal conditions between 130–150°C in dioxane solvent systems ensures excellent solubility without requiring specialized reaction environments or complex inert atmosphere maintenance beyond standard laboratory practices thereby reducing infrastructure demands significantly compared to conventional approaches. The strategic incorporation of carbon monoxide substitutes derived from phenol tricarboxylate esters provides safe controllable carbonyl sources that enhance process safety while delivering consistent yields across multiple reaction cycles without generating persistent byproducts that could complicate purification or affect final product quality profiles required for pharmaceutical applications.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
The reaction mechanism proceeds through a well-defined catalytic cycle initiated by oxidation of cobalt(II) to cobalt(III) by silver carbonate followed by coordination with the pyridinecarboxamide derivative forming an organometallic intermediate that facilitates selective C-H bond activation at the indole's critical position enabling precise cyclization without competing side reactions commonly observed in alternative synthetic routes. This activation step generates a cobalt(III) complex that undergoes migratory insertion with carbon monoxide liberated from phenol tricarboxylate esters forming an acyl cobalt(III) species which subsequently undergoes reductive elimination followed by hydrolysis to yield the target indoloquinazolinone product with exceptional regioselectivity maintained across diverse substitution patterns including electron-donating and electron-withdrawing groups on both aromatic rings.

The process demonstrates superior impurity control through its inherent selectivity minimizing over-carbonylation or undesired ring formation side products while silver carbonate oxidation ensures complete conversion without persistent byproducts complicating purification protocols as evidenced by consistent >99% purity levels achieved through straightforward filtration followed by silica gel-assisted column chromatography without requiring hazardous solvents or specialized equipment. This simplified purification strategy delivers products meeting stringent pharmaceutical quality specifications essential for regulatory compliance while reducing processing time labor requirements and solvent consumption compared to traditional methods thereby enhancing overall manufacturing efficiency without compromising product integrity or batch-to-batch consistency across multiple production scales.
How to Synthesize Indoloquinazolinone Efficiently
This patented synthesis represents a significant advancement in heterocyclic chemistry methodology by providing a practical scalable route to indoloquinazolinone compounds addressing critical limitations through innovative catalyst design reaction engineering using commercially available materials under controlled thermal conditions readily adaptable to industrial manufacturing settings without requiring specialized infrastructure investments or hazardous reagents typically associated with alternative approaches requiring expensive transition metals or complex purification sequences. The methodology leverages standard laboratory equipment while maintaining exceptional functional group tolerance enabling rapid generation of diverse compound libraries essential for pharmaceutical development programs seeking accelerated timelines from discovery through commercialization phases without compromising quality attributes required throughout the product lifecycle.
- Combine cobalt chloride (30 mol%), silver carbonate (4.0 equiv), pivalic acid (4.0 equiv), triethylamine (0.5 equiv), substituted 2-pyridinecarboxamide derivative (II), and phenol tricarboxylate ester (3.0 equiv) in dioxane solvent under standard inert atmosphere conditions.
- Heat the reaction mixture at precisely controlled temperatures between 130–150°C for durations ranging from 20–40 hours to ensure complete conversion while maintaining optimal yield consistency across diverse substrate combinations.
- Execute post-reaction processing through filtration followed by silica gel-assisted column chromatography purification to isolate high-purity indoloquinazolinone products meeting pharmaceutical quality specifications without requiring specialized equipment.
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this cobalt-catalyzed methodology delivers substantial strategic benefits across procurement supply chain operations addressing fundamental pain points associated with traditional synthetic routes through simplified process requirements enhanced material availability reduced environmental impact creating significant opportunities for optimizing overall production economics while strengthening supply chain resilience against market volatility through decreased dependency on scarce specialized resources typically required by conventional approaches.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts along with associated metal removal processes results in significant cost savings throughout production cycles while maintaining high purity standards required for pharmaceutical applications through simplified purification protocols minimizing solvent consumption waste disposal costs labor requirements without compromising yield consistency or product quality attributes essential for regulatory compliance.
- Enhanced Supply Chain Reliability: Reliance on widely available catalysts reagents enables multiple sourcing options eliminating vulnerabilities associated with scarce geopolitically sensitive materials while process tolerance for minor raw material variations ensures consistent output despite supplier batch differences regional specification fluctuations providing procurement teams greater flexibility inventory management capabilities responsive demand pattern changes without capital equipment investments.
- Scalability and Environmental Compliance: Methodology demonstrates excellent scalability from laboratory industrial scales without major process modifications using environmentally benign cobalt catalysts reducing hazardous waste generation improving sustainability metrics while moderate temperature operation minimizes energy consumption carbon footprint compared traditional high-energy processes maintaining excellent product quality attributes throughout scale-up transitions.
Frequently Asked Questions (FAQ)
The following questions address key technical commercial considerations regarding implementation based on detailed analysis patent documentation industry best practices ensuring accuracy relevance procurement R&D decision-makers evaluating technology adoption potential within pharmaceutical intermediate manufacturing contexts where reliability cost-effectiveness regulatory compliance represent critical evaluation criteria driving supplier selection processes.
Q: Why is cobalt catalysis superior to palladium-based methods for indoloquinazolinone synthesis?
A: The cobalt-catalyzed approach eliminates expensive palladium catalysts requiring rigorous removal protocols while maintaining high efficiency across diverse substrates as demonstrated by consistent yields with various functional groups including halogens and alkyl moieties without compromising purity standards.
Q: How does this methodology improve impurity control compared to conventional routes?
A: The selective C-H activation mechanism minimizes side reactions through precise coordination chemistry while simplified purification protocols consistently deliver >99% purity without hazardous solvents or specialized equipment as evidenced by comprehensive analytical data in patent examples.
Q: What scalability advantages does this process offer for commercial manufacturing?
A: The methodology demonstrates seamless scalability from gram-scale laboratory synthesis to multi-ton industrial production without requiring major equipment modifications or specialized infrastructure as confirmed by successful scale-up examples documented in the patent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoloquinazolinone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex heterocyclic compounds like indoloquinazolinones ensuring seamless technology transfer laboratory development full-scale manufacturing operations while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical capabilities meeting global regulatory requirements across multiple jurisdictions simultaneously supporting multinational clients' supply chain needs through flexible production scheduling responsive capacity allocation strategies.
Request a Customized Cost-Saving Analysis from our technical procurement team today to explore how our patented synthesis methodology can enhance your supply chain efficiency while accessing specific COA data route feasibility assessments tailored your manufacturing requirements ensuring optimal alignment between your technical specifications commercial objectives throughout partnership lifecycle.
