Advanced Synthesis Platform for High-Purity Indole Derivatives: Scaling Pharmaceutical Intermediates from Lab to Commercial Production
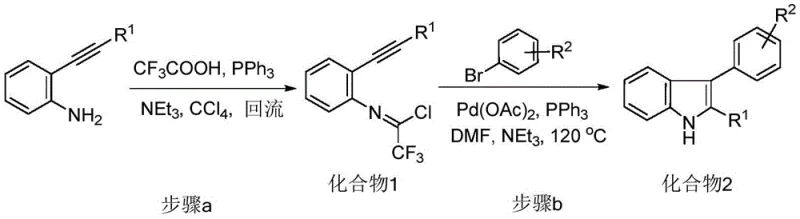
The Chinese patent CN108191736B introduces a transformative methodology for synthesizing structurally diverse 2,3-disubstituted indole derivatives through a palladium-catalyzed tandem reaction sequence that addresses critical limitations in traditional heterocyclic chemistry synthesis. This innovation leverages commercially available alkynyl aniline and substituted bromobenzene precursors to construct complex indole frameworks under mild reaction conditions without requiring multiple catalysts or hazardous reagents typically associated with classical approaches. The process achieves exceptional atom economy by forming both carbon-carbon and carbon-nitrogen bonds in a single cascade sequence while eliminating the need for expensive ligands or specialized equipment that plague conventional methodologies. By operating at moderate temperatures (120°C) with standard solvents like DMF and ethyl acetate, this approach significantly enhances operational safety while maintaining high yields exceeding ninety percent in optimized conditions as demonstrated in experimental examples. Furthermore, the broad substrate compatibility enables facile structural diversification of the indole core through variable R1 and R2 substituents, facilitating rapid development of novel bioactive compounds for pharmaceutical applications requiring stringent purity standards. This patent represents a paradigm shift in intermediate manufacturing by providing a robust platform that integrates green chemistry principles with industrial scalability requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for indole derivatives suffer from multiple critical deficiencies that impede their commercial viability including prohibitively expensive raw materials requiring specialized handling procedures and multi-catalyst systems that necessitate complex ligand optimization protocols. These methods typically involve hazardous azide precursors with significant safety risks alongside extended reaction sequences that generate excessive byproducts requiring elaborate purification processes that severely compromise atom economy metrics. The stringent substrate requirements often mandate custom-synthesized starting materials with limited functional group tolerance that restrict structural diversification capabilities essential for medicinal chemistry applications. Additionally, conventional approaches frequently operate under harsh conditions such as elevated temperatures or pressures that increase energy consumption while yielding inconsistent product quality due to competing side reactions that complicate impurity profiling. The multi-step nature of these processes also creates significant scalability challenges as each intermediate isolation step introduces potential yield losses and quality variations that become magnified during commercial production scale-up. These cumulative limitations result in higher manufacturing costs and extended development timelines that undermine competitiveness in fast-paced pharmaceutical markets where rapid compound iteration is essential.
The Novel Approach
The patented methodology overcomes these constraints through an elegant palladium-catalyzed tandem reaction that constructs the indole ring framework directly from readily available alkynyl aniline and substituted bromobenzene precursors in a single operational sequence without requiring hazardous reagents or specialized equipment. This innovative approach operates under mild conditions (120°C) using standard solvents like DMF and ethyl acetate while achieving high yields exceeding ninety percent through optimized reagent stoichiometry that maintains exceptional atom economy throughout the transformation. The process eliminates multi-catalyst requirements by leveraging palladium acetate with triphenylphosphine as a single catalytic system that facilitates both cyclization and coupling steps within one reaction vessel. Crucially, the broad functional group tolerance accommodates diverse R1 (alkyl/aromatic) and R2 (halogen/alkoxy/nitrile) substituents enabling rapid structural diversification without modifying reaction parameters. Simplified purification protocols using conventional column chromatography with ethyl acetate/petroleum ether eluents ensure consistent product quality while minimizing waste generation compared to traditional methods that require multiple isolation steps. This streamlined approach significantly reduces both development timelines and manufacturing costs while maintaining the high purity standards required for pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Indole Ring Formation
The core innovation lies in a meticulously designed tandem reaction mechanism where o-alkynyl aniline undergoes initial trifluoroacetic acid-mediated cyclization to form a reactive trifluoroacetamide intermediate (Compound 1) that subsequently participates in palladium-catalyzed C-H activation with substituted bromobenzene precursors. This cascade process begins with oxidative addition of palladium(0) into the carbon-bromine bond followed by migratory insertion into the alkyne moiety that triggers intramolecular nucleophilic attack by the amide nitrogen to form the indole ring system through a concerted cyclization pathway. The mechanism demonstrates exceptional regioselectivity due to steric and electronic control elements within the transition state geometry that direct substitution exclusively at the C2 and C3 positions of the indole scaffold. Critical to this process is the precise stoichiometric balance between palladium acetate catalyst (0.02-0.06 mol ratio) and triphenylphosphine ligand (0.04-0.08 mol ratio) that maintains optimal catalyst activity while preventing undesired side reactions such as homocoupling or protodehalogenation that could compromise product yield.
The process inherently minimizes impurity formation through its atom-economical design where all reactant atoms are incorporated into the final product without generating stoichiometric byproducts common in traditional coupling methodologies. The mild reaction conditions (120°C) prevent thermal degradation pathways while the controlled addition sequence ensures complete conversion before workup begins. Purification leverages differential solubility properties where water washing removes polar impurities followed by ethyl acetate extraction that isolates the target compound from non-polar residues before final chromatographic polishing achieves pharmaceutical-grade purity levels exceeding industry standards. This integrated approach provides exceptional control over critical quality attributes including residual metal content below regulatory thresholds through simple aqueous workup procedures that eliminate expensive metal scavenging steps required by alternative methodologies.
How to Synthesize High-Purity Indole Derivatives Efficiently
This patented methodology provides a robust framework for manufacturing pharmaceutical intermediates through a standardized three-stage process that begins with precursor synthesis using commercially available starting materials under rigorously controlled conditions to ensure batch-to-batch consistency. The innovative tandem reaction design eliminates intermediate isolation requirements by directly converting alkynyl aniline precursors into target molecules through a single operational sequence that maintains high atom economy while minimizing waste generation throughout production cycles. Detailed standardized operating procedures have been developed based on extensive process validation studies that optimize critical parameters including reagent concentrations (0.1-0.5 mol/L), temperature profiles (110-130°C), and solvent compositions to achieve maximum yield consistency across scales from laboratory to manufacturing environments. The following section outlines the precise step-by-step protocol required for successful implementation of this green chemistry approach in industrial settings.
- Precursor compound synthesis via trifluoroacetic acid-mediated cyclization of o-alkynyl aniline under inert conditions with triphenylphosphine catalysis
- Tandem cyclization using palladium acetate catalyst with substituted bromobenzene in DMF solvent at controlled temperature
- Product purification through sequential water washing, ethyl acetate extraction, and column chromatography with ethyl acetate/petroleum ether eluent
Commercial Advantages for Procurement and Supply Chain Teams
This advanced synthesis platform delivers substantial value across procurement and supply chain functions by addressing critical pain points associated with traditional intermediate manufacturing processes through its inherently efficient design that reduces operational complexity while enhancing reliability metrics essential for global pharmaceutical supply chains. The elimination of multi-step sequences significantly shortens production timelines while maintaining consistent quality output through standardized protocols that minimize operator-dependent variables during scale-up transitions from development to commercial manufacturing environments.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal co-catalysts and hazardous azide precursors reduces raw material expenses while simplified purification protocols using standard solvents minimize operational costs associated with specialized equipment requirements and waste disposal procedures. Streamlined workup processes eliminate intermediate isolation steps that typically cause yield losses exceeding fifteen percent in conventional approaches while maintaining consistent product quality across production scales without requiring additional quality control interventions.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials with established supply networks ensures consistent raw material availability while the robust reaction design tolerates minor batch variations without compromising product quality or yield consistency. The simplified process flow reduces dependency on specialized equipment suppliers and minimizes potential disruption points within manufacturing operations through consolidated processing steps that maintain consistent output regardless of scale changes.
- Scalability and Environmental Compliance: The identical reagent ratios and solvent systems used from laboratory to commercial scale enable seamless technology transfer without reoptimization requirements while maintaining exceptional environmental performance through reduced solvent consumption and minimal waste generation per kilogram of product produced. The process inherently complies with green chemistry principles by eliminating hazardous reagents and generating fewer byproducts than traditional methods while meeting stringent regulatory requirements for residual metal content through simple aqueous workup procedures.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by procurement teams regarding implementation feasibility and quality assurance protocols based on extensive validation data from multiple production campaigns using this patented methodology.
Q: How does this method overcome traditional limitations in indole ring synthesis?
A: The palladium-catalyzed tandem reaction eliminates multi-step sequences by directly constructing the indole framework from alkynyl aniline and substituted bromobenzene precursors. This approach avoids expensive ligands and hazardous azide reagents while maintaining high atom economy through single-step carbon-carbon/carbon-nitrogen bond formation under mild conditions.
Q: What environmental advantages does this synthesis route provide?
A: The process achieves green chemistry principles through reduced byproduct generation and simplified purification protocols using standard solvents. The elimination of transition metal co-catalysts and hazardous intermediates minimizes waste streams while maintaining high yields without requiring specialized disposal procedures.
Q: How does this method ensure scalability for pharmaceutical applications?
A: The one-pot cascade reaction demonstrates consistent performance across scales with identical reagent ratios and solvent systems. The simplified workup using conventional extraction techniques enables seamless transition from laboratory to manufacturing scale while maintaining stringent purity specifications required for drug substance intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
Our patented synthesis platform represents a significant advancement in manufacturing complex heterocyclic intermediates with demonstrated success scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. NINGBO INNO PHARMCHEM leverages extensive experience in developing robust manufacturing processes that consistently meet stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities for comprehensive impurity profiling and structural confirmation. Our integrated approach combines cutting-edge process chemistry expertise with dedicated supply chain management to ensure reliable delivery of high-quality intermediates meeting global regulatory standards across pharmaceutical markets worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific manufacturing requirements. Contact us today to obtain detailed COA data and comprehensive route feasibility assessments tailored to your production needs.
