Advanced Convergent Synthesis of JAK Inhibitor Intermediates for Commercial Scale-Up
Advanced Convergent Synthesis of JAK Inhibitor Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for kinase inhibitors, particularly those targeting the JAK-STAT pathway, which plays a pivotal role in immunoregulation and cell proliferation. Patent CN111943959A, published in late 2020, discloses a highly efficient synthesis method for a specific JAK inhibitor that addresses critical bottlenecks in traditional manufacturing. This technology leverages a convergent strategy, splitting the synthesis into two distinct branches—Intermediate A series and Intermediate B series—before a final coupling step. This approach not only simplifies the purification process but also significantly enhances the overall yield and purity of the target molecule. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials for clinical and commercial needs.
The structural complexity of JAK inhibitors often necessitates multi-step syntheses involving sensitive heterocyclic systems. The disclosed method utilizes 4-nitropyrazole and 3-oxetanone as the foundational building blocks. By breaking down the synthesis into manageable fragments, the process minimizes the accumulation of impurities that typically plague linear synthesis routes. The final target molecule features a thieno[3,2-d]pyrimidine core linked to a pyrazole moiety and an oxetane ring, a structural motif known for its metabolic stability and binding affinity. The ability to construct this complex architecture from simple, commercially available precursors represents a significant advancement in cost reduction in API manufacturing, offering a viable pathway for large-scale production without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for JAK inhibitors often suffer from low atom economy and the requirement for harsh reaction conditions that can degrade sensitive functional groups. Many legacy processes rely on late-stage functionalization of complex cores, which leads to difficult-to-remove impurities and necessitates extensive chromatographic purification. Furthermore, the use of expensive transition metal catalysts without efficient recovery systems can drastically inflate the cost of goods sold (COGS). In many cases, the linear nature of these older methods means that a failure in any single step results in the loss of all prior value-added work, creating significant supply chain risks. The accumulation of side products, particularly in nucleophilic substitution reactions on electron-deficient heterocycles, often requires repeated recrystallization, further reducing the overall yield and extending the production lead time.
The Novel Approach
The methodology outlined in CN111943959A overcomes these challenges through a modular design that isolates potential failure points. By synthesizing Key Intermediate A-2 and Key Intermediate B-3 separately, manufacturers can ensure high purity of each fragment before the final assembly. The use of Chan-Lam coupling for the introduction of the cyclopropyl group onto the pyrazole ring is a standout feature, offering milder conditions compared to traditional nucleophilic aromatic substitution. Additionally, the final coupling step utilizes n-butanol as a solvent at 100°C, a condition that is easily manageable in standard stainless steel reactors. This strategic separation of concerns allows for better process control, effectively reducing the generation of impurities and byproducts. Consequently, this novel approach provides a clear path toward commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply continuity.
Mechanistic Insights into Chan-Lam Coupling and Pd-Catalyzed Substitution
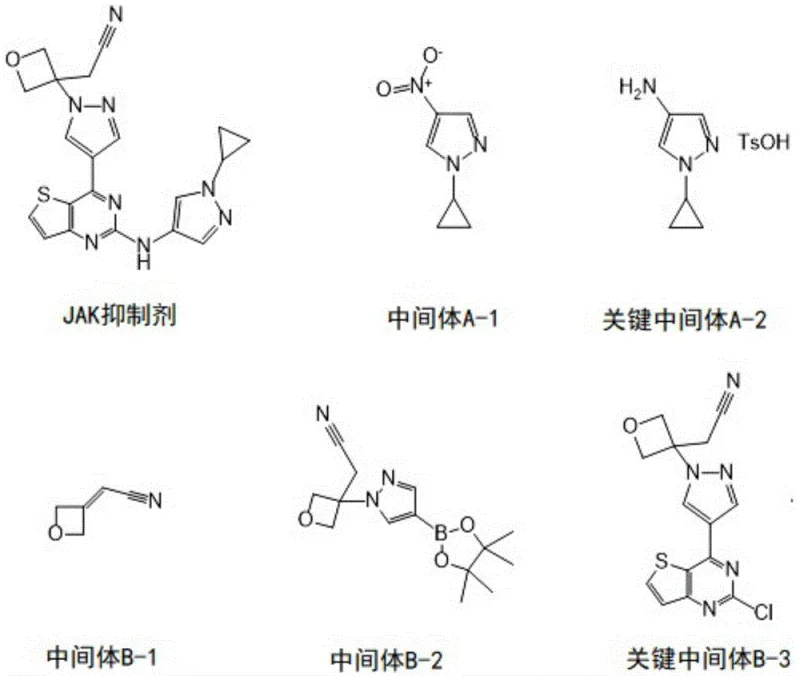
The synthesis of Key Intermediate A-2 begins with a copper-mediated Chan-Lam coupling reaction between 4-nitropyrazole and cyclopropylboronic acid. This transformation is critical as it installs the cyclopropyl moiety, which is essential for the biological activity of the final inhibitor. The reaction proceeds in 1,2-dichloroethane at 70°C in the presence of anhydrous copper acetate and a base like sodium carbonate. Mechanistically, this involves the transmetallation of the boronic acid to the copper center, followed by oxidation and reductive elimination to form the C-N bond. This method is superior to direct alkylation as it avoids the formation of poly-alkylated byproducts. Following this, the nitro group is reduced to an amine using hydrogen and a palladium-carbon catalyst, followed by protection with p-toluenesulfonic acid (TsOH) to stabilize the intermediate for subsequent steps.
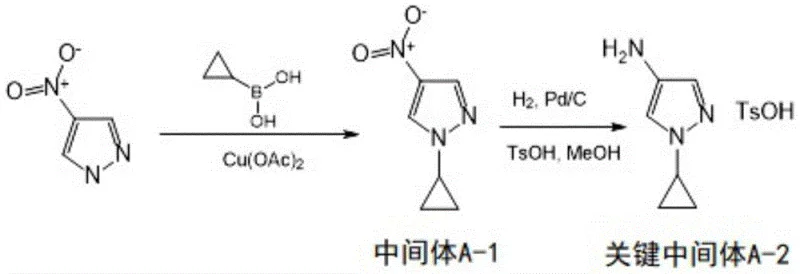
Parallel to this, the construction of the B-fragment involves a Wittig reaction to convert 3-oxetanone into an unsaturated nitrile (Intermediate B-1), followed by a conjugate addition with a pyrazole boronic ester. The subsequent formation of Key Intermediate B-3 employs a palladium-catalyzed cross-coupling with 2,4-dichlorothieno[3,2-d]pyrimidine. This step utilizes [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium (Pd(dppf)Cl2) as the catalyst, which is highly effective for coupling sterically hindered substrates. The selectivity of this reaction is paramount, as it must distinguish between the two chlorine atoms on the thienopyrimidine ring to ensure the correct regioisomer is formed. The careful optimization of these catalytic cycles ensures that the high-purity pharmaceutical intermediates required for downstream processing are obtained with minimal metal contamination.
How to Synthesize JAK Inhibitor Efficiently
The complete synthesis involves the convergence of the two prepared fragments. Key Intermediate A-2 acts as the nucleophile, attacking the electrophilic center of Key Intermediate B-3. This final nucleophilic substitution is performed in n-butanol at elevated temperatures to drive the reaction to completion. The choice of n-butanol is strategic, providing a balance between solubility of the polar intermediates and the boiling point required to overcome the activation energy barrier. Detailed operational parameters, including specific stoichiometric ratios and workup procedures, are critical for maximizing the 40% isolated yield reported in the patent examples. For a comprehensive understanding of the exact laboratory protocols, please refer to the standardized synthesis guide below.
- Synthesize Key Intermediate A-2 via Chan-Lam coupling of 4-nitropyrazole with cyclopropylboronic acid followed by hydrogenation.
- Prepare Key Intermediate B-3 through Wittig reaction of 3-oxetanone, followed by addition and Pd-catalyzed substitution.
- Perform final nucleophilic substitution between Intermediate A-2 and B-3 in n-butanol at 100°C to obtain the JAK inhibitor.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits. The reliance on commodity chemicals such as 3-oxetanone and 4-nitropyrazole mitigates the risk of raw material shortages that often plague specialized reagent markets. Furthermore, the avoidance of cryogenic conditions or ultra-high pressure equipment simplifies the capital expenditure requirements for manufacturing facilities. This accessibility translates directly into a more resilient supply chain, capable of responding rapidly to fluctuations in market demand. For supply chain heads, the predictability of this process is a major asset, as it reduces the likelihood of batch failures and ensures a steady flow of materials for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of complex protecting group strategies and the use of catalytic rather than stoichiometric amounts of expensive metals significantly lower the variable costs per kilogram. By streamlining the purification steps through improved impurity profiles, the consumption of silica gel and organic solvents is drastically reduced. This efficiency gain allows for a more competitive pricing structure without sacrificing margin, making the final API more accessible for healthcare systems.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the parallel production of Intermediate A-2 and Intermediate B-3. This decoupling means that if one line encounters a delay, the other can continue to operate, maintaining inventory levels of the sub-components. Additionally, the stability of the key intermediates allows for stockpiling, providing a buffer against unexpected disruptions in the global logistics network and ensuring reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions described, such as the use of n-butanol and moderate temperatures, are inherently safer and easier to scale from pilot plant to commercial tonnage. The process generates fewer hazardous waste streams compared to traditional methods involving heavy metal oxidants or chlorinated solvents in excessive quantities. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this JAK inhibitor. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the described technology. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or long-term supply agreements.
Q: What are the key starting materials for this JAK inhibitor synthesis?
A: The process utilizes 4-nitropyrazole and 3-oxetanone as primary initial raw materials, which are commercially available and cost-effective.
Q: How does this method improve impurity control compared to traditional routes?
A: By employing mild reaction conditions such as Chan-Lam coupling at 70°C and controlled nucleophilic substitutions, the generation of byproducts is effectively reduced, enhancing overall purity.
Q: Is this synthetic route suitable for industrial production?
A: Yes, the patent explicitly states the route is simple, mature, and suitable for industrial production due to easily obtained raw materials and strong controllability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable JAK Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the development of life-saving therapies. Our team of expert chemists has extensively analyzed the route disclosed in CN111943959A and is fully equipped to execute this chemistry with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry, and our rigorous QC labs guarantee that every batch meets stringent purity specifications required by global regulatory bodies.
We invite you to collaborate with us to optimize this synthesis for your specific needs. Whether you require custom process development to further enhance yields or a dedicated supply of the key intermediates, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals in the oncology and immunology sectors.
