Advanced Metal-Free Synthesis of Isocoumarin Aromatic Ethers for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly isocoumarins, which serve as critical cores in numerous bioactive molecules. Patent CN111253358A introduces a groundbreaking methodology for synthesizing isocoumarin aromatic ether compounds, addressing long-standing challenges in efficiency and environmental impact. This innovation leverages hypervalent iodine chemistry to construct the aryl-oxygen bond, bypassing the need for traditional transition metal catalysts that often plague large-scale manufacturing with toxicity and purification issues. By shifting the paradigm from metal-catalyzed cross-coupling to oxidative coupling, this technology offers a cleaner, more direct route to high-value intermediates. For R&D teams and procurement strategists alike, understanding this shift is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) in API manufacturing. The following analysis details how this novel approach outperforms legacy methods in both technical metrics and commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aromatic ether linkages has relied heavily on transition metal-catalyzed coupling reactions, such as Ullmann or Buchwald-Hartwig type transformations. These conventional processes, while effective in small-scale discovery, introduce significant bottlenecks for commercial production. The primary drawback is the reliance on expensive and often toxic metals like palladium, copper, or nickel, which necessitate rigorous downstream purification to meet stringent regulatory limits for residual metals in drug substances. Furthermore, these reactions frequently require harsh conditions, including high temperatures and strong bases, which can compromise sensitive functional groups on complex molecular scaffolds. The environmental footprint of these methods is also substantial, generating heavy metal waste streams that require specialized disposal protocols, thereby inflating operational costs and complicating environmental compliance.

The Novel Approach
In stark contrast, the methodology disclosed in CN111253358A utilizes a metal-free oxidative coupling strategy mediated by diacetoxyiodobenzene (DAIB). This approach fundamentally alters the reaction landscape by replacing transition metals with a hypervalent iodine oxidant, which acts as a mild and selective electrophile. The reaction proceeds under remarkably mild conditions, typically at room temperature or slightly elevated temperatures, preserving the integrity of sensitive substituents on the isocoumarin ring. This not only simplifies the operational protocol but also drastically reduces the formation of side products associated with thermal degradation. The result is a streamlined process that delivers the target isocoumarin aromatic ethers with exceptional purity and yield, effectively eliminating the need for expensive metal scavengers and complex workup procedures.
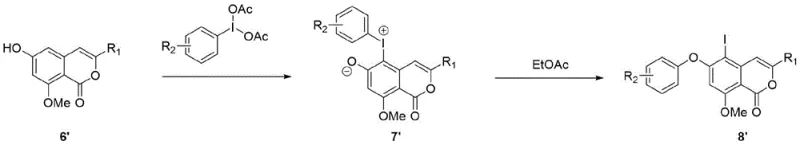
Mechanistic Insights into Hypervalent Iodine Oxidative Coupling
The core of this technological advancement lies in the unique reactivity of the lambda-3-iodane species generated in situ. When the phenolic isocoumarin precursor reacts with DAIB, it forms a reactive aryliodonium intermediate. This intermediate is highly electrophilic and susceptible to nucleophilic attack by the solvent or internal nucleophiles, facilitating the formation of the C-O bond with high regioselectivity. Unlike radical-based metal catalysis which can lead to indiscriminate coupling, this ionic mechanism ensures that the etherification occurs specifically at the desired position on the aromatic ring. The subsequent thermal rearrangement step, conducted in ethyl acetate at 60°C, drives the conversion of the iodonium salt into the stable aromatic ether product, releasing iodobenzene as a benign byproduct that is easily separated. This mechanistic clarity allows for precise control over the reaction trajectory, minimizing the generation of difficult-to-remove impurities.
From an impurity control perspective, the absence of transition metals is a game-changer for pharmaceutical quality assurance. Metal-catalyzed reactions often generate metal-containing organic impurities that are structurally similar to the product and difficult to purge. By utilizing an organic oxidant like DAIB, the impurity profile is significantly simplified, consisting primarily of organic byproducts that can be removed via standard crystallization or chromatography. This leads to a much cleaner crude product, reducing the burden on downstream purification units. For process chemists, this means fewer development cycles are needed to define control strategies for genotoxic impurities or residual metals, accelerating the timeline from lab bench to pilot plant. The high yields reported, such as 93% for the coupling step and 95% for the rearrangement, further attest to the efficiency of this mechanistic pathway.
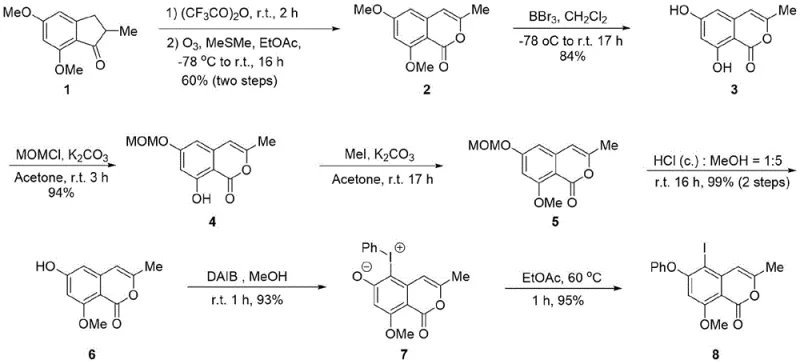
How to Synthesize Isocoumarin Aromatic Ethers Efficiently
Implementing this synthesis requires careful attention to the preparation of the phenolic precursor and the stoichiometry of the oxidant. The process begins with the construction of the isocoumarin core, followed by selective deprotection to reveal the phenol necessary for coupling. The key oxidative step is performed in anhydrous methanol to ensure the stability of the hypervalent iodine species. Following the coupling, a thermal treatment in ethyl acetate completes the transformation. This two-stage sequence is robust and forgiving, making it ideal for transfer to manufacturing suites. For a comprehensive breakdown of the specific experimental parameters, reagent grades, and workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Preparation of the isocoumarin precursor involves protecting group manipulation and ozonolysis to establish the core lactone structure.
- Oxidative coupling is performed by reacting the phenolic isocoumarin intermediate with diacetoxyiodobenzene (DAIB) in anhydrous methanol at room temperature.
- Thermal rearrangement of the iodonium intermediate in ethyl acetate at 60°C yields the final aromatic ether product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this metal-free methodology represents a significant opportunity for cost optimization and risk mitigation. Traditional metal-catalyzed routes are vulnerable to supply chain disruptions related to precious metal availability and price volatility. By eliminating the dependency on palladium or copper catalysts, this process insulates the manufacturing budget from fluctuations in the commodities market. Additionally, the simplification of the purification process translates directly into reduced processing time and lower consumption of specialized scavenging resins. The use of common, commodity-grade solvents like methanol and ethyl acetate further enhances the economic attractiveness of this route, as these materials are readily available globally and do not require specialized handling or storage infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost driver from the bill of materials. Furthermore, the high yields achieved in each step minimize raw material waste, maximizing the output per kilogram of input. The simplified workup procedure reduces the consumption of silica gel and solvents during purification, leading to substantial savings in waste disposal and utility costs. Overall, the process economics are improved through a combination of higher efficiency and lower input costs.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including diacetoxyiodobenzene and standard organic solvents, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metals make this process inherently safer and easier to scale from pilot to commercial production. The reduced environmental burden aligns with modern green chemistry principles, facilitating easier permitting and regulatory approval. The benign nature of the byproducts simplifies waste treatment, allowing for more sustainable manufacturing practices that meet increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this route for their own portfolios. Understanding these details is essential for assessing the feasibility of adopting this method for large-scale production.
Q: Why is this metal-free synthesis preferred for pharmaceutical intermediates?
A: Traditional methods rely on transition metal catalysts like palladium or copper, which leave toxic residues requiring costly removal steps. This hypervalent iodine method eliminates heavy metals entirely, simplifying purification and ensuring compliance with strict ICH Q3D guidelines for elemental impurities.
Q: What are the typical yields for this synthetic route?
A: The patent data indicates exceptionally high yields for the key transformation steps. The oxidative coupling step achieves a 93% yield, and the subsequent thermal rearrangement proceeds with a 95% yield, demonstrating robust efficiency suitable for industrial production.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the process utilizes mild reaction conditions (room temperature to 60°C) and common solvents like methanol and ethyl acetate. The absence of sensitive metal catalysts and the high stability of the intermediates make this route highly amenable to scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isocoumarin Aromatic Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the one described in CN111253358A to enhance product quality and process efficiency. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our state-of-the-art facilities are equipped to handle hypervalent iodine chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality standards for every batch produced.
We invite you to leverage our technical expertise to optimize your supply chain for isocoumarin derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this metal-free technology can drive value for your organization.
