Advanced Green Synthesis of 3-Thioether Indoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing privileged scaffolds, among which the indole nucleus stands out as a cornerstone for drug discovery. Patent CN112409236B presents a significant technological breakthrough in the field of organic synthesis by disclosing a highly selective method for preparing 3-thioether indoles. This innovation addresses the longstanding challenges associated with traditional sulfuration techniques, which often suffer from harsh reaction conditions, poor atom economy, and the generation of hazardous waste. By leveraging a novel oxidative coupling strategy involving sulfonyl hydrazides, this technology enables the efficient construction of carbon-sulfur bonds at the C3 position of the indole ring. For R&D directors and process chemists, this represents a pivotal shift towards greener, more sustainable manufacturing protocols that align with modern regulatory standards. The ability to access diverse 3-substituted indole derivatives with high purity and yield is critical for the development of therapeutic agents targeting cardiovascular diseases, cancer, and other serious conditions, making this patent a valuable asset for any organization focused on high-purity pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-thioether indoles has relied heavily on methods that impose significant operational and environmental burdens on manufacturing facilities. Prior art, such as the technology disclosed in Chinese patent CN201110411943.X, necessitates cryogenic conditions as low as -30°C, which drastically increases energy consumption and requires specialized refrigeration equipment that is costly to maintain on an industrial scale. Furthermore, these legacy processes often depend on large volumes of toxic polar aprotic solvents like N,N-dimethylformamide (DMF), creating severe challenges for solvent recovery and waste disposal. Another common approach involves the use of aryl diphenyl disulfides as sulfur sources, as seen in patent CN201310204244.7, which typically requires prolonged heating at high temperatures for 9 to 15 hours. These disulfide-based routes are frequently plagued by the unpleasant odor of sulfur compounds, posing safety risks to personnel and requiring extensive ventilation systems. The reliance on such苛刻 conditions not only inflates the cost of goods sold but also limits the substrate scope, making it difficult to synthesize complex derivatives needed for advanced drug candidates without compromising yield or selectivity.
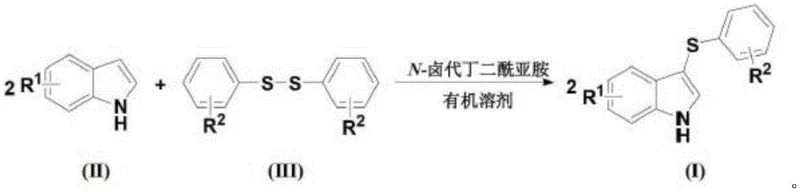
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN112409236B introduces a streamlined and environmentally benign pathway that utilizes sulfonyl hydrazides as the sulfur source. This innovative approach operates under significantly milder conditions, with reaction temperatures ranging from 60°C to 100°C, eliminating the need for energy-intensive cryogenic cooling. The use of absolute ethanol as the reaction solvent marks a substantial improvement in green chemistry metrics, replacing hazardous solvents with a safer, bio-based alternative that is easier to recycle and dispose of. Crucially, this method achieves high regioselectivity for the C3 position of the indole ring without the need for directing groups or protecting group manipulations, thereby shortening the synthetic sequence and improving overall process efficiency. The reaction system is remarkably simple, requiring only the mixing of indole, sulfonyl hydrazide, a bromine source, and an oxidant, which facilitates easy scale-up from laboratory benchtop to commercial production vessels. This simplicity translates directly into reduced operational complexity and lower capital expenditure for manufacturing plants aiming to produce reliable pharmaceutical intermediates.
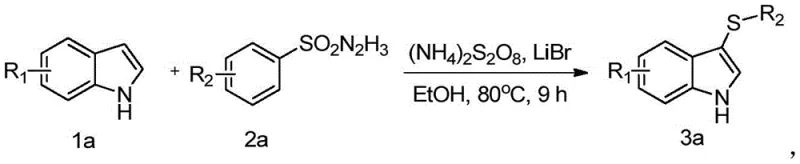
Mechanistic Insights into Ammonium Persulfate-Mediated Oxidative Coupling
The success of this synthetic route hinges on a sophisticated interplay between the oxidant and the bromine source, which together facilitate a unique radical or electrophilic substitution mechanism. Detailed mechanistic studies within the patent reveal that ammonium persulfate acts not merely as a terminal oxidant but plays a critical promotional role in the self-coupling of the sulfonyl hydrazide species. This initial activation step generates a reactive sulfur-containing intermediate that is primed for nucleophilic attack. Simultaneously, the presence of lithium bromide is found to be decisive, as it likely generates an active brominating species in situ that activates the electron-rich C3 position of the indole nucleus. This dual-catalyst system ensures that the reaction proceeds with exceptional specificity, avoiding the formation of polysubstituted byproducts or oxidation of the indole nitrogen that often plagues similar oxidative transformations. The synergy between the persulfate and the bromide ion creates a highly reactive environment that drives the reaction to completion within 8 to 10 hours, a timeframe that is competitive with much harsher thermal processes. Understanding this mechanism allows process chemists to fine-tune reagent ratios, specifically maintaining the molar ratio of indole to sulfonyl hydrazide between 1:1 and 1:2, to maximize conversion while minimizing raw material waste.
From an impurity control perspective, the mechanism offers inherent advantages that simplify downstream purification and ensure the delivery of high-purity OLED material or API precursors. Because the reaction avoids the use of transition metal catalysts such as palladium or copper, there is no risk of heavy metal contamination in the final product, which is a stringent requirement for pharmaceutical regulatory filings. The absence of metal residues eliminates the need for expensive and time-consuming scavenging steps, such as treatment with activated carbon or silica-bound thiols, which are standard in cross-coupling reactions. Furthermore, the high regioselectivity observed implies that isomeric impurities, such as 2-substituted indoles, are formed in negligible quantities, reducing the burden on chromatographic separation. The stability of the sulfonyl hydrazide starting materials also contributes to a cleaner reaction profile, as they do not decompose into volatile sulfur gases that could complicate reactor headspace management. This clean reaction profile is essential for maintaining consistent batch-to-batch quality, a key metric for supply chain heads managing the commercial scale-up of complex polymer additives or fine chemicals.
How to Synthesize 3-Thioether Indole Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of a homogeneous mixed solution containing the indole substrate, the specific sulfonyl hydrazide derivative, and the lithium bromide promoter dissolved in ethanol. Once the mixture is uniform, the oxidant, ammonium persulfate, is introduced, and the system is heated to the target temperature range. It is critical to monitor the reaction progress to determine the optimal endpoint, typically falling within the 8 to 10-hour window, to prevent over-oxidation. Following the reaction, the workup procedure involves standard unit operations including drying, solvent removal via concentration, and final purification using column chromatography with an ethyl acetate and petroleum ether gradient. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Prepare a mixed solution by combining the indole substrate, sulfonyl hydrazide compound, and lithium bromide in absolute ethanol.
- Add ammonium persulfate as the oxidant to the mixture and heat the reaction system to a temperature range of 60-100°C.
- Maintain heating for 8-10 hours, then proceed with drying, concentration, and column chromatography purification to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of transition metal catalysts represents a direct reduction in raw material costs, as precious metals like palladium are not only expensive to purchase but also incur significant costs for recovery and disposal. By utilizing commodity chemicals such as ammonium persulfate and lithium bromide, the process insulates manufacturers from the volatility of the precious metals market, ensuring more predictable budgeting and cost reduction in pharmaceutical intermediate manufacturing. Additionally, the use of ethanol as a primary solvent simplifies the supply chain logistics, as it is widely available, inexpensive, and does not require the special handling permits associated with chlorinated or amide solvents. This shift to greener solvents also reduces the environmental compliance burden, potentially lowering waste treatment fees and insurance premiums associated with hazardous chemical storage. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or mixing, leading to higher first-pass yields and reduced batch failure rates.
- Cost Reduction in Manufacturing: The economic advantage of this method is primarily driven by the substitution of expensive and toxic reagents with cost-effective alternatives. Traditional methods often require stoichiometric amounts of strong bases or specialized organometallic reagents, whereas this protocol utilizes catalytic amounts of lithium bromide and inexpensive oxidants. The avoidance of cryogenic conditions significantly lowers utility costs, as maintaining reactors at -30°C requires substantial energy input compared to heating at 80°C. Furthermore, the simplified workup procedure reduces the consumption of silica gel and eluents during purification, contributing to substantial cost savings in consumables. These factors combine to create a leaner manufacturing process that improves the overall margin profile for high-volume production runs without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reagents required for this synthesis are globally available commodity chemicals with stable supply chains. Sulfonyl hydrazides are stable solids that can be stored for extended periods without degradation, unlike liquid thiols or disulfides which may oxidize or emit odors over time. This stability allows for bulk purchasing strategies that further drive down unit costs and mitigate the risk of supply disruptions. The use of ethanol also ensures that solvent availability is never a bottleneck, as it is produced at massive scales for various industries. By reducing dependence on niche reagents or custom-synthesized catalysts, procurement teams can secure long-term supply agreements with greater confidence, ensuring continuous production schedules for critical drug intermediates.
- Scalability and Environmental Compliance: The scalability of this process is supported by its straightforward exothermic profile and the use of a low-boiling point solvent that is easy to recover. The absence of heavy metals simplifies the environmental impact assessment, making it easier to obtain necessary permits for plant expansion or modification. Waste streams generated from this process are less hazardous, facilitating easier treatment and disposal in accordance with increasingly strict environmental regulations. The high atom economy of the reaction means that less raw material ends up as waste, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing operation. This environmental compatibility makes the technology future-proof against tightening global regulations on chemical emissions and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to assist technical teams in evaluating feasibility. Understanding these nuances is vital for successful technology transfer and process validation.
Q: Why is sulfonyl hydrazide preferred over traditional disulfides for indole sulfuration?
A: Sulfonyl hydrazides are odorless, stable solids that do not require external reducing agents due to the presence of the hydrazine group, unlike malodorous and less stable disulfides used in conventional methods.
Q: What are the critical reaction conditions for high regioselectivity in this process?
A: The simultaneous presence of ammonium persulfate and lithium bromide is crucial; the temperature must be strictly controlled between 60-100°C to prevent side reactions and ensure exclusive C3-position functionalization.
Q: How does this method improve environmental compliance compared to prior art?
A: This protocol eliminates the need for toxic polar aprotic solvents like DMF or DMSO and avoids heavy metal catalysts, utilizing ethanol as a green solvent which simplifies waste treatment and reduces environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Thioether Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensively evaluated the technology described in CN112409236B and is fully prepared to leverage this methodology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-thioether indole delivered meets the highest international standards for pharmaceutical applications. We are committed to providing a reliable pharmaceutical intermediate supplier partnership that prioritizes quality, consistency, and technical excellence.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener protocol for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your upcoming projects. Let us collaborate to optimize your supply chain and accelerate the delivery of life-saving medicines to the market through superior chemical manufacturing solutions.
