Advancing DNA-Encoded Library Synthesis with Robust On-DNA Mannich Reaction Technology
The landscape of drug discovery has been revolutionized by DNA-encoded library (DEL) technology, allowing for the screening of billions of compounds against therapeutic targets with unprecedented speed. However, the utility of DELs is strictly bounded by the chemical reactions compatible with the fragile DNA tag. Patent CN112779606A, filed in May 2021, addresses a critical gap in this field by disclosing a novel method for performing On-DNA Mannich reactions. This innovation enables the direct construction of beta-aminocarbonyl compounds, a privileged scaffold prevalent in bioactive natural products and pharmaceutical agents, directly on the DNA strand. Unlike traditional solution-phase synthesis which often relies on harsh conditions incompatible with nucleic acids, this patented methodology operates efficiently in mixed aqueous-organic solvents under mild conditions. For R&D directors and procurement specialists in the fine chemical sector, this represents a significant leap forward in expanding the chemical diversity of encoded libraries without compromising the integrity of the genetic barcode.
The implementation of this technology allows for the large-scale introduction of aldehydes, amines, and active hydrogen compounds as modular building blocks. This modularity is essential for the combinatorial explosion required in modern hit identification campaigns. By validating the feasibility of three-component coupling on DNA, the patent provides a robust pathway for generating complex small molecules that were previously inaccessible via standard DEL chemistry. The ability to perform these reactions in multi-well plates further underscores its suitability for high-throughput automation, a key requirement for industrial-scale library synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-aminocarbonyl compounds via the Mannich reaction has been a staple in organic chemistry, yet translating this to an On-DNA format has proven exceptionally difficult. Conventional Mannich protocols typically necessitate strong Lewis acids, Brønsted acids, or elevated temperatures that pose a severe threat to the stability of the DNA oligonucleotide. The phosphodiester backbone of DNA is susceptible to hydrolysis under acidic or basic extremes, and the nucleobases can undergo depurination or alkylation, leading to library corruption and loss of encoding information. Furthermore, many traditional catalysts are insoluble in the aqueous buffers required to maintain DNA solubility, resulting in heterogeneous reaction conditions that lead to poor conversion rates and inconsistent product quality. These limitations have historically restricted the chemical space of DELs, forcing chemists to rely on a narrower set of biocompatible reactions that may not access the most pharmacologically relevant scaffolds.
The Novel Approach
The methodology described in CN112779606A overcomes these barriers by utilizing a carefully optimized solvent system comprising water mixed with organic co-solvents such as DMSO, DMA, or acetonitrile. This biphasic-like environment ensures the solubility of organic building blocks while maintaining the hydration shell necessary for DNA stability. The patent demonstrates that the reaction can proceed efficiently at room temperature or slightly elevated temperatures (up to 100°C in specific buffered conditions) without degrading the DNA tag. Crucially, the method tolerates a wide range of catalysts, including organocatalysts like L-proline and mild acids like p-toluenesulfonic acid, which facilitate the formation of the reactive imine intermediate and subsequent nucleophilic attack without damaging the genetic material. This approach effectively decouples the harshness of the chemical transformation from the fragility of the biological tag.
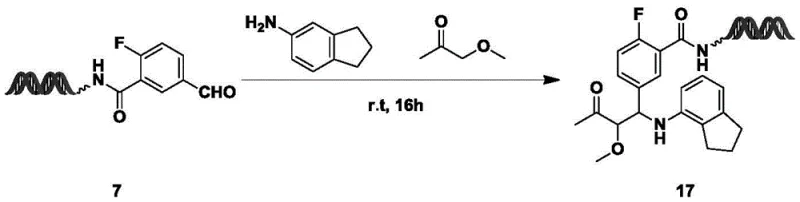
As illustrated in the reaction schemes provided in the patent, such as the synthesis of Compound 17 shown above, the process achieves remarkable conversion rates, reaching up to 98% in optimized examples. This high efficiency is critical for DEL synthesis, where low yields can result in the loss of valuable library members during the enrichment and sequencing phases. The ability to use diverse amines, including anilines and cyclic amines, alongside various ketones and esters, confirms the broad substrate scope of this novel approach. This flexibility allows medicinal chemists to explore structure-activity relationships (SAR) more comprehensively by incorporating varied steric and electronic properties into the beta-aminocarbonyl core.
Mechanistic Insights into On-DNA Multicomponent Coupling
The mechanistic pathway of this On-DNA Mannich reaction follows a classic condensation-addition sequence but is adapted for the unique constraints of the DNA conjugate. Initially, the On-DNA aldehyde reacts with the amine component to form an imine or iminium ion intermediate. In the context of the patent, this step is facilitated by the specific pH of the buffer systems used, such as MES or PBS, which optimize the nucleophilicity of the amine while preventing DNA degradation. The presence of catalysts like L-proline can further accelerate this step by forming a transient enamine or activating the carbonyl electrophile. Once the imine is generated, the active hydrogen compound (such as a ketone or beta-keto ester) undergoes tautomerization to its enol or enolate form. This nucleophilic species then attacks the electrophilic carbon of the imine, forming the new carbon-carbon bond that characterizes the beta-aminocarbonyl product.
Impurity control in this system is inherently managed by the mild reaction conditions and the specificity of the catalyst. Harsh conditions often lead to side reactions such as aldol condensations of the ketone component or hydrolysis of the DNA linker. By operating at near-neutral pH or using mild organic acids, the patent minimizes these competing pathways. Furthermore, the use of excess reagents (10 to 1000 equivalents) drives the equilibrium towards the desired product, ensuring high conversion even with the steric hindrance potentially imposed by the bulky DNA tag. The purification strategy outlined, involving ethanol precipitation, effectively removes unreacted small molecules and salts while retaining the high molecular weight DNA-conjugated product, ensuring a clean impurity profile suitable for downstream screening applications.
How to Synthesize On-DNA Beta-Aminocarbonyl Compounds Efficiently
The synthesis protocol detailed in the patent offers a straightforward, two-stage procedure that is highly amenable to automation. The process begins with the preparation of the On-DNA aldehyde, which serves as the anchor for the library diversification. This substrate is dissolved in a buffered solution, creating the foundation for the multicomponent coupling. The reaction is designed to be operationally simple, requiring only the sequential addition of reagents without the need for complex inert atmosphere techniques or specialized equipment beyond standard laboratory shakers and centrifuges. This simplicity is a key factor in its potential for widespread adoption in both academic and industrial settings.
- Prepare the On-DNA aldehyde substrate solution in a suitable buffer such as MES or PBS at a concentration of 0.5-5mM.
- Add the amine component (10-1000 equivalents) and optional catalyst (0-100 equivalents) to the aldehyde solution and react for 0.5-2 hours to form the imine intermediate.
- Introduce the active hydrogen compound (10-1000 equivalents) and allow the mixture to react at 5-100°C for 1-24 hours to complete the Mannich addition.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this On-DNA Mannich reaction technology presents compelling economic and logistical advantages. The primary value driver is the significant reduction in process complexity compared to alternative methods for generating similar scaffolds. Traditional routes might require protecting group strategies or multi-step sequences to install the beta-amino ketone motif prior to DNA conjugation, which increases material costs and reduces overall throughput. By enabling a direct, one-pot construction on the DNA, this method drastically simplifies the synthetic route, thereby reducing the consumption of expensive DNA starting materials and minimizing the labor hours required for synthesis and purification.
- Cost Reduction in Manufacturing: The elimination of harsh reagents and the use of commercially available, commodity-grade building blocks (aldehydes, amines, ketones) significantly lowers the raw material cost profile. Furthermore, the high conversion rates reported in the patent examples mean that less starting material is wasted, improving the overall atom economy of the library synthesis. The avoidance of transition metal catalysts, which often require expensive removal steps to prevent interference with biological assays, further contributes to cost savings by streamlining the downstream processing workflow.
- Enhanced Supply Chain Reliability: The reagents required for this protocol are standard fine chemicals that are widely available from multiple global suppliers, mitigating the risk of supply chain disruptions associated with proprietary or exotic catalysts. The robustness of the reaction in aqueous buffers also reduces the dependency on specialized anhydrous solvents, simplifying inventory management and storage requirements. This reliability ensures consistent production schedules for large-scale library generation projects, allowing R&D teams to adhere to tight timelines for hit identification campaigns.
- Scalability and Environmental Compliance: The use of water as a major component of the solvent system aligns with green chemistry principles, reducing the volume of hazardous organic waste generated during synthesis. This environmental benefit translates to lower waste disposal costs and simplifies regulatory compliance for manufacturing facilities. Additionally, the compatibility of the method with multi-well plate formats indicates high scalability; the process can be easily parallelized to synthesize thousands of compounds simultaneously, supporting the massive scale required for modern drug discovery pipelines without a linear increase in operational overhead.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this On-DNA Mannich reaction technology. These insights are derived directly from the experimental data and specifications provided in patent CN112779606A, offering clarity on the practical application of this method in a production environment. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into existing DEL platforms.
Q: Why is the On-DNA Mannich reaction challenging compared to solution-phase chemistry?
A: Traditional Mannich reactions often require harsh acidic or basic conditions and organic solvents that can degrade the DNA tag or hydrolyze the phosphate backbone. This patent solves this by utilizing mild aqueous-organic mixed solvents and specific catalysts like L-proline that preserve DNA integrity while driving the reaction.
Q: What is the substrate scope for this On-DNA reaction method?
A: The method demonstrates broad compatibility with various On-DNA aldehydes (aryl, heteroaryl), diverse amines (anilines, alkyl amines), and active hydrogen compounds (ketones, esters). This versatility allows for the rapid generation of large, diverse libraries for high-throughput screening.
Q: How does this method improve library diversity for drug discovery?
A: By enabling the efficient construction of beta-aminocarbonyl scaffolds directly on DNA, this method introduces a privileged structure found in many natural products and drugs into DNA-encoded libraries. This significantly expands the chemical space available for screening against biological targets.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable On-DNA Beta-Aminocarbonyl Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic patents into commercially viable chemical solutions. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative On-DNA Mannich reaction described in CN112779606A can be seamlessly integrated into your supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for DNA-encoded library intermediates, guaranteeing that every batch delivered supports the integrity of your screening data.
We invite pharmaceutical and biotech companies to leverage our expertise for their next-generation library synthesis projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific scaffold requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your transition to this advanced chemistry is smooth, efficient, and economically optimized for long-term success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
