Revolutionizing Alpha-Cyano-Beta-Amino Acrylate Production via Green Ammonium Salt Catalysis
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing complex intermediates, particularly those serving as precursors for biologically active compounds. Patent CN114874109A introduces a groundbreaking methodology for the preparation of novel alpha-cyano-beta-amino acrylate compounds, addressing critical limitations in current synthetic protocols. This technology leverages a unique Knoevenagel-Michael cascade reaction catalyzed by ammonium or amine salts, which function dually as both the catalyst and the nitrogen source. By operating under neutral conditions in a mixed solvent system of dimethyl sulfoxide (DMSO) and phosphate buffer, this process eliminates the need for hazardous strong acids or bases, offering a significantly greener alternative for producing high-purity pharmaceutical intermediates. The implications for supply chain stability and environmental compliance are profound, as this route minimizes toxic waste generation while maintaining exceptional product yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-amino ester compounds, which are vital precursors for gene delivery vectors and anticancer drug carriers, has relied heavily on the Mannich reaction or Michael addition reactions catalyzed by Lewis acids. Traditional protocols often necessitate the use of corrosive and toxic catalysts such as ZrOCl2·8H2O or CdCl2, which pose significant environmental hazards and require complex post-reaction purification to remove heavy metal residues. Furthermore, these conventional methods frequently demand harsh reaction conditions, including extreme pH levels that can lead to the formation of undesirable by-products and compromise the structural integrity of sensitive functional groups. The reliance on excessive alkali or strong acidic activators not only increases the cost of raw materials but also creates a substantial burden on wastewater treatment facilities, making these processes increasingly untenable in the context of modern green chemistry regulations and corporate sustainability goals.
The Novel Approach
In stark contrast, the methodology disclosed in CN114874109A represents a paradigm shift by utilizing inexpensive and readily available ammonium or amine salts, such as ammonium sulfate or methylamine hydrochloride, to drive the reaction. This innovative approach operates effectively at mild temperatures ranging from 40°C to 50°C within a neutral pH environment, thereby circumventing the corrosion issues and safety risks associated with traditional Lewis acid catalysis. The reaction system employs a specific ratio of organic solvent to aqueous buffer (7:3 DMSO to PB buffer), which facilitates the solubility of diverse substrates while maintaining the stability of the catalytic species. This results in a streamlined workflow where the catalyst is integrated into the product structure or easily separated, drastically reducing the complexity of the workup procedure and enhancing the overall atom economy of the synthesis.
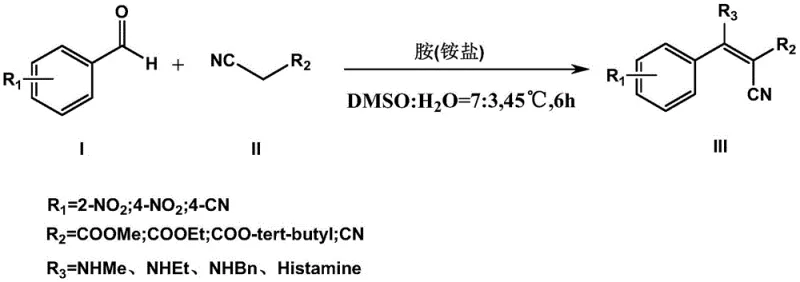
The mechanistic elegance of this process lies in its ability to expand the substrate scope significantly without compromising yield or purity. As illustrated in the reaction scheme, the method accommodates a wide variety of aromatic aldehydes, including those with electron-withdrawing groups like nitro and cyano substituents at ortho or para positions. Additionally, the active methylene component can be varied between methyl, ethyl, and tert-butyl cyanoacetates, as well as malononitrile, allowing for the modular construction of diverse alpha-cyano-beta-amino acrylate derivatives. This flexibility is crucial for R&D teams aiming to generate libraries of analogs for structure-activity relationship (SAR) studies, as it provides a reliable platform for rapid iteration and optimization of lead compounds.
Mechanistic Insights into Ammonium Salt-Catalyzed Cascade Reaction
The core of this synthetic breakthrough is the ammonium or amine salt-catalyzed Knoevenagel-Michael cascade reaction, which proceeds through a highly efficient multi-step mechanism under neutral conditions. Initially, the amine species derived from the salt activates the active methylene compound, facilitating the nucleophilic attack on the carbonyl carbon of the aromatic aldehyde to form an intermediate olefin via Knoevenagel condensation. Subsequently, the second equivalent of the amine or ammonium species participates in a Michael addition across the activated double bond, leading to the final alpha-cyano-beta-amino acrylate structure. This cascade sequence is meticulously balanced by the buffer system, which ensures that the proton transfer steps occur smoothly without the accumulation of acidic or basic by-products that could degrade the product or inhibit the catalyst.
From an impurity control perspective, the neutral reaction environment plays a pivotal role in minimizing side reactions such as polymerization or hydrolysis of the ester groups, which are common pitfalls in acid or base-catalyzed variants. The use of DMSO as a co-solvent enhances the polarity of the medium, stabilizing the transition states and ensuring homogeneous mixing of the organic and inorganic components. This results in a cleaner reaction profile with fewer impurities, simplifying the downstream purification process typically involving silica gel column chromatography. The high selectivity observed in this system allows for the isolation of products with excellent purity, meeting the stringent quality standards required for pharmaceutical applications and reducing the need for extensive recrystallization or additional refining steps.
How to Synthesize Alpha-Cyano-Beta-Amino Acrylates Efficiently
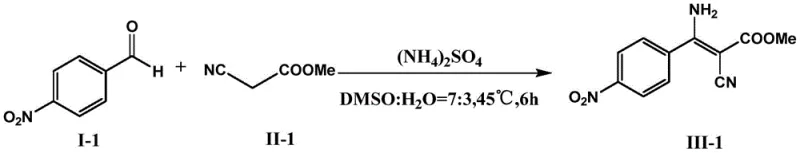
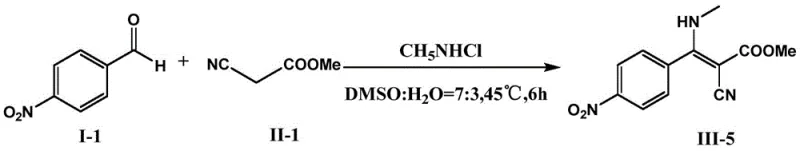
Implementing this synthesis protocol requires precise adherence to the molar ratios and solvent compositions specified in the patent to achieve optimal results. The standard procedure involves combining the aromatic aldehyde, active methylene compound, and the ammonium or amine salt in a reaction vessel containing the DMSO-buffer mixture. The reaction is then agitated at controlled temperatures for a defined period, typically between 4 to 8 hours, depending on the specific substrate reactivity. Following the reaction completion, a simple liquid-liquid extraction with ethyl acetate effectively separates the organic product from the aqueous phase, followed by drying and concentration to yield the crude material. For detailed operational parameters and specific variations for different substrates, please refer to the standardized guide below.
- Mix ammonium/amine salt, aromatic aldehyde, and active methylene compound in a DMSO: PB buffer (7:3) solvent system.
- Shake the reaction mixture at 40-50°C for 4-8 hours under neutral pH conditions.
- Terminate with ethyl acetate extraction, dry over Na2SO4, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive and hazardous Lewis acid catalysts translates directly into a reduction in raw material costs and a simplification of the supply chain, as ammonium salts are commodity chemicals with stable global availability. Furthermore, the absence of toxic heavy metals in the process removes the need for specialized waste disposal contracts and expensive metal scavenging resins, leading to significant operational expenditure savings. The robustness of the reaction at the gram scale, as demonstrated in the patent data, suggests a low risk of failure during technology transfer to pilot or commercial plants, ensuring consistent supply continuity for downstream API manufacturing.
- Cost Reduction in Manufacturing: The substitution of proprietary or high-cost Lewis acid catalysts with ubiquitous ammonium salts drastically lowers the bill of materials for each production batch. Additionally, the simplified workup procedure, which avoids complex neutralization and heavy metal removal steps, reduces labor hours and solvent consumption, contributing to a leaner and more cost-effective manufacturing process that enhances overall margin potential.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials such as substituted benzaldehydes and cyanoacetates, the risk of supply disruption due to single-source dependency is minimized. The mild reaction conditions also reduce the wear and tear on reactor equipment, extending asset life and decreasing maintenance downtime, which collectively strengthens the reliability of the production schedule and ensures timely delivery to customers.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly strict environmental regulations, mitigating the risk of regulatory fines or production halts due to non-compliance. The process generates minimal hazardous waste and operates under safe, non-corrosive conditions, making it inherently easier to scale from laboratory to industrial volumes without requiring massive capital investment in specialized containment or treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their supply chains.
Q: How does this method improve upon traditional Lewis acid catalysis?
A: Unlike traditional methods requiring corrosive Lewis acids or strong bases, this patent utilizes benign ammonium or amine salts under neutral conditions, eliminating toxic waste and simplifying downstream processing.
Q: What is the substrate scope for this synthesis?
A: The method demonstrates broad compatibility with various substituted aromatic aldehydes (e.g., nitro, cyano groups) and active methylene compounds including methyl, ethyl, and tert-butyl cyanoacetates.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly validates the robustness of the reaction at the gram scale (1L reactor), maintaining high yields and stable performance, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyano-Beta-Amino Acrylate Supplier
As the global demand for high-performance pharmaceutical intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies such as the one described in CN114874109A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of alpha-cyano-beta-amino acrylates meets the highest international standards, providing our clients with the confidence needed to advance their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this green synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this ammonium salt-catalyzed method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering value through innovation and operational excellence in the fine chemical sector.
