Transforming Triazole Chemistry: Scalable Production of 5-Trifluoromethyl 1,2,3-Triazoles for Pharmaceutical Applications
Patent CN113121462B introduces a groundbreaking methodology for synthesizing 5-trifluoromethyl substituted 1,2,3-triazole compounds through a novel metal-free approach that eliminates hazardous reagents while maintaining high efficiency. This innovation addresses critical limitations in traditional triazole synthesis by utilizing readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials. The process operates under mild reaction conditions (60°C) with simple workup procedures that significantly enhance operational safety and scalability. Unlike conventional methods requiring toxic azides and transition metal catalysts, this new route provides a sustainable pathway for producing high-purity triazole intermediates essential for pharmaceutical development. The patent demonstrates exceptional versatility across diverse substrate combinations while achieving consistent product quality suitable for commercial manufacturing applications. This advancement represents a significant leap forward in the synthesis of fluorinated heterocyclic compounds with broad implications for drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethyl-substituted triazoles predominantly rely on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides followed by subsequent trifluoromethylation steps. These methods present significant operational hazards due to the inherent instability and explosive nature of organic azide compounds that require specialized handling protocols and stringent safety measures. The multi-step processes often necessitate expensive transition metal catalysts that introduce contamination risks requiring extensive purification procedures to meet pharmaceutical purity standards. Additionally, these conventional approaches exhibit limited substrate scope with poor functional group tolerance that restricts their applicability across diverse molecular architectures. The requirement for specialized trifluoromethyl reagents further complicates supply chain logistics while increasing overall production costs. These combined limitations create substantial barriers to commercial scale-up and hinder the widespread adoption of triazole-based compounds in pharmaceutical manufacturing despite their valuable biological properties.
The Novel Approach
The patented methodology represents a paradigm shift by employing a base-promoted reaction between diazo compounds and trifluoroethylimidoyl chloride that completely eliminates the need for hazardous azides and transition metal catalysts. This innovative approach utilizes cesium carbonate as an economical base promoter in acetonitrile solvent at moderate temperatures (60°C), creating a significantly safer manufacturing environment without compromising reaction efficiency. The process demonstrates remarkable substrate flexibility with broad functional group tolerance that accommodates various aryl and alkyl substituents while maintaining consistent product quality. By avoiding expensive metal catalysts and hazardous reagents, the method substantially reduces purification requirements and eliminates costly heavy metal removal steps from the manufacturing workflow. The simplified reaction pathway enables direct conversion to final products with minimal intermediate handling, thereby enhancing overall process robustness and scalability potential. This streamlined approach delivers superior operational efficiency while meeting the stringent purity requirements essential for pharmaceutical applications.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism proceeds through a base-promoted intermolecular nucleophilic addition where the diazo compound attacks the electrophilic carbon of trifluoroethylimidoyl chloride. This initial step forms a key intermediate through carbon-carbon bond formation that subsequently undergoes intramolecular cyclization via a 5-endo-dig pathway to generate the triazole ring structure. The cesium carbonate base facilitates deprotonation events that drive the reaction forward while maintaining optimal reaction kinetics under mild thermal conditions. This mechanistic pathway avoids radical intermediates or unstable species that typically complicate traditional triazole syntheses. The absence of transition metals prevents potential coordination complexes that could lead to side reactions or impurities. The well-defined reaction trajectory ensures high regioselectivity at the desired positions while minimizing unwanted byproduct formation through controlled stepwise transformations that maintain molecular integrity throughout the process.
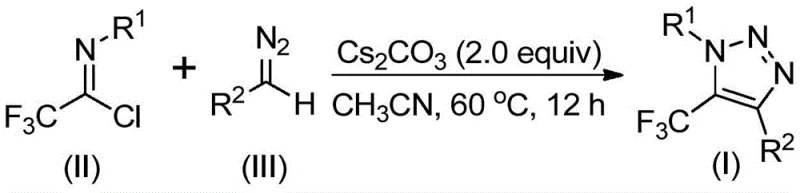
The process inherently minimizes impurity formation through its carefully orchestrated reaction sequence that avoids unstable intermediates common in conventional methods. The absence of transition metals eliminates potential metal-catalyzed side reactions that typically generate difficult-to-remove impurities requiring extensive purification. The mild reaction conditions prevent thermal degradation pathways that could produce decomposition products affecting final product quality. The well-defined mechanistic pathway ensures consistent regioselectivity that maintains structural integrity across diverse substrate combinations. This controlled transformation sequence results in cleaner reaction profiles with fewer byproducts compared to traditional approaches that rely on less predictable radical or metal-mediated pathways. The simplified purification workflow through standard column chromatography yields products meeting pharmaceutical-grade purity specifications without requiring specialized separation techniques.
How to Synthesize 5-Trifluoromethyl 1,2,3-Triazoles Efficiently
This innovative synthesis route offers pharmaceutical manufacturers a practical pathway to produce high-value triazole intermediates with exceptional operational simplicity and safety profile. The methodology leverages commercially available starting materials that eliminate supply chain vulnerabilities associated with hazardous reagents required by conventional approaches. By operating at moderate temperatures with standard laboratory equipment, the process demonstrates excellent compatibility with existing manufacturing infrastructure while delivering superior product quality. The straightforward workup procedure involving filtration and column chromatography ensures consistent batch-to-batch reproducibility essential for regulatory compliance in pharmaceutical production environments. Detailed standardized synthesis steps are provided below to facilitate seamless technology transfer from laboratory to commercial manufacturing settings.
- Prepare the reaction mixture by combining trifluoroethylimidoyl chloride (II), diazo compound (III), and cesium carbonate in acetonitrile solvent under nitrogen atmosphere.
- Heat the reaction mixture to 60°C and maintain for 12 hours with continuous stirring to ensure complete conversion.
- Purify the crude product through filtration, silica gel mixing, and column chromatography to obtain high-purity 5-trifluoromethyl substituted triazole compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing approach delivers substantial value to procurement and supply chain organizations by addressing critical pain points in traditional triazole production methods. The elimination of hazardous materials reduces regulatory compliance burdens while enhancing workplace safety across the manufacturing ecosystem. The use of readily available starting materials creates more resilient supply chains less vulnerable to single-source dependencies or geopolitical disruptions affecting specialized reagent suppliers. These improvements collectively enhance overall operational reliability while positioning manufacturers to better meet evolving regulatory requirements in global pharmaceutical markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes significant material costs while avoiding complex purification steps required to remove metal contaminants from final products. This streamlined approach reduces overall processing time by eliminating multiple intermediate steps common in conventional syntheses, resulting in substantial cost savings through reduced labor requirements and lower utility consumption per production batch without requiring specialized equipment or infrastructure investments.
- Enhanced Supply Chain Reliability: Utilizing commercially available starting materials creates more robust supply chains with multiple sourcing options that reduce vulnerability to single-supplier dependencies. The absence of hazardous reagents eliminates special handling requirements and transportation restrictions that typically complicate logistics planning and increase lead times. This improved material availability profile enables more predictable production scheduling while reducing inventory holding costs associated with safety stock requirements for unstable or regulated compounds.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward purification process enable seamless scale-up from laboratory to commercial production volumes without requiring specialized equipment modifications. The elimination of heavy metals from the manufacturing process significantly reduces environmental impact by avoiding toxic waste streams that require expensive treatment protocols. This environmentally friendly approach aligns with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives through reduced waste generation and lower energy consumption per unit produced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this innovative triazole synthesis methodology based on detailed patent analysis and practical manufacturing considerations. These insights reflect our extensive experience in scaling similar complex organic transformations for global pharmaceutical clients.
Q: How does this method eliminate the need for hazardous azide compounds?
A: The novel process utilizes diazo compounds and trifluoroethylimidoyl chloride as starting materials instead of toxic azides, significantly improving workplace safety while maintaining high reaction efficiency.
Q: What are the key advantages of this metal-free synthesis approach?
A: This method eliminates transition metal catalysts and associated purification steps, reducing production costs and ensuring higher purity levels required for pharmaceutical applications without heavy metal contamination risks.
Q: How does this process support scalable manufacturing for commercial production?
A: The reaction operates under mild conditions (60°C) with readily available reagents and straightforward purification, enabling seamless scale-up from laboratory to industrial production volumes while maintaining consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl 1,2,3-Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex fluorinated heterocycles while maintaining stringent purity specifications required by global regulatory authorities. With state-of-the-art facilities featuring rigorous QC labs equipped for comprehensive analytical testing including advanced NMR spectroscopy and mass spectrometry capabilities, we ensure consistent product quality across all production scales. Our technical team possesses deep expertise in optimizing triazole-based syntheses to meet specific client requirements while navigating complex regulatory landscapes across multiple jurisdictions worldwide.
Leverage our specialized knowledge through a Customized Cost-Saving Analysis tailored to your specific manufacturing needs. Contact our technical procurement team today to request detailed COA data and comprehensive route feasibility assessments that demonstrate how our innovative approach can enhance your supply chain resilience while delivering superior product quality for your pharmaceutical development programs.
