Optimized Synthesis of N-Cyanomethylbis Trifluoromethyl Nicotinamide for Commercial Scale-Up
The global agrochemical sector demands rigorous quality control standards, particularly for potent insecticides like flonicamid, where impurity profiling is critical for regulatory compliance and efficacy. Patent CN107286086B introduces a groundbreaking preparation method for N-cyanomethylbis(trifluoromethyl)nicotinamide, a pivotal impurity standard used to monitor flonicamid synthesis. This compound is not merely a byproduct but a essential analytical tool that ensures the safety and consistency of the final pesticide formulation. The disclosed technology addresses the historical challenges of low yield and poor purity associated with conventional synthesis routes, offering a robust pathway for producing high-grade reference materials. By leveraging a specific three-step reaction sequence involving hydrolysis, hydrogenolysis, and amidation, manufacturers can now achieve unprecedented consistency in producing this complex pyridine derivative. This advancement supports the broader supply chain by ensuring that quality control laboratories have access to reliable standards, thereby facilitating smoother regulatory approvals and market entry for agrochemical products worldwide.
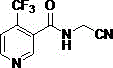
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nicotinamide derivatives often relied on direct acyl chlorination methods which, while straightforward in theory, suffer from significant practical drawbacks in an industrial setting. Traditional approaches frequently utilize thionyl chloride for activation, leading to reaction mixtures that are difficult to purify and often result in dark-colored crude products requiring extensive downstream processing. These conventional routes typically struggle to control side reactions, leading to the formation of unknown impurities that complicate the analytical validation of the final insecticide. Furthermore, the yields associated with these older methodologies are often suboptimal, frequently hovering around 43.5%, which represents a substantial loss of valuable starting materials and increased production costs. The inability to effectively recycle solvents or intermediates in these legacy processes exacerbates environmental concerns and drives up the overall cost of goods sold, making them less attractive for large-scale commercial manufacturing where efficiency is paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated multi-step strategy that prioritizes both yield optimization and impurity control through precise reaction engineering. By starting with 2,6-dichloro-3-cyano-4-trifluoromethylpyridine and subjecting it to controlled nitrile hydrolysis followed by catalytic hydrogenolysis, the process establishes a clean intermediate profile before the final amidation step. A key innovation lies in the treatment of the amidation reaction filtrate, which is subjected to hot filtration and cooling to recover valuable components that are recycled back into the process as reaction solvents. This closed-loop system not only minimizes waste generation but also capitalizes on unreacted starting materials present in the mother liquor, effectively boosting the overall material efficiency of the plant. The result is a streamlined manufacturing protocol that consistently delivers yields between 71.1% and 85.7% with purity levels exceeding 99.1%, representing a quantum leap over previous technological benchmarks.
Mechanistic Insights into Pyridine Derivative Functionalization
The chemical transformation begins with the hydrolysis of the nitrile group, a critical step where the choice of base and solvent ratio dictates the success of the entire sequence. Using a potassium hydroxide solution with a concentration of 11.2% to 22.4% under reflux conditions ensures complete conversion to the picolinic acid intermediate while minimizing the formation of carboxamide byproducts. Following this, the hydrogenolysis step employs a palladium on carbon catalyst in the presence of triethylamine to selectively remove chlorine atoms without affecting the sensitive trifluoromethyl group. This step requires careful control of hydrogen pressure and temperature to prevent over-reduction or catalyst poisoning, ensuring that the resulting 4-trifluoromethyl nicotinic acid is obtained in high purity. The precision in these early stages is vital, as any residual impurities carried forward would be amplified in the final amidation reaction, compromising the integrity of the analytical standard.
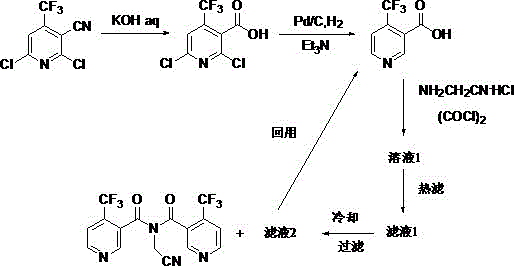
The final amidation reaction represents the most complex mechanistic challenge, requiring the activation of the carboxylic acid using oxalyl chloride in the presence of aminoacetonitrile hydrochloride. The protocol specifies a molar ratio of oxalyl chloride to acid between 1.05:1 and 1.1:1 to ensure complete activation while avoiding excess reagent that could lead to side reactions. Temperature control is equally critical, with the reaction initiated at low temperatures (-20°C to 0°C) to manage exotherms and then heated to 95-110°C to drive the coupling to completion. The subsequent workup involves a strategic hot filtration to remove organic salts, followed by controlled cooling crystallization that leverages the solubility differences to isolate the target molecule. This meticulous attention to thermodynamic parameters ensures that the final product meets the stringent purity requirements necessary for its role as a calibration standard in high-performance liquid chromatography.
How to Synthesize N-Cyanomethylbis(Trifluoromethyl)Nicotinamide Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction conditions and material ratios to replicate the high yields and purity reported in the patent literature. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to pilot plant operations with minimal re-optimization required. Operators must focus on the precise preparation of the aminoacetonitrile solution and the controlled addition of the chlorinating agent to maintain reaction homogeneity and safety. Detailed standard operating procedures regarding the recycling of filtrate 2 are essential, as this step is the primary driver of the process economic advantages and environmental benefits. For a comprehensive breakdown of the specific equipment requirements, safety protocols, and step-by-step operational guidelines, please refer to the technical documentation provided below.
- Perform nitrile group hydrolysis on 2,6-dichloro-3-cyano-4-trifluoromethylpyridine using KOH solution under reflux.
- Execute hydrogenolysis on the resulting acid intermediate using Pd/C catalyst and triethylamine in methanol.
- Conduct amidation with aminoacetonitrile hydrochloride and oxalyl chloride, recycling the mother liquor filtrate for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this patented synthesis method offers substantial strategic advantages by fundamentally altering the cost structure of producing high-purity agrochemical intermediates. The ability to recycle the amidation filtrate directly translates into a significant reduction in solvent consumption and raw material waste, which are major cost drivers in fine chemical manufacturing. By recovering unreacted acids and other valuable components from the mother liquor, the process effectively lowers the net input cost per kilogram of finished product, enhancing the overall margin profile for suppliers. This efficiency gain is particularly relevant for procurement managers seeking to secure long-term contracts for analytical standards, as it provides a buffer against volatility in raw material pricing and ensures more stable quotation structures over time.
- Cost Reduction in Manufacturing: The elimination of expensive solvent disposal costs and the reduction in fresh raw material requirements through filtrate recycling create a leaner manufacturing model. This process optimization removes the need for extensive purification steps often required in conventional routes, thereby lowering energy consumption and labor hours associated with downstream processing. Consequently, suppliers can offer more competitive pricing structures without compromising on the quality or purity specifications required by regulatory bodies. The economic logic is driven by the circular nature of the solvent system, which turns what was previously waste into a valuable resource for subsequent production batches.
- Enhanced Supply Chain Reliability: The robustness of this three-step synthesis ensures a consistent and reliable supply of N-cyanomethylbis(trifluoromethyl)nicotinamide, mitigating the risk of stockouts that can delay quality control testing for flonicamid production. Because the process tolerances are well-defined and the yield is significantly higher than legacy methods, manufacturers can plan production schedules with greater confidence and accuracy. This predictability is crucial for supply chain heads who need to guarantee the availability of critical reference materials to support continuous manufacturing operations at downstream pesticide facilities. The reduced dependency on complex purification also shortens the overall production cycle time, allowing for faster turnaround on orders.
- Scalability and Environmental Compliance: The method is inherently designed for commercial scale-up, utilizing common reagents and standard unit operations that are easily replicated in large-scale reactors. The reduction in hazardous waste generation, particularly through the minimization of chlorinated byproducts and solvent waste, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. This compliance advantage reduces the regulatory burden on manufacturing sites and minimizes the risk of production stoppages due to environmental permitting issues. Furthermore, the high purity of the final product reduces the need for re-processing, further lowering the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized agrochemical intermediate. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation to ensure accuracy and relevance. Understanding these details is essential for technical teams evaluating the feasibility of integrating this standard into their quality control workflows. The information provided here serves as a foundational guide for further discussions regarding specification alignment and supply agreements.
Q: What is the primary application of N-cyanomethylbis(trifluoromethyl)nicotinamide?
A: It serves as a critical reference standard for detecting and calibrating impurity levels during the industrial synthesis of the insecticide flonicamid.
Q: How does the patented method improve yield compared to conventional routes?
A: By implementing a specific three-step sequence and recycling the amidation filtrate, the method achieves yields between 71.1% and 85.7%, significantly higher than traditional direct acyl chlorination methods.
Q: What purity levels can be achieved with this synthesis protocol?
A: The optimized process, particularly through controlled crystallization and recrystallization steps, consistently delivers product purity ranging from 99.1% to 99.9%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Cyanomethylbis(Trifluoromethyl)Nicotinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity reference standards play in the development and quality assurance of next-generation agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We employ stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-cyanomethylbis(trifluoromethyl)nicotinamide meets the exacting standards required for HPLC calibration and impurity profiling. Our commitment to technical excellence means that we do not just supply chemicals; we provide solutions that enhance the reliability of your entire analytical workflow.
We invite you to collaborate with us to optimize your supply chain for flonicamid monitoring and related agrochemical applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and production goals efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
