Advanced Manufacturing of Tildipirosin: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of Tildipirosin: Technical Breakthroughs and Commercial Scalability
The pharmaceutical and veterinary industries are constantly seeking robust synthetic routes for high-value macrolide antibiotics, and the recent disclosure in patent CN112679570B offers a compelling solution for the production of Tildipirosin. This semi-synthetic derivative of tylosin represents a significant advancement in treating respiratory diseases in cattle and pigs, characterized by its unique tribasic structure and prolonged half-life. The patent outlines a novel synthesis and purification method that addresses critical pain points in prior art, specifically focusing on yield optimization and process stability. By leveraging a streamlined four-step transformation starting from tylosin phosphate, the disclosed methodology achieves a final product purity exceeding 99% through a series of controlled reactions and simplified workup procedures. This technical evolution is not merely an academic exercise but a strategic enabler for reliable veterinary drug intermediate supplier networks aiming to secure consistent quality.
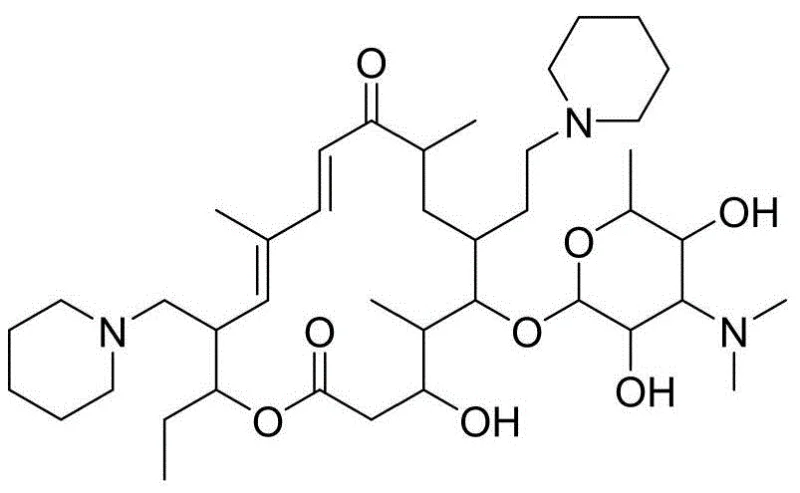
Furthermore, the integration of mild reaction conditions ranging from 60°C to 90°C allows for greater flexibility in reactor selection and energy management during commercial scale-up of complex veterinary drug intermediates. The process effectively mitigates the risks associated with unstable aldehyde intermediates often encountered in traditional oxidation pathways. For R&D directors evaluating technology transfer, the emphasis on avoiding column chromatography in favor of extraction and recrystallization signals a mature process ready for multi-ton manufacturing. The ability to recycle filtrates from recrystallization steps further underscores the economic viability of this approach, ensuring that raw material utilization is maximized while waste generation is minimized. Consequently, this patent provides a foundational blueprint for enhancing supply chain continuity in the global animal health sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tildipirosin has been plagued by inefficiencies that hinder large-scale adoption, particularly regarding yield and intermediate stability. Prior art, such as the methods disclosed in international patent WO 2008012343, often relies on amination reduction followed by strong acid hydrolysis and iodide activation. While conceptually straightforward, these legacy routes suffer from significant byproduct formation, resulting in dismal product yields reported as low as 12.2 percent. Such low efficiency translates directly into exorbitant production costs and substantial waste disposal challenges, creating a bottleneck for procurement managers focused on cost reduction in veterinary drug manufacturing. Additionally, other existing methods, like those in Chinese patent CN103880903B, involve oxidation steps that are hypersensitive to temperature variations.
When reaction temperatures exceed 0°C in these conventional oxidative pathways, the instability of the aldehyde intermediates leads to rapid degradation and the formation of complex impurity profiles that are difficult to separate. This sensitivity necessitates expensive cryogenic cooling infrastructure and rigorous process control, which increases capital expenditure and operational complexity. Furthermore, the reliance on column chromatography for purification in many older protocols is inherently non-scalable, limiting throughput and introducing variability in batch-to-batch consistency. For supply chain heads, these factors represent significant risks to delivery timelines and inventory reliability. The cumulative effect of these technical limitations is a fragile supply network that struggles to meet the growing global demand for effective livestock therapeutics without compromising on quality or price.
The Novel Approach
In stark contrast, the methodology presented in CN112679570B introduces a paradigm shift by prioritizing system stability and operational simplicity throughout the synthetic sequence. The novel approach utilizes tylosin phosphate as a robust starting material, subjecting it to a controlled reductive amination with formic acid and piperidine at elevated temperatures between 60°C and 90°C. This thermal window is significantly more forgiving than the sub-zero conditions required by previous methods, thereby reducing energy consumption and equipment strain. A key innovation lies in the iodination step, where iodine is introduced as a solution rather than a solid. This modification eliminates the exothermic spikes associated with dissolving solid iodine, maintaining a stable reaction environment that preserves the integrity of the sensitive macrolide backbone.
Moreover, the purification strategy has been completely re-engineered to favor liquid-liquid extraction and crystallization over chromatographic separation. By implementing a precise pH swing protocol—adjusting from neutral to highly alkaline conditions—the process effectively partitions the desired product from impurities into the organic phase. This not only simplifies the downstream processing but also facilitates the recycling of mother liquors, contributing to a theoretical yield of over 75% prior to final crystallization. The final product, obtained after secondary recrystallization, consistently demonstrates purity levels above 99%, meeting the stringent specifications required for high-purity veterinary drug intermediates. This holistic improvement in process design ensures that the manufacturing of Tildipirosin is not only chemically efficient but also commercially sustainable for long-term production.
Mechanistic Insights into Reductive Amination and Iodination Sequence
The core of this synthetic breakthrough lies in the precise orchestration of functional group transformations on the tylosin macrocycle, specifically targeting the C20 and C23 positions. The initial step involves a Leuckart-Wallach type reductive amination where tylosin phosphate reacts with piperidine in the presence of formic acid. Heating the mixture to 75°C facilitates the formation of an iminium ion intermediate, which is subsequently reduced in situ to install the first piperidine moiety at the C20 position. Following this, the mycaminose sugar moiety is hydrolyzed under acidic conditions to expose the C23 hydroxyl group, generating the critical 23-hydroxy-20-piperidinyl-tylonolide intermediate. The stability of this intermediate is paramount, and the patent specifies a careful pH adjustment sequence to prevent degradation during the aqueous workup, ensuring that the molecule remains intact for the subsequent activation step.
The subsequent iodination mechanism is particularly noteworthy for its deviation from standard protocols. Instead of direct addition, the use of triphenylphosphine and pyridine activates the C23 hydroxyl group, while the dropwise addition of iodine solution ensures a controlled concentration of the halogenating agent. This prevents local overheating and over-iodination, which are common sources of side reactions in macrolide chemistry. The resulting 23-iodo intermediate is then subjected to a nucleophilic substitution with a second equivalent of piperidine. The use of an alkali catalyst in this final amination step drives the displacement of the iodide leaving group, completing the installation of the second piperidine ring. This mechanistic pathway avoids the formation of unstable aldehyde species entirely, bypassing the primary failure mode of earlier synthetic routes and providing a cleaner, more predictable reaction profile for industrial execution.
Impurity control is intrinsically built into this mechanism through the physical properties of the intermediates. The sequential pH adjustments exploit the basicity of the piperidine nitrogen atoms. At neutral pH, certain acidic impurities remain in the aqueous phase, while at pH > 13, the free base form of the product partitions efficiently into the organic solvent. This selective extraction acts as a powerful purification tool at each stage, preventing the carryover of contaminants into the final crystallization. By maintaining water content below 0.1% before the iodination step, the process also suppresses hydrolysis side reactions that could cleave the glycosidic bonds. This rigorous control over reaction parameters ensures that the final impurity profile is minimal, reducing the burden on the final purification steps and guaranteeing a high-quality API precursor.
How to Synthesize Tildipirosin Efficiently
Implementing this synthesis requires strict adherence to the specified thermal and stoichiometric parameters to replicate the high yields reported in the patent data. The process begins with the dissolution of tylosin phosphate in a solvent such as ethyl acetate, followed by the addition of formic acid and piperidine for the initial amination. After the reaction reaches completion, the mixture is cooled and treated with hydrobromic acid to facilitate phase separation. The aqueous layer is then subjected to hydrolysis at controlled temperatures, followed by a meticulous extraction protocol involving dichloromethane and sodium hydroxide to isolate the hydroxy-intermediate. Critical to the success of the subsequent steps is the removal of water via atmospheric distillation to achieve a moisture level of less than 0.1%, preparing the substrate for the sensitive iodination reaction.
- Dissolve tylosin phosphate in organic solvent, add formic acid and piperidine, heat to 60-90°C for reductive amination, then separate aqueous layer.
- Hydrolyze the aqueous solution at 55-75°C, adjust pH sequentially to neutral then >13, extract, and dry to <0.1% water content.
- Perform iodination by dropwise adding iodine solution to the dried intermediate with triphenylphosphine and pyridine at 20-60°C.
- React the iodo-intermediate with piperidine and base catalyst at 40-90°C, followed by pH adjustment, extraction, and double recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the technical refinements in this patent translate directly into tangible operational benefits and risk mitigation. The elimination of cryogenic conditions and the shift to atmospheric pressure distillation significantly lower the barrier to entry for manufacturing partners, allowing for production in facilities with standard glass-lined or stainless steel reactors. This accessibility broadens the potential supplier base, enhancing competition and potentially driving down costs without sacrificing quality. The robust nature of the intermediates means that storage and transport between synthesis stages are less prone to degradation, reducing the incidence of batch failures and the associated financial losses. These factors collectively contribute to a more resilient supply chain capable of withstanding market fluctuations and demand surges.
- Cost Reduction in Manufacturing: The most significant economic driver in this new process is the dramatic increase in overall yield compared to legacy methods. By achieving a final yield of over 50% through secondary recrystallization, compared to the single-digit yields of older routes, the amount of raw tylosin phosphate required per kilogram of finished Tildipirosin is drastically reduced. Furthermore, the removal of column chromatography from the purification train eliminates the need for expensive silica gel and large volumes of elution solvents, which are major cost centers in fine chemical production. The ability to recycle mother liquors from the crystallization steps further enhances material efficiency, ensuring that valuable intermediates are not discarded but rather reprocessed into saleable product. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: The stability of the reaction system described in the patent ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply to pharmaceutical formulators. Traditional methods that rely on unstable aldehyde intermediates are prone to sudden yield drops if temperature control slips, leading to unpredictable lead times. In contrast, the dropwise iodination technique and the use of stable phosphate salts create a forgiving process window that tolerates minor operational variances. This reliability allows supply chain planners to forecast production schedules with greater accuracy and maintain lower safety stock levels. Additionally, the use of common, commercially available solvents like ethyl acetate and dichloromethane ensures that raw material sourcing remains straightforward and unaffected by niche chemical shortages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the simplified workup procedures offer distinct advantages. The reliance on extraction and crystallization rather than chromatography reduces the volume of hazardous waste generated per unit of product, aligning with increasingly strict environmental regulations in major manufacturing hubs. The process operates at moderate temperatures, reducing the energy footprint associated with heating and cooling large reactor vessels. This "green chemistry" aspect not only lowers utility costs but also simplifies the permitting process for facility expansions. The straightforward nature of the unit operations means that scaling from pilot plant to commercial production involves minimal re-engineering, allowing manufacturers to rapidly ramp up capacity to meet global demand for this essential veterinary antibiotic.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. They are derived from the specific experimental data and comparative examples provided in the patent documentation, offering clarity on critical process parameters. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or contract manufacturing agreements. The answers reflect the empirical findings of the inventors and highlight the specific advantages of the disclosed method over alternative approaches.
Q: Why is dropwise addition of iodine solution preferred over solid iodine in this synthesis?
A: Dropwise addition prevents temperature fluctuations caused by the heat of dissolution associated with solid iodine. This ensures a stable reaction system, minimizes byproduct formation, and significantly improves both yield and purity compared to batch solid addition methods.
Q: How does the new purification method improve industrial feasibility?
A: The method eliminates the need for complex column chromatography. By utilizing sequential pH adjustments and solvent extractions followed by simple recrystallization, the process achieves >99% purity. This simplifies equipment requirements and reduces processing time, making it highly suitable for large-scale production.
Q: What is the critical water content control parameter for the iodination step?
A: The water content must be controlled to less than 0.1% via atmospheric distillation prior to iodination. This strict dehydration ensures the iodination reaction proceeds smoothly without the need for extreme drying conditions (like 100ppm), balancing operational simplicity with chemical efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tildipirosin Supplier
The technical advancements detailed in patent CN112679570B underscore the complexity and precision required to produce high-quality Tildipirosin at a commercial scale. As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this sophisticated chemistry to life. Our state-of-the-art facilities are equipped to handle the specific solvent systems and pH control requirements of this macrolide synthesis, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we are uniquely positioned to deliver Tildipirosin intermediates and APIs that adhere to the highest global standards for veterinary pharmaceuticals.
We invite industry partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic impact of switching to this high-yield process. We encourage you to request specific COA data and route feasibility assessments to verify our capability to support your long-term production goals. Let us partner with you to secure a stable, cost-effective, and high-quality supply of Tildipirosin for the global animal health market.
