Advanced Pd-Catalyzed Synthesis of Indoline Tetrahydropyrans for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Indoline Tetrahydropyrans for Commercial Pharmaceutical Applications
The field of organic synthesis is constantly evolving to meet the demands of modern drug discovery, particularly in the construction of complex polycyclic scaffolds. Patent CN114292277A introduces a groundbreaking methodology for the preparation of indoline tetrahydropyran compounds, a structural motif frequently encountered in bioactive molecules. This innovation addresses a significant gap in synthetic chemistry by utilizing a palladium-catalyzed dearomatization [4+2] cycloaddition strategy. Unlike traditional approaches that often struggle with electron-deficient heterocycles, this process leverages the unique reactivity of 3-nitroindoles. The resulting compounds possess easily functionalized groups, making them invaluable precursors for the derivatization and synthesis of other chiral polycyclic systems. For research and development teams focused on expanding chemical space, this technology offers a robust platform for generating novel candidate molecules for high-throughput screening and new drug development.
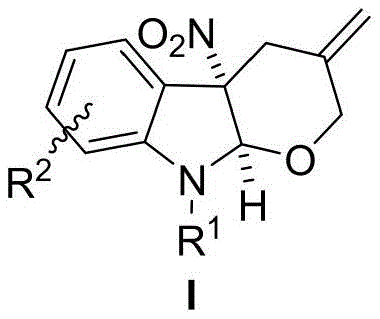
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, asymmetric dearomatization has been a formidable challenge in organic synthesis, with the majority of successful methodologies relying heavily on electron-rich aromatic compounds such as indoles and naphthols. These traditional pathways exploit the inherent high nucleophilicity of such substrates to facilitate ring-forming reactions. However, when chemists attempt to apply these standard protocols to electron-poor heteroaromatics like 3-nitroindoles, the reactions often fail or proceed with poor efficiency. Furthermore, prior art regarding palladium-catalyzed dearomatization involving 3-nitroindoles is extremely scarce, with only two documented cases of [3+2] cycloaddition reactions involving Π-allylpalladium 1,4-[N,C] dipolar active intermediates. The scarcity of [4+2] cycloaddition reports is attributed to the significantly weaker nucleophilic capability of oxygen compared to carbon and nitrogen, which creates a high energy barrier for the initial dearomatization addition step, rendering many potential synthetic routes unviable for industrial application.
The Novel Approach
The technology disclosed in patent CN114292277A represents a paradigm shift by successfully realizing the dearomatization [4+2] cycloaddition between a Π-allylpalladium 1,4-[O,C] dipolar active intermediate and 3-nitroindole. This is the first reported instance of utilizing such an intermediate to achieve dearomatization of electron-poor aromatic heterocycles, effectively overcoming the nucleophilicity limitations that hindered previous efforts. The reaction utilizes alkenyl carbonates, which under palladium catalysis, generate the requisite reactive species to couple with the indole core. This novel approach not only expands the toolbox available to synthetic chemists but also provides direct access to indoline-tetrahydropyran compounds that are widely prevalent in biologically active molecules. The method is characterized by its novelty, operational simplicity, mild reaction conditions, and exceptionally high yields, making it a superior alternative to existing synthetic strategies for constructing these complex fused ring systems.
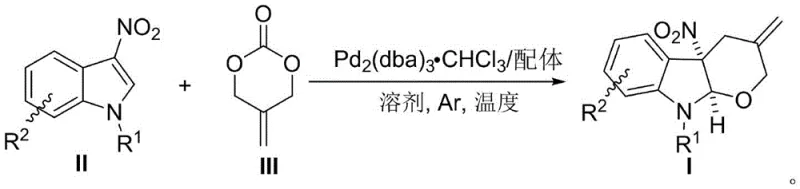
Mechanistic Insights into Pd-Catalyzed Dearomatization [4+2] Cycloaddition
The core of this synthetic breakthrough lies in the sophisticated manipulation of palladium catalysis to generate a specific reactive intermediate. The mechanism initiates with the oxidative addition of the palladium catalyst, typically Pd2(dba)3·CHCl3, to the alkenyl carbonate substrate. This step is crucial as it leads to the formation of the Π-allylpalladium 1,4-[O,C] dipolar active intermediate, a species that had previously been difficult to harness for dearomatization purposes. The presence of the phosphine ligand, optimally triphenylphosphine (PPh3), stabilizes this intermediate and modulates its electronic properties to facilitate the subsequent attack on the electron-deficient 3-nitroindole. The nitro group at the 3-position of the indole plays a dual role; while it reduces the electron density of the aromatic ring, it also activates the system for nucleophilic attack under the right catalytic conditions. The coordination of the palladium complex allows the oxygen nucleophile to overcome its inherent weakness and engage in the [4+2] cyclization, forging the tetrahydropyran ring with high precision.
Controlling the stereochemistry and purity of the final product is paramount for pharmaceutical applications, and this methodology excels in that regard. The reaction conditions are finely tuned to ensure excellent diastereoselectivity, consistently achieving diastereomeric ratios (dr) greater than 20:1. This high level of stereocontrol is likely driven by the rigid transition state imposed by the palladium-ligand complex during the ring-closing step. Furthermore, the use of mild temperatures, specifically around 40°C, minimizes side reactions and decomposition pathways that could lead to impurity formation. The process tolerates a wide range of substituents on both the indole and the carbonate components, including halogens and various electron-withdrawing or donating groups, without compromising the integrity of the catalytic cycle. This robustness ensures that the impurity profile remains manageable, simplifying downstream purification processes such as column chromatography and enhancing the overall feasibility of the synthesis for large-scale production.
How to Synthesize Indoline Tetrahydropyran Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these valuable compounds. The procedure begins with the preparation of the catalytic system in a dry reaction vessel, ensuring that moisture does not interfere with the sensitive palladium species. The catalyst and ligand are stirred in the solvent, preferably acetonitrile, to allow for complete complexation before the introduction of the substrates. This pre-activation step is critical for maximizing the reaction rate and yield. Once the catalyst system is ready, the 3-nitroindole and alkenyl carbonate are added sequentially, and the mixture is maintained at the optimal temperature until conversion is complete. The detailed standardized synthesis steps for implementing this protocol in a laboratory or pilot plant setting are provided in the guide below.
- Prepare the catalyst system by mixing Pd2(dba)3·CHCl3 and a phosphine ligand (preferably PPh3) in a dry reaction vessel with solvent.
- Stir the mixture for approximately 5 minutes to ensure complete complexation before adding the substrates.
- Add 3-nitroindole and alkenyl carbonate sequentially, maintain reaction temperature at 40°C, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The process is designed with scalability and cost-efficiency in mind, addressing common pain points associated with the manufacturing of complex heterocyclic intermediates. By utilizing readily available starting materials and a catalyst system that operates under mild conditions, the technology reduces the dependency on exotic reagents and extreme processing parameters. This translates directly into a more resilient supply chain, as the risk of disruption due to specialized raw material shortages is significantly minimized. Furthermore, the operational simplicity of the method means that it can be implemented using standard reactor equipment without the need for extensive retrofitting or specialized high-pressure infrastructure, facilitating a smoother transition from laboratory scale to commercial production.
- Cost Reduction in Manufacturing: One of the most significant economic advantages of this method is the remarkably low catalyst loading required to achieve high yields. The patent specifies that the catalyst dosage can be as low as 1 mol%, which drastically reduces the consumption of palladium, a precious metal with considerable cost implications. Additionally, the preferred ligand, triphenylphosphine, is inexpensive and commercially abundant, further driving down the raw material costs compared to processes requiring complex, proprietary ligands. The elimination of harsh reaction conditions also leads to substantial energy savings, as the process operates effectively at moderate temperatures rather than requiring cryogenic cooling or high-temperature heating. These factors combined result in a significantly optimized cost structure for the manufacturing of indoline tetrahydropyran derivatives, enhancing the overall margin potential for downstream pharmaceutical products.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain stability. Because the reaction tolerates a broad scope of substrates and uses common solvents like acetonitrile, sourcing flexibility is greatly improved. Manufacturers are not locked into single-source suppliers for niche reagents, allowing for competitive bidding and better inventory management. The high yields reported, reaching up to 99% in optimized examples, mean that less raw material is wasted per unit of product produced, improving the overall material throughput efficiency. This efficiency reduces the frequency of production runs needed to meet demand, thereby lowering the logistical burden and ensuring a more consistent flow of high-purity intermediates to the formulation stage. Such reliability is crucial for maintaining uninterrupted production schedules in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this synthesis aligns well with green chemistry principles. The mild reaction conditions reduce the generation of hazardous byproducts and lower the overall energy footprint of the manufacturing process. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, facilitates effective waste management and reduces the volume of hazardous waste requiring disposal. The high selectivity of the reaction minimizes the formation of isomeric impurities, which simplifies the purification process and reduces the solvent consumption associated with extensive chromatographic separations. These attributes make the process highly scalable, allowing for seamless expansion from kilogram to multi-ton production capacities while maintaining strict compliance with environmental regulations and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their supply chains. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing workflows and for anticipating potential challenges during scale-up.
Q: What makes this Pd-catalyzed method unique compared to traditional dearomatization?
A: This method represents the first reported instance of a Π-allylpalladium 1,4-[O,C] dipolar active intermediate reacting with electron-poor 3-nitroindoles. Unlike conventional methods that rely on electron-rich aromatics or [3+2] cycloadditions, this [4+2] pathway successfully overcomes the low nucleophilicity of oxygen, enabling the construction of complex indoline-tetrahydropyran scaffolds with excellent diastereoselectivity.
Q: What are the optimal reaction conditions for maximizing yield?
A: According to the patent data, the optimal conditions involve using acetonitrile as the solvent with a catalyst loading of 5 mol% Pd2(dba)3·CHCl3 and 15 mol% triphenylphosphine (PPh3). The reaction proceeds efficiently at a mild temperature of 40°C, achieving yields up to 99% with a diastereomeric ratio greater than 20:1.
Q: Can this synthesis be adapted for diverse substrate libraries?
A: Yes, the methodology demonstrates broad substrate tolerance. The 3-nitroindole component can accommodate various substituents including halogens (Cl, Br, F), alkyl groups, and electron-withdrawing groups like cyano. Additionally, the nitrogen protecting group can be varied (Ts, Ac, Boc, etc.), making it highly suitable for generating diverse compound libraries for drug screening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline Tetrahydropyran Supplier
The technological advancements described in patent CN114292277A highlight the immense potential of palladium-catalyzed dearomatization in modern drug synthesis. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative pathways and possess the technical expertise to bring them to life on a commercial scale. As a dedicated CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of indoline tetrahydropyran intermediate meets the highest quality standards required for pharmaceutical applications.
We invite you to leverage our capabilities to optimize your supply chain and accelerate your drug development timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs, helping you identify opportunities for efficiency and budget optimization. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to quality and innovation can support your strategic goals and ensure a reliable supply of critical pharmaceutical intermediates.
