Advanced Catalytic Synthesis of Pyrazoline Nucleoside Analogs with Quaternary Carbon Centers for Commercial Scale-Up
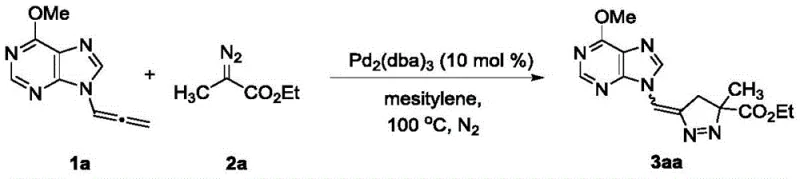
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those containing sterically demanding quaternary carbon centers. Patent CN108774229B discloses a groundbreaking synthetic route for pyrazoline nucleoside analogs, utilizing a palladium-catalyzed 1,3-dipolar cycloaddition between 9-allene purines and α-alkyl/aryl diazoacetates. This technology addresses a significant gap in organic synthesis, as the construction of pyrazolines with quaternary centers via allene-diazo cycloaddition has been historically underexplored compared to traditional alkene or alkyne counterparts. The innovation lies not only in the successful formation of the core structure but also in the precise control over product diversity and regioselectivity through catalyst modulation. For R&D directors and process chemists, this represents a versatile platform for generating novel antiviral and anticancer lead compounds with enhanced metabolic stability due to the quaternary center.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of pyrazoline derivatives predominantly relies on the 1,3-dipolar cycloaddition of diazo compounds with simple alkenes or alkynes. While effective for generating tertiary carbon centers, these classical methods often struggle when attempting to introduce quaternary stereocenters due to severe steric hindrance and unfavorable electronic interactions. Furthermore, the use of unactivated allenes in such cycloadditions is exceptionally rare in literature, limiting the structural diversity available to medicinal chemists. Conventional approaches frequently suffer from poor regioselectivity, resulting in complex mixtures of isomers that are difficult and costly to separate on a commercial scale. The lack of efficient catalytic systems capable of handling the cumulative double bonds of allenes has previously restricted the accessibility of this valuable chemical space, forcing researchers to rely on multi-step sequences that degrade overall atom economy and increase waste generation.
The Novel Approach
The methodology described in the patent overcomes these hurdles by employing a specialized palladium catalytic system that activates the allene moiety for efficient cycloaddition. By simply switching the ligand environment—using either Pd2(dba)3 or DPPB—chemists can dictate the outcome of the reaction. When Pd2(dba)3 is utilized, the process exhibits remarkable selectivity, favoring the formation of a single 1-pyrazoline isomer, which simplifies downstream purification significantly. Conversely, employing DPPB allows for the generation of diverse isomeric mixtures, providing a powerful tool for diversity-oriented synthesis. This dual-pathway capability ensures that the process can be tailored either for high-purity single-entity production or for library generation, offering unparalleled flexibility for drug discovery programs aiming to optimize structure-activity relationships rapidly.
Mechanistic Insights into Pd-Catalyzed 1,3-Dipolar Cycloaddition
The core of this transformation involves the generation of a reactive metal-carbene or π-allyl palladium intermediate that facilitates the cycloaddition with the electron-deficient diazo species. The palladium catalyst coordinates with the cumulative double bonds of the 9-allene purine, lowering the activation energy required for the nucleophilic attack by the diazo compound. This coordination is critical for overcoming the inherent stability of the allene system and directing the regiochemistry of the ring closure. The steric bulk of the ligands plays a pivotal role; bulky phosphine ligands in the Pd2(dba)3 system likely enforce a specific conformation that blocks alternative reaction pathways, thereby ensuring the formation of the thermodynamically favored 1-pyrazoline product. In contrast, the bidentate nature of DPPB may allow for a more flexible coordination sphere, permitting the formation of multiple isomeric products including the 4-methylene pyrazolines.
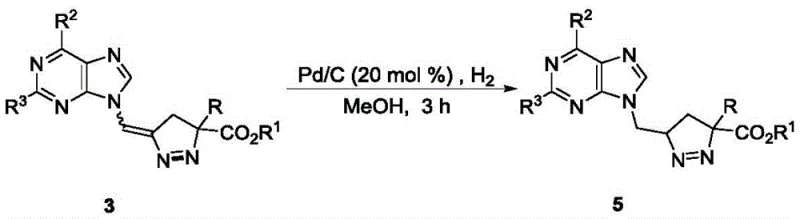
Impurity control is inherently built into this catalytic design. The high selectivity observed with the Pd2(dba)3 system minimizes the formation of regioisomeric byproducts, which are often the most challenging impurities to remove via crystallization or chromatography. Additionally, the reaction conditions utilize inert atmospheres and controlled temperatures to prevent the premature decomposition of the sensitive diazo reagents, which can lead to hazardous nitrogen gas evolution and non-specific carbene insertion side reactions. The subsequent hydrogenation step, converting the exocyclic double bond to a single bond using Pd/C, further demonstrates the robustness of the scaffold, proceeding with yields as high as 95% without affecting the sensitive purine base. This level of chemoselectivity ensures that the final API intermediates meet stringent purity specifications required for regulatory submission.
How to Synthesize Pyrazoline Nucleoside Analogs Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the precise weighing of 9-allene purine and the corresponding diazoacetate, followed by the addition of the selected palladium catalyst in an aromatic solvent. The reaction is conducted under a strict nitrogen blanket to maintain anhydrous and oxygen-free conditions, which are essential for catalyst longevity and safety. Heating the mixture to 100°C drives the cycloaddition to completion, typically within 24 to 48 hours depending on the substrate electronics.
- Prepare the reaction vessel with 9-allene purine and alpha-alkyl/aryl diazoacetate under inert nitrogen atmosphere.
- Select the appropriate catalyst system: Pd2(dba)3 for单一 product selectivity or DPPB for diverse isomer generation.
- Heat the reaction mixture in toluene or mesitylene at 100°C for 24-48 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers substantial strategic benefits regarding cost efficiency and supply reliability. The ability to tune the reaction outcome through catalyst selection eliminates the need for developing entirely different synthetic routes for different analogs, thereby consolidating the supply chain and reducing the inventory complexity of specialized reagents. The high yields reported, ranging from moderate to excellent levels across various substrates, directly translate to reduced raw material consumption per kilogram of finished product. This efficiency is crucial for maintaining healthy margins in the competitive pharmaceutical intermediate market, where raw material costs can fluctuate wildly.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences traditionally required to build quaternary centers significantly lowers the operational expenditure. By achieving the core scaffold in a single catalytic step, manufacturers save on solvent usage, energy consumption for heating and cooling cycles, and labor hours associated with intermediate isolations. The high atom economy of the cycloaddition reaction ensures that a maximum proportion of the starting materials ends up in the final product, minimizing waste disposal costs which are increasingly regulated and expensive in the fine chemical sector.
- Enhanced Supply Chain Reliability: The starting materials, specifically 9-allene purines and diazoacetates, are commercially accessible or can be synthesized via established protocols, reducing the risk of supply bottlenecks associated with exotic reagents. The robustness of the reaction conditions, tolerating a variety of substituents on the purine ring and the diazo component, means that supply disruptions for one specific derivative can be mitigated by switching to alternative analogs within the same chemical family without retooling the entire production line. This flexibility ensures continuous availability of critical intermediates for downstream drug manufacturing.
- Scalability and Environmental Compliance: The process utilizes standard aromatic solvents like toluene and mesitylene, which are well-understood in large-scale chemical engineering and have established recovery and recycling protocols. The absence of toxic heavy metals other than the trace palladium catalyst, which can be effectively scavenged, aligns with modern green chemistry principles and environmental regulations. The scalability is further supported by the straightforward workup procedures involving filtration and column chromatography, which are easily adaptable to continuous flow processing or large batch reactors for ton-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope limitations defined in the patent documentation, providing a realistic view of the process capabilities. Understanding these nuances is vital for project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of using Pd2(dba)3 over DPPB in this synthesis?
A: Pd2(dba)3 provides high regioselectivity, yielding primarily the single 1-pyrazoline product, whereas DPPB leads to a mixture of isomers, allowing for diversity-oriented synthesis depending on the target application.
Q: Can the double bond in the pyrazoline ring be further functionalized?
A: Yes, the exocyclic double bond in the pyrazoline product can be efficiently hydrogenated using Pd/C catalyst to yield saturated pyrazoline derivatives with excellent yields up to 95%.
Q: What are the typical reaction conditions for this cycloaddition?
A: The reaction typically proceeds in aromatic solvents like toluene or mesitylene at elevated temperatures around 100°C under a nitrogen atmosphere to ensure safety and prevent diazo decomposition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazoline Nucleoside Analog Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to manufacture these complex pyrazoline nucleoside analogs. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch meets the exacting standards required for pharmaceutical applications. We understand the critical nature of supply continuity and have optimized our processes to deliver high-purity intermediates consistently.
We invite potential partners to engage with our technical procurement team for a Customized Cost-Saving Analysis tailored to your specific project needs. Whether you require specific COA data for regulatory filings or detailed route feasibility assessments for novel derivatives, our experts are ready to provide the support necessary to accelerate your drug development timeline. Contact us today to discuss how our advanced catalytic capabilities can enhance your supply chain resilience.
