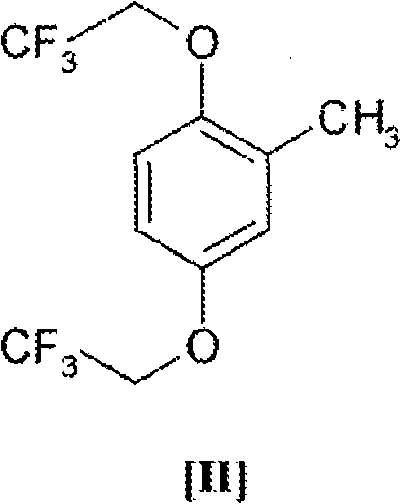
Advanced Copper-Catalyzed Synthesis of 2,5-Bis(2,2,2-trifluoroethoxy)toluene for Commercial API Production
Advanced Copper-Catalyzed Synthesis of 2,5-Bis(2,2,2-trifluoroethoxy)toluene for Commercial API Production
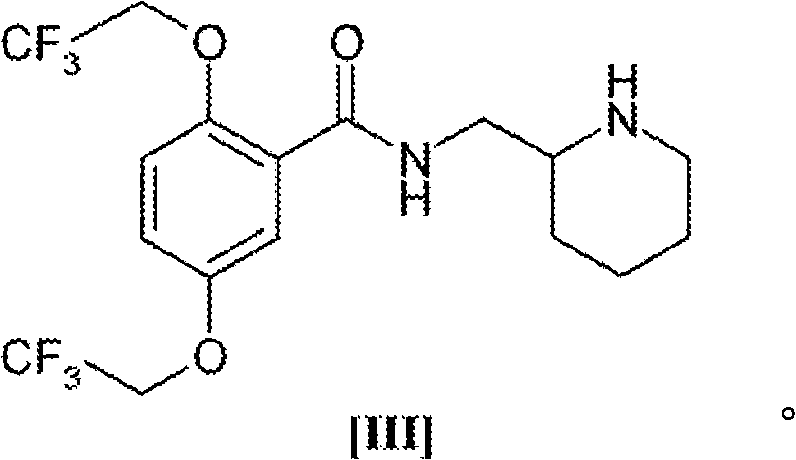
The pharmaceutical industry continuously demands more efficient and safer pathways for synthesizing critical antiarrhythmic agents, particularly Flecainide. A pivotal breakthrough in this domain is documented in patent CN101516817A, which discloses a robust method for preparing 2,5-bis(2,2,2-trifluoroethoxy)toluene, a high-value intermediate essential for the commercial manufacture of Flecainide acetate. This novel approach addresses long-standing challenges regarding safety, yield, and scalability that have plagued previous synthetic routes. By leveraging a copper-catalyzed etherification strategy starting from 2,5-dihalotoluene, the process achieves superior conversion rates while mitigating the thermal hazards associated with traditional strong base systems. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this technology is crucial for securing a stable supply chain. The structural integrity of the target molecule, as depicted below, is fundamental to its biological activity in the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-bis(2,2,2-trifluoroethoxy)benzene derivatives relied heavily on the reaction of 1,4-dibromobenzene with 2,2,2-trifluoroethanol in the presence of sodium hydride and cuprous iodide. A critical flaw in this legacy methodology, referenced in GB 2045760, is the stoichiometric inefficiency; it necessitates a massive excess of trifluoroethanol, often up to 8 equivalents, to drive the reaction to completion. Even with such excess, the conversion remains incomplete, resulting in a complex mixture containing significant amounts of starting material and mono-substituted intermediates like 1-bromo-4-(2,2,2-trifluoroethoxy)benzene. Furthermore, alternative methods utilizing sodium hydride in DMF present unacceptable safety profiles for commercial scale-up of complex pharmaceutical intermediates. The combination of NaH and DMF is known to undergo uncontrollable exothermic decomposition, posing severe thermal runaway risks that are untenable for modern GMP manufacturing environments.
The Novel Approach
In stark contrast, the innovative process described in the patent utilizes 2,5-dihalotoluene as the starting scaffold, which fundamentally alters the reaction kinetics and thermodynamics. By employing metallic sodium instead of sodium hydride, the protocol eliminates the hazardous exothermic decomposition risks inherent to the older NaH/DMF systems. This substitution not only enhances operator safety but also streamlines the reaction profile, allowing for precise temperature control during the alkoxide formation phase. The new method operates effectively with a much more favorable reagent ratio, typically between 2 to 10 moles of trifluoroethanol per mole of dihalide, drastically reducing raw material waste. This shift represents a paradigm change in cost reduction in pharmaceutical intermediate manufacturing, as it minimizes the burden on downstream purification units and reduces the environmental footprint associated with solvent and reagent disposal.
Mechanistic Insights into Copper-Catalyzed Etherification
The core of this synthetic advancement lies in the copper-catalyzed nucleophilic aromatic substitution, often referred to as an Ullmann-type condensation. The reaction initiates with the formation of the sodium trifluoroethoxide species in situ, generated by the careful addition of sodium metal to 2,2,2-trifluoroethanol in a dioxane medium. This step is critical; the patent specifies maintaining the temperature between 22°C and 90°C during sodium addition to manage the exotherm safely. Once the alkoxide is formed, the introduction of the dipolar aprotic solvent, N,N-dimethylformamide (DMF), facilitates the solubility of the organic substrates and the copper catalyst. The copper(II) sulfate acts as a pre-catalyst, likely reducing to an active Cu(I) species under the reaction conditions, which then coordinates with the aromatic halide and the alkoxide nucleophile to lower the activation energy for the C-O bond formation.
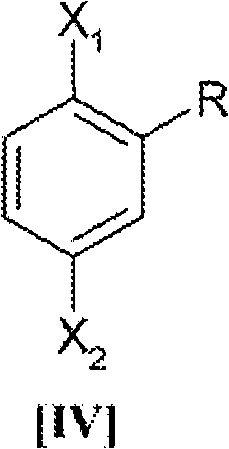
Regioselectivity and impurity control are paramount in the production of high-purity intermediates for API synthesis. The use of 2,5-dihalotoluene, specifically 2,5-dibromotoluene where X1 and X2 are bromine, ensures that the substitution occurs at the correct positions relative to the methyl group. The methyl group exerts a subtle electronic and steric influence that, combined with the catalytic system, directs the trifluoroethoxy groups to the 2 and 5 positions efficiently. The reaction is typically conducted at an optimal temperature range of 85°C to 105°C, often held at 95-100°C for approximately 4 hours. This thermal window is wide enough to ensure complete conversion of the dihalide but controlled enough to prevent side reactions or solvent degradation. The result is a crude product that, upon simple acidification and crystallization, yields the target compound with high chemical purity, minimizing the presence of mono-substituted byproducts that plague the 1,4-dibromobenzene route.
How to Synthesize 2,5-Bis(2,2,2-trifluoroethoxy)toluene Efficiently
Implementing this synthesis requires strict adherence to the specified addition sequences and thermal profiles to maximize yield and safety. The process begins with the generation of the nucleophile in a separate vessel or the main reactor under inert atmosphere, followed by the sequential addition of the catalyst and substrate. The patent emphasizes the importance of the workup procedure, where the reaction mixture is quenched into cold aqueous methanol and acidified to pH 1-2 to precipitate the product. This straightforward isolation technique avoids complex extraction protocols, making it highly attractive for process chemistry teams aiming for reducing lead time for high-purity pharmaceutical intermediates. The detailed standardized operating procedures for this transformation are outlined below.
- Preparation of Alkoxide: Dissolve sodium metal in dioxane and add 2,2,2-trifluoroethanol carefully to manage exotherm, stirring until sodium is fully consumed.
- Coupling Reaction: Add DMF solvent, 2,5-dibromotoluene substrate, and copper(II) sulfate catalyst, then heat the mixture to 95-100°C for 4 hours.
- Isolation and Purification: Quench the reaction with cold aqueous methanol and acidify to pH 1-2 to precipitate the pure white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the substantial optimization of the cost structure associated with raw material consumption and waste management. By moving away from the 8-equivalent excess requirement of the legacy process, manufacturers can achieve significant savings on trifluoroethanol, a relatively expensive fluorinated building block. Additionally, the replacement of sodium hydride with metallic sodium reduces the costs associated with hazardous material handling, storage, and specialized disposal, thereby lowering the overall operational expenditure. These efficiencies translate directly into a more competitive pricing model for the final intermediate, supporting cost reduction in API manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of excessive reagent usage and the simplification of the purification process lead to a leaner manufacturing cost base. Traditional methods often require extensive chromatography or recrystallization steps to remove stubborn mono-substituted impurities, which drives up solvent and labor costs. The new method's high selectivity minimizes these downstream processing requirements, allowing for a more streamlined production flow. Furthermore, the use of standard solvents like DMF and dioxane, which are readily available in bulk quantities, ensures that supply chain volatility for niche reagents does not impact production schedules or budgets.
- Enhanced Supply Chain Reliability: Sourcing 2,5-dihalotoluene derivatives is generally more stable and predictable compared to managing the logistics of large volumes of sodium hydride slurry, which requires stringent safety protocols for transport and storage. The robustness of the new reaction conditions means that production batches are less prone to failure due to thermal excursions or reagent instability. This reliability is critical for maintaining continuous supply to downstream API manufacturers, preventing costly stockouts or delays in the drug development pipeline. Suppliers adopting this technology can offer more consistent lead times and volume flexibility to their global partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed to function effectively in standard industrial reactors without the need for specialized high-pressure or cryogenic equipment. This ease of scale-up facilitates rapid capacity expansion to meet market demand surges. From an environmental perspective, the reduction in reagent excess and the avoidance of hazardous decomposition byproducts align with modern green chemistry principles. This compliance reduces the regulatory burden on waste treatment facilities and supports the sustainability goals of multinational pharmaceutical corporations seeking eco-friendly supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior choice for industrial applications. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and vendor qualification.
Q: Why is the new sodium metal method safer than the conventional sodium hydride process?
A: Conventional methods utilizing sodium hydride (NaH) in DMF pose severe thermal runaway risks due to uncontrollable exothermic decomposition. The novel process replaces NaH with metallic sodium, which allows for better thermal management during the alkoxide formation stage, significantly enhancing operational safety in large-scale reactors.
Q: What are the purity advantages of using 2,5-dihalotoluene over 1,4-dibromobenzene?
A: Traditional routes starting from 1,4-dibromobenzene often suffer from incomplete conversion, requiring excessive reagents (up to 8 equivalents) and leaving difficult-to-remove mono-substituted impurities. The 2,5-dihalotoluene route offers superior regioselectivity and conversion efficiency, simplifying downstream purification and ensuring higher chemical purity for API synthesis.
Q: Can this process be scaled for industrial production without specialized equipment?
A: Yes, the process is designed for industrial applicability using standard chemical processing equipment. It operates at moderate temperatures (85°C to 105°C) and utilizes common solvents like DMF and dioxane, eliminating the need for exotic high-pressure or cryogenic infrastructure typically associated with complex fluorinated intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Bis(2,2,2-trifluoroethoxy)toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiarrhythmic therapies depends on the availability of high-quality key intermediates. Our technical team has extensively evaluated the process described in CN101516817A and integrated its core principles into our manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in clinical trials or full-scale commercial launch. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2,5-bis(2,2,2-trifluoroethoxy)toluene meets the exacting standards required for Flecainide synthesis.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our adoption of this advanced copper-catalyzed route can lower your total cost of ownership. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us be your partner in delivering safe, efficient, and cost-effective pharmaceutical solutions.