Advanced Catalyst-Free N-Methylation Technology for High-Purity Pharmaceutical Intermediates
Advanced Catalyst-Free N-Methylation Technology for High-Purity Pharmaceutical Intermediates
The synthesis of N-methylated aromatic amines represents a critical transformation in the manufacturing of active pharmaceutical ingredients (APIs), agrochemicals, and advanced fine chemicals. These structural motifs are ubiquitous in drug discovery and process chemistry, serving as key building blocks for a vast array of therapeutic compounds. A recent technological breakthrough, documented in patent CN103288660A, introduces a highly efficient, catalyst-free methodology for achieving N-methylation. This novel approach utilizes a system comprising formic acid and triethylamine in dimethylsulfoxide (DMSO) under inert conditions. Unlike traditional methods that rely on hazardous alkyl halides or expensive metal catalysts, this protocol offers a greener, safer, and economically superior alternative. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this technology is essential for optimizing supply chains and reducing manufacturing costs in fine chemical production.
The significance of this patent lies in its ability to handle a diverse range of substrates with remarkable consistency. From simple aniline derivatives to complex fused heterocyclic systems like tetrahydroquinolines and indolines, the method demonstrates exceptional versatility. The reaction conditions are robust, typically operating between 130°C and 160°C, which ensures complete conversion without the need for sensitive catalytic species. This stability is paramount for commercial scale-up of complex pharmaceutical intermediates, where batch-to-batch reproducibility is non-negotiable. By leveraging formic acid as both the carbon source and the reducing agent, the process elegantly bypasses the generation of stoichiometric salt waste associated with classical alkylation, aligning perfectly with modern green chemistry principles and stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the N-methylation of aromatic amines has been fraught with significant technical and safety challenges that impact both operational efficiency and regulatory compliance. Traditional protocols frequently employ methylating agents such as methyl iodide, dimethyl sulfate, or methyl triflate. While effective, these reagents are potent alkylating agents known for their high toxicity, mutagenicity, and carcinogenicity, posing severe health risks to operators and requiring elaborate containment infrastructure. Furthermore, alternative methods utilizing formaldehyde often necessitate the use of reducing agents like sodium borohydride or cyanoborohydride, which generate substantial quantities of boron-containing waste streams that are difficult and costly to treat. Another common approach involves the use of transition metal catalysts for reductive amination; however, this introduces the risk of heavy metal contamination in the final product, necessitating expensive and time-consuming purification steps to meet the rigorous ppm-level specifications required by global pharmacopeias.
The Novel Approach
The methodology disclosed in CN103288660A fundamentally disrupts these conventional paradigms by introducing a metal-free, reagent-minimalist system. By employing formic acid and triethylamine in DMSO, the reaction proceeds through a unique mechanism where formic acid serves a dual role: it initially formylates the amine and subsequently acts as the hydride donor for the reduction of the intermediate formamide to the methyl amine. This eliminates the need for external reducing agents or toxic alkyl halides entirely. The absence of transition metals is a game-changer for supply chain reliability, as it removes the bottleneck of metal scavenging and validation. Moreover, the reaction tolerates a wide array of functional groups, including electron-donating methoxy groups and electron-withdrawing halogens, without compromising yield. This robustness allows manufacturers to streamline their synthetic routes, reducing the number of unit operations and significantly lowering the overall cost of goods sold (COGS) for high-value intermediates.
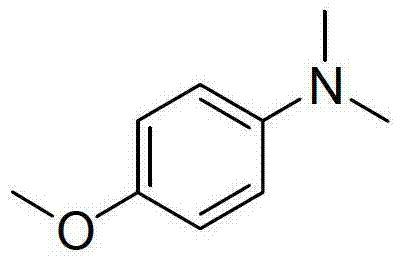
Mechanistic Insights into Formic Acid-Mediated Reductive Methylation
The core of this innovation lies in the intricate interplay between formic acid, triethylamine, and the aromatic amine substrate under thermal conditions. The mechanism is postulated to proceed via an initial nucleophilic attack of the amine on the carbonyl carbon of formic acid, facilitated by the basic environment provided by triethylamine, leading to the formation of an N-formyl intermediate. This formamide species is then subjected to a transfer hydrogenation process, where a second equivalent of formic acid (activated by the amine base) decomposes to release carbon dioxide and a hydride equivalent. This hydride attacks the electrophilic carbon of the formyl group, reducing it to a methyl group. This sequence—formylation followed by reduction—occurs twice to achieve the N,N-dimethylated product. The use of DMSO as a polar aprotic solvent is critical, as it stabilizes the charged transition states and facilitates the high-temperature conditions (up to 160°C) required to drive the dehydration and reduction steps to completion without the aid of a metal catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over competitive pathways. Because the reaction does not involve radical intermediates or harsh acidic conditions typical of alkyl halide substitutions, side reactions such as ring halogenation or polymerization are minimized. The primary byproducts are carbon dioxide and water (from the condensation steps), along with triethylammonium salts, which are easily removed during the aqueous workup. This clean reaction profile is evident in the high purity of the isolated products, such as the N,N-dimethyl-p-anisidine shown below, which was obtained in 92% yield with minimal chromatographic purification. For R&D teams, this implies a simpler impurity profile, easier method development for QC, and a lower risk of genotoxic impurity (GTI) formation, thereby accelerating the regulatory filing timeline for new drug applications.
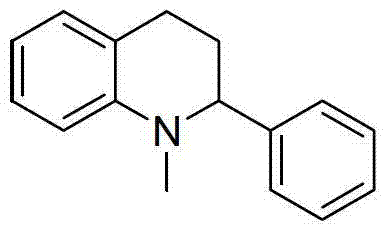
How to Synthesize N-Methylated Aromatic Amines Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize side products. The general protocol involves charging a pressure-rated vessel with the aromatic amine, a significant excess of formic acid (15 to 25 equivalents), and triethylamine (15 to 25 equivalents) in DMSO. The system must be purged with an inert gas like argon or nitrogen to prevent oxidative degradation of the reagents at high temperatures. The mixture is then heated to between 130°C and 160°C for 10 to 24 hours, depending on the steric hindrance of the substrate. Following the reaction, the mixture is cooled and carefully basified with sodium hydroxide solution to liberate the free amine from its salt form. Extraction with dichloromethane, followed by drying and solvent removal, yields the crude product, which can be further purified by flash column chromatography if necessary. Detailed standardized synthetic steps for specific substrates are provided in the guide below.
- Prepare the reaction mixture by combining the aromatic amine substrate, formic acid, and triethylamine in dimethylsulfoxide (DMSO) solvent under an inert gas atmosphere, maintaining a molar ratio of approximately 1: 20:20.
- Heat the sealed reaction vessel to a temperature range of 130°C to 160°C and stir continuously for a duration of 10 to 24 hours to facilitate the formylation and subsequent reduction steps.
- Upon completion, cool the mixture, adjust pH to alkaline using sodium hydroxide, extract with dichloromethane, dry the organic phase, and purify via column chromatography to isolate the high-purity N-methylated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free N-methylation technology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of expensive and regulated reagents like methyl iodide or palladium catalysts drastically simplifies the raw material sourcing landscape. Formic acid and triethylamine are commodity chemicals available in bulk quantities from multiple global suppliers, ensuring supply continuity and shielding the manufacturing process from the volatility of the precious metals market. Furthermore, the simplified workup procedure—requiring only basic extraction and distillation rather than complex metal scavenging or crystallization from hazardous solvents—reduces the consumption of auxiliary materials and shortens the production cycle time. This operational efficiency allows for faster turnaround on custom synthesis orders and enhances the overall agility of the supply chain in responding to market demands for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this formic acid-based protocol is profound. By removing the need for transition metal catalysts, manufacturers eliminate the capital expenditure associated with catalyst recovery systems and the recurring cost of purchasing noble metals. Additionally, the avoidance of toxic alkyl halides reduces the costs linked to hazardous waste disposal and regulatory compliance reporting. The high atom economy of using formic acid, which decomposes into gaseous CO2, means less solid waste is generated per kilogram of product. These factors combine to significantly lower the variable cost of production, allowing for more competitive pricing strategies in the global marketplace for fine chemical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Unlike specialized ligands or catalysts that may have single-source suppliers and long lead times, formic acid and triethylamine are produced on a massive industrial scale worldwide. This redundancy in the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions (high temperature, ambient pressure in sealed vessels) makes the process highly scalable from pilot plant to multi-ton commercial production. This scalability ensures that suppliers can reliably meet increasing volume requirements as a drug candidate progresses from clinical trials to commercial launch, securing the long-term availability of critical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is vastly superior to traditional alkylation. The absence of carcinogenic methylating agents creates a safer working environment for plant personnel and reduces the liability associated with handling Schedule 1 substances. The waste stream is primarily aqueous and organic solvent, devoid of heavy metals, which simplifies wastewater treatment and lowers the cost of environmental compliance. This "green" profile is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies who are committed to sustainability goals. Consequently, manufacturers adopting this technology position themselves as preferred vendors for eco-conscious clients, enhancing their brand reputation and market access in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-methylation technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. For further technical discussions or specific data requests, our team is prepared to provide comprehensive support tailored to your project requirements.
Q: What are the primary advantages of this catalyst-free N-methylation method over traditional alkyl halide methods?
A: This method eliminates the use of toxic and carcinogenic alkylating agents such as methyl iodide or dimethyl sulfate. Furthermore, it avoids the need for expensive transition metal catalysts, significantly simplifying the purification process and reducing heavy metal contamination risks in the final pharmaceutical intermediate.
Q: What is the typical yield range for this synthesis across different substrates?
A: The patent data demonstrates robust yields across a wide variety of substrates. Simple anilines achieve yields up to 92%, while more complex heterocyclic systems like tetrahydroquinolines and indolines consistently deliver yields between 70% and 88%, indicating high process reliability.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial suitability. It utilizes common, inexpensive reagents (formic acid, triethylamine, DMSO) and operates under relatively straightforward conditions (130-160°C) without requiring specialized high-pressure hydrogenation equipment, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methylated Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that efficient N-methylation technologies play in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering high-purity N-methylated aromatic amines that meet stringent purity specifications, utilizing rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our capability to implement catalyst-free, green chemistry solutions like the formic acid-mediated method described above underscores our dedication to sustainable and cost-effective manufacturing practices that align with the evolving needs of the global pharmaceutical industry.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your upcoming projects. Whether you require custom synthesis of complex heterocyclic intermediates or large-scale production of commodity amines, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis for your specific target molecules. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized processes can enhance your supply chain efficiency and reduce your overall development timelines.
