Advanced Synthesis of All-Carbon Spiro-Fused Polycyclic Compounds for High-Performance Materials
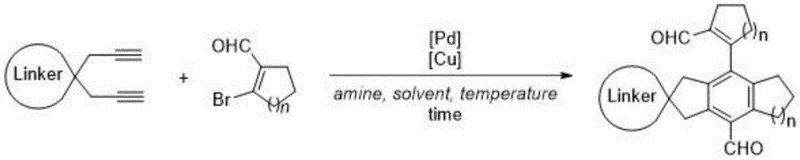
The chemical industry is constantly seeking robust methodologies to construct complex molecular architectures, particularly those with rigid spiro-fused frameworks that impart unique physical and chemical properties. Patent CN112358388A introduces a groundbreaking approach to synthesizing all-carbon spiro-fused polycyclic compounds, addressing a significant gap in current synthetic capabilities. These compounds are not merely academic curiosities; they serve as critical precursors for polyols used in high-performance polymers such as polyethers, polyesters, and polyurethanes, as well as promising candidates for organic optoelectronic materials. The disclosed technology leverages a sophisticated three-component tandem reaction sequence, integrating Sonogashira coupling, [4+2] cyclization, and a subsequent aldehyde group migration. This innovation represents a paradigm shift from traditional multi-step syntheses, offering a streamlined pathway that enhances both efficiency and structural diversity. For R&D directors and procurement specialists alike, this patent outlines a route that balances molecular complexity with operational simplicity, utilizing accessible starting materials and cost-effective catalytic systems to generate high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of all-carbon spiro-condensed compounds has been fraught with synthetic challenges due to the inherent rigidity and steric congestion of the target molecular skeletons. Conventional strategies often necessitate lengthy multi-step sequences, each requiring distinct reaction conditions, isolation procedures, and purification protocols, which cumulatively drive up production costs and extend lead times. Many existing methods rely on harsh reaction conditions, such as extreme temperatures or highly reactive reagents, which can compromise functional group tolerance and lead to significant formation of impurities. Furthermore, the lack of atom economy in traditional stepwise approaches results in substantial waste generation, posing environmental compliance issues for large-scale manufacturing. The difficulty in controlling stereochemistry and regioselectivity during the formation of the quaternary spiro-center often leads to low overall yields, making these processes economically unviable for commercial applications in the polymer and fine chemical sectors. Consequently, there has been a persistent demand for a more direct and efficient synthetic methodology that can overcome these structural and operational bottlenecks.
The Novel Approach
The methodology described in patent CN112358388A offers a transformative solution by employing a one-pot cascade reaction that efficiently assembles complex spiro-fused frameworks from simple precursors. This novel approach utilizes 1,1-di(propargyl) substituted cycloalkanes and beta-bromocycloalkene aldehydes as building blocks, which are reacted in the presence of a dual palladium and copper catalytic system. By operating under relatively mild thermal conditions of 70-80°C in a nitrogen atmosphere, the process ensures high functional group compatibility while minimizing energy consumption. The use of diisopropylamine as a base and ethyl acetate as the preferred solvent further underscores the practicality of this method, as these reagents are inexpensive and widely available in the global supply chain. This streamlined protocol not only reduces the number of unit operations required but also significantly improves the overall yield, with specific examples demonstrating isolated yields ranging from 49% to 70%. The ability to access a wide range of substrates with varying ring sizes and substituents highlights the versatility of this chemistry, making it an attractive option for the commercial scale-up of complex polymer additives and specialty intermediates.
Mechanistic Insights into Pd/Cu Catalyzed Cascade Cyclization
The success of this synthetic strategy lies in the intricate interplay between the palladium and copper catalysts, which orchestrate a seamless sequence of bond-forming events. The reaction initiates with a Sonogashira cross-coupling between the terminal alkyne moieties of the cycloalkane substrate and the vinyl bromide functionality of the aldehyde component. This step generates a transient diyne intermediate that is primed for the subsequent cyclization event. Following the coupling, the system undergoes an intramolecular [4+2] cycloaddition, likely proceeding through a cyclic allene intermediate, which constructs the central benzene ring skeleton with full substitution. This pericyclic process is critical for establishing the rigid spiro-core that defines the physical properties of the final product. The elegance of this mechanism is further enhanced by a spontaneous 1,3-aldehyde migration that occurs post-cyclization, stabilizing the aromatic system and delivering the final dicarbonyl-functionalized spiro compound. Understanding this mechanistic pathway is vital for high-purity organic optoelectronic materials development, as it allows chemists to predict and control the formation of specific isomers and minimize the generation of structural impurities that could detrimentally affect material performance.
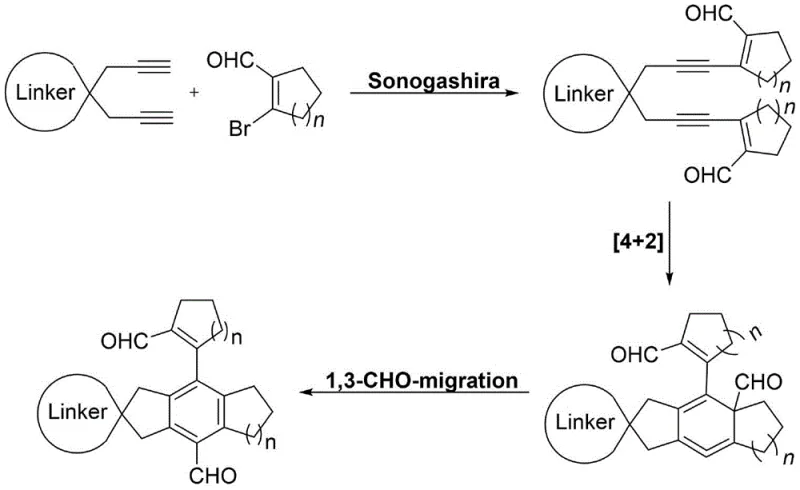
From an impurity control perspective, the choice of ligands and catalyst loading plays a pivotal role in ensuring the fidelity of the cascade. The patent specifies the use of tetrakis(triphenylphosphine)palladium and tris(triphenylphosphine)copper(I) bromide, which provide the necessary electronic and steric environment to facilitate the coupling without promoting unwanted side reactions such as homocoupling of the alkynes. The moderate reaction temperature of 70-80°C is sufficient to drive the cyclization and migration steps without causing thermal degradation of the sensitive aldehyde groups or the newly formed spiro-junction. Additionally, the use of ethyl acetate as the solvent contributes to a cleaner reaction profile, as it effectively solubilizes the organic components while remaining inert under the basic reaction conditions. This precise control over the reaction parameters ensures that the resulting crude product contains a high proportion of the desired target molecule, thereby simplifying the downstream purification process and reducing the burden on quality control laboratories to detect and quantify trace impurities in the final batch.
How to Synthesize All-Carbon Spiro Polycyclic Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized reaction parameters to maximize yield and reproducibility. The process begins by establishing an inert nitrogen atmosphere to prevent oxidation of the sensitive catalysts and intermediates. The stoichiometry is critical, with a molar ratio of 100:240:2:4:500 recommended for the cycloalkane, aldehyde, palladium catalyst, copper catalyst, and base, respectively. This specific balance ensures that the coupling reaction proceeds to completion while maintaining sufficient catalytic turnover. The detailed standardized synthesis steps, including specific workup procedures and chromatographic conditions, are outlined below to guide technical teams in replicating these results effectively.
- Under nitrogen atmosphere, mix 1,1-di(propargyl) substituted cycloalkane, beta-bromocycloalkene aldehyde, Pd(PPh3)4, and Cu(PPh3)3Br in ethyl acetate.
- Add diisopropylamine as the base and stir the reaction mixture at 70-80°C for 24 to 72 hours depending on substrate reactivity.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the manufacturing workflow, which directly translates to operational efficiencies and cost reduction in polymer additive manufacturing. By consolidating multiple synthetic transformations into a single vessel, the process eliminates the need for intermediate isolation, drying, and transfer steps, thereby reducing labor costs and minimizing material loss. The reliance on commodity chemicals such as ethyl acetate and diisopropylamine, rather than exotic or hazardous solvents, ensures a stable and predictable supply chain, mitigating the risks associated with raw material volatility. Furthermore, the use of earth-abundant transition metal catalysts like copper, alongside palladium, helps to keep catalyst costs manageable, especially when considering the potential for catalyst recovery and recycling in larger-scale operations.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and reduced processing time. Unlike traditional methods that may require cryogenic conditions or expensive protecting group strategies, this cascade reaction operates at moderate temperatures using straightforward reagents. The elimination of multiple purification stages between steps drastically cuts down on solvent consumption and waste disposal costs. Additionally, the high selectivity of the reaction means that less raw material is wasted on byproduct formation, leading to a more efficient utilization of inputs. This lean manufacturing approach allows producers to offer competitive pricing for these high-value spiro intermediates, making them accessible for broader applications in the plastics and coatings industries without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route contributes significantly to supply chain resilience. The starting materials, specifically the propargyl-substituted cycloalkanes and bromocycloalkene aldehydes, are structurally simple and can be sourced from multiple vendors or synthesized via established pathways. This diversification of supply sources reduces dependency on single-point failures and ensures continuity of production even during market fluctuations. The mild reaction conditions also mean that the process can be executed in standard stainless steel reactors without the need for specialized glass-lined equipment or extreme pressure vessels, facilitating easier technology transfer between different manufacturing sites. This flexibility is crucial for maintaining consistent delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram or tonne scale is facilitated by the homogeneous nature of the reaction mixture and the absence of gas-evolving steps that could complicate reactor headspace management. The use of ethyl acetate, a solvent with a favorable environmental profile compared to chlorinated alternatives, aligns with increasingly stringent global regulations regarding volatile organic compound (VOC) emissions and worker safety. The simplified workup procedure, involving a standard aqueous quench and extraction, generates waste streams that are easier to treat and dispose of in compliance with environmental standards. This green chemistry aspect not only reduces the regulatory burden on the manufacturer but also enhances the sustainability credentials of the final product, a factor that is becoming increasingly important for end-users in the consumer goods and automotive industries who are striving to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these all-carbon spiro-fused polycyclic compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. These insights are intended to clarify the operational feasibility and strategic value of integrating this technology into existing production portfolios.
Q: What are the key advantages of this Pd/Cu catalyzed cascade method?
A: This method utilizes a one-pot tandem reaction strategy that combines Sonogashira coupling, [4+2] cyclization, and aldehyde migration. It eliminates the need for multiple isolation steps, uses relatively inexpensive catalysts like Pd(PPh3)4, and operates under mild conditions (70-80°C), significantly simplifying the production process compared to traditional multi-step syntheses.
Q: Can this synthesis be scaled for industrial polymer additive production?
A: Yes, the process demonstrates excellent scalability potential. It employs common organic solvents like ethyl acetate and standard bases like diisopropylamine, which are readily available in bulk. The reaction tolerates a wide range of substrates, allowing for the production of diverse spiro-fused structures essential for tuning the properties of polyols used in polyurethanes and polyesters.
Q: How is product purity ensured in this complex cascade reaction?
A: High purity is achieved through the specificity of the palladium and copper catalytic system, which minimizes side reactions. Furthermore, the final purification step involves silica gel column chromatography using an ethyl acetate and petroleum ether gradient, effectively separating the target all-carbon spiro polycyclic compounds from any unreacted starting materials or minor byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable All-Carbon Spiro Polycyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced cascade reactions in driving innovation within the fine chemical and polymer sectors. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require these spiro-compounds as precursors for next-generation polyurethanes or as building blocks for organic electronics, our manufacturing infrastructure is designed to handle complex synthetic challenges with precision and reliability.
We invite you to collaborate with us to explore how this patented technology can optimize your supply chain and enhance your product portfolio. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and competitiveness in your market.
