Advanced Suzuki Coupling Strategy for High-Purity Biphenyl Glycine Pharmaceutical Intermediates
Advanced Suzuki Coupling Strategy for High-Purity Biphenyl Glycine Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for complex amino acid derivatives, particularly those serving as critical building blocks for protease inhibitors and other therapeutic agents. Patent CN111848430B, published in December 2022, introduces a groundbreaking synthetic method for 2-([1,1'-biphenyl]-4-yl)-2-glycine compounds that addresses significant safety and efficiency bottlenecks found in prior art. This technology leverages a microwave-assisted Suzuki coupling reaction to construct the biaryl backbone, followed by a mild deprotection step, offering a superior alternative to traditional aldehyde-based condensations. For R&D directors and procurement specialists, this patent represents a pivotal shift towards safer, higher-yielding processes that align with modern green chemistry principles while ensuring the supply continuity of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
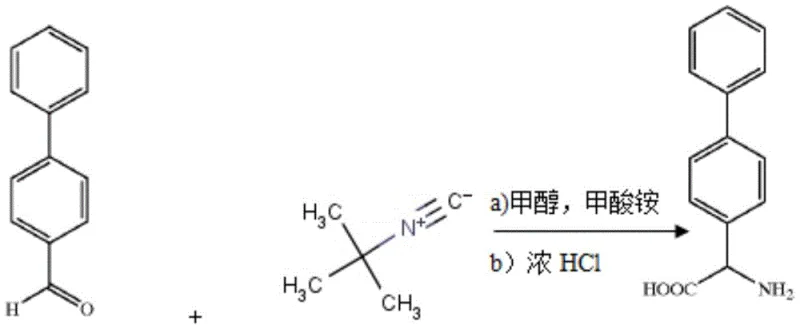
Historically, the synthesis of 2-([1,1'-biphenyl]-4-yl)-2-glycine derivatives has relied heavily on routes exemplified by patent WO2003010130, which utilizes 4-biphenylcarboxaldehyde as the primary starting material. As illustrated in the reaction scheme above, this conventional pathway involves heating the aldehyde with ammonium formate and tert-butyl isocyanate in methanol, followed by a harsh hydrolysis step using concentrated hydrochloric acid at 100°C for 24 hours. This methodology presents severe drawbacks, primarily stemming from the use of tert-butyl isocyanate, a substance known for its high toxicity and flammability, which poses significant safety risks during large-scale manufacturing. Furthermore, the requirement for prolonged heating at elevated temperatures with strong acids not only compromises equipment integrity but also leads to potential side reactions that degrade product purity and reduce overall yield, making it an unsustainable choice for modern cost reduction in API manufacturing.
The Novel Approach
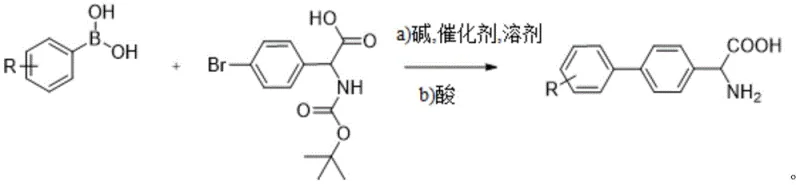
In stark contrast, the novel approach disclosed in CN111848430B employs a strategic Suzuki-Miyaura cross-coupling reaction between a substituted phenylboronic acid (Formula I) and a protected bromo-glycine derivative (Formula II). As depicted in the general reaction scheme, this method constructs the biphenyl core under inert gas protection using a palladium catalyst and a base, critically enhanced by microwave assistance. This innovation allows the reaction to proceed under much milder conditions, typically between 80-100°C, and significantly shortens the reaction timeline compared to the multi-day protocols of the past. By shifting the bond-forming strategy from aldehyde condensation to transition metal catalysis, the process eliminates the need for hazardous isocyanides and aggressive acid hydrolysis, thereby establishing a reliable pharmaceutical intermediate supplier pathway that prioritizes both operator safety and environmental compliance without sacrificing chemical efficiency.
Mechanistic Insights into Microwave-Assisted Suzuki Coupling
The core of this technological advancement lies in the precise orchestration of the palladium-catalyzed cycle, facilitated by dielectric heating. The reaction initiates with the oxidative addition of the aryl bromide (Formula II) to the zero-valent palladium species, such as Pd(PPh3)4, forming an organopalladium intermediate. The introduction of microwave energy at 70-90W for a brief 2-4 minute window serves to rapidly activate the catalytic species and overcome the activation energy barrier more efficiently than conventional conductive heating. Following this activation, the transmetallation step occurs where the phenylboronic acid, activated by the base (e.g., K2CO3), transfers its aryl group to the palladium center. Finally, reductive elimination releases the coupled product, regenerating the active catalyst. This mechanism ensures high selectivity for the biaryl bond formation, minimizing homocoupling byproducts that often plague traditional methods, thus delivering the high-purity OLED material or pharmaceutical grade precursors required by discerning clients.
Impurity control is inherently managed through the mildness of the subsequent deprotection step. Unlike the prior art which subjects the intermediate to 100°C concentrated HCl, this patent specifies deprotection using trifluoroacetic acid (TFA) or dilute HCl at temperatures ranging from 15-35°C. This gentle removal of the tert-butoxycarbonyl (Boc) protecting group prevents racemization and thermal degradation of the sensitive amino acid backbone. The use of solvents like THF and toluene, combined with aqueous base solutions, creates a biphasic system that facilitates efficient mass transfer during the coupling phase. By optimizing the molar ratios, specifically maintaining a slight excess of the boronic acid (1 to 1.5:1), the process drives the equilibrium towards completion, ensuring that residual starting materials are minimized and downstream purification burdens are significantly reduced for commercial scale-up of complex polymer additives or drug substances.
How to Synthesize 2-([1,1'-biphenyl]-4-yl)-2-glycine Efficiently
The practical implementation of this synthesis requires careful attention to the microwave parameters and catalyst loading to maximize the benefits observed in the patent examples. The process begins with the suspension of the boronic acid and the protected bromo-amino acid in a mixture of THF and aqueous potassium carbonate under a nitrogen atmosphere. Upon addition of the palladium catalyst, the mixture is subjected to microwave irradiation before being stirred at reflux. This specific sequence is crucial for activating the catalyst without causing premature decomposition. Following the coupling, the workup involves standard extraction and chromatographic purification to isolate the Boc-protected intermediate, which is then treated with acid to reveal the free amine. For detailed operational parameters and specific stoichiometric calculations tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- Perform a Suzuki reaction between a substituted phenylboronic acid and a protected bromo-glycine derivative using a palladium catalyst and base under microwave assistance.
- Maintain reaction temperature between 80-100°C for 6-10 hours following a brief 2-4 minute microwave activation period.
- Execute a deprotection step using trifluoroacetic acid or hydrochloric acid at mild temperatures (15-35°C) to obtain the final amino acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Suzuki-based methodology offers profound strategic benefits beyond mere chemical elegance. The primary advantage is the substantial cost savings derived from the elimination of hazardous reagents like tert-butyl isocyanate, which require specialized handling, storage, and disposal protocols that inflate operational expenditures. By replacing these with commodity chemicals such as phenylboronic acids and standard palladium catalysts, the raw material costs are stabilized, and the supply chain becomes more resilient against regulatory shocks associated with toxic substance management. Furthermore, the significant reduction in reaction time—from days to hours—enhances the throughput of manufacturing facilities, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations without the need for capital-intensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the high yields consistently reported across various substituents, often exceeding 81% and reaching up to 96% in optimized examples. This efficiency directly translates to lower cost per kilogram of the final active ingredient, as less raw material is wasted in side reactions or lost during extensive purification steps necessitated by lower-yielding routes. Additionally, the avoidance of high-temperature acid hydrolysis reduces energy consumption and extends the lifespan of reactor vessels and linings, contributing to long-term capital preservation and reduced maintenance downtime for the production line.
- Enhanced Supply Chain Reliability: The reliance on widely available phenylboronic acids ensures a stable supply of key starting materials, mitigating the risk of shortages that can occur with specialized aldehyde derivatives or isocyanides. The modular nature of the Suzuki reaction means that a single protected glycine intermediate can be coupled with a diverse array of boronic acids to produce a library of analogues, providing flexibility in production planning. This versatility allows manufacturers to pivot quickly between different product grades or custom derivatives, ensuring continuous supply even when specific market demands shift unexpectedly.
- Scalability and Environmental Compliance: From an environmental perspective, the milder reaction conditions and the absence of highly toxic reagents simplify waste stream management, reducing the burden on effluent treatment plants and lowering the overall environmental footprint of the manufacturing process. The scalability is further supported by the use of standard organic solvents like THF and ethyl acetate, which are easily recovered and recycled in industrial settings. This alignment with green chemistry principles not only meets stringent regulatory requirements but also enhances the corporate sustainability profile, a critical factor for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this technology for their specific production needs.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: The novel method eliminates the use of toxic and flammable tert-butyl isocyanate and avoids high-temperature reactions with concentrated hydrochloric acid, significantly improving operational safety profiles.
Q: How does microwave assistance impact the reaction efficiency?
A: Microwave assistance for 2-4 minutes effectively promotes the Suzuki coupling reaction, reducing overall reaction time and improving yields to over 81% compared to conventional heating methods.
Q: Is this process scalable for commercial API intermediate production?
A: Yes, the process utilizes commercially available phenylboronic acids and standard palladium catalysts, making it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-([1,1'-biphenyl]-4-yl)-2-glycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the potential of the microwave-assisted Suzuki coupling route described in CN111848430B and is fully prepared to translate this laboratory-scale innovation into robust commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of biphenyl glycine intermediate delivered meets the highest standards required for downstream API synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this safer, more efficient route for your organization. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex organic synthesis can drive value and reliability in your supply chain.
