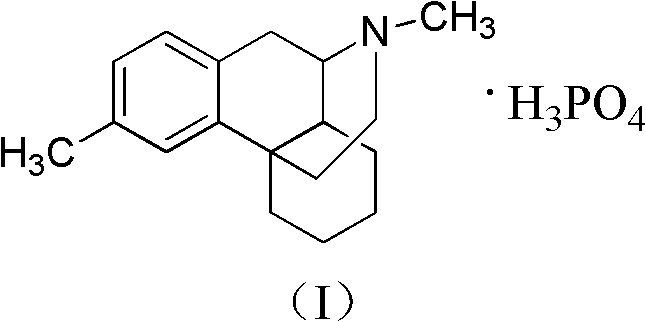
Advanced Synthesis of Dimemorfan Phosphate Intermediates for Commercial Scale-Up
The pharmaceutical landscape for antitussive agents continues to evolve, with Dimemorfan Phosphate standing out as a potent, non-addictive cough suppressant developed originally by Yamanouchi Pharmaceutical. Central to the efficient manufacturing of this active pharmaceutical ingredient is the robust synthesis of its key precursor, dl-(4-methylphenyl)-2-methyl-1,2,3,4,5,6,7,8-octahydroisoquinoline. Patent CN102267944A introduces a transformative methodology that addresses historical bottlenecks in producing this complex intermediate. By shifting away from scarce starting materials toward abundant commodity chemicals, this innovation offers a pathway to significantly enhanced supply chain resilience and cost efficiency. The structural integrity of the final API relies heavily on the stereochemical and chemical purity of this intermediate, making the optimization of its synthesis a critical priority for R&D and procurement teams alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
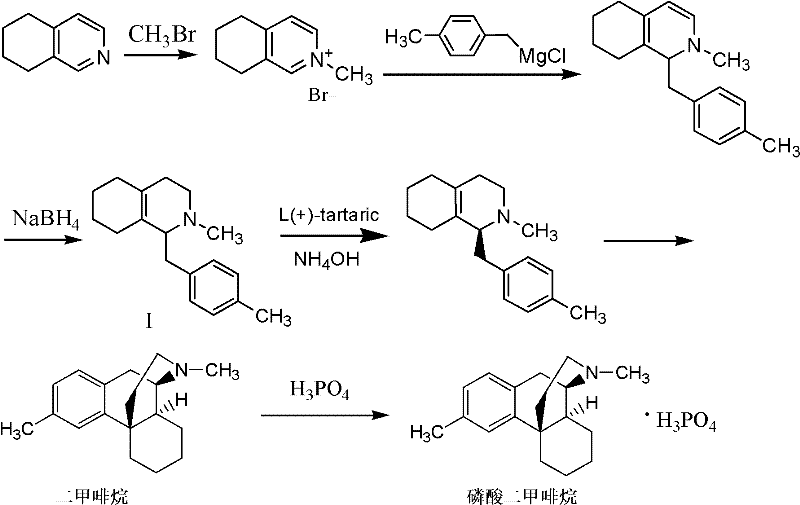
Historically, the synthesis of the dimemorfan intermediate relied on pathways described in literature such as Chem. Pharm. Bull. 1972 and US Patent 3786054, which utilized 5,6,7,8-tetrahydroisoquinoline as the foundational starting material. This approach presents severe logistical and economic challenges for modern manufacturers, primarily because this specific tetrahydroisoquinoline derivative is not domestically available in many regions and must be imported at exorbitant costs. Furthermore, the conventional route necessitates the use of methyl bromide, a highly toxic gaseous reagent that imposes stringent safety regulations and requires specialized containment equipment, thereby inflating capital expenditure. The reliance on Grignard reagents introduces additional complexity due to their extreme sensitivity to moisture and the need for strictly anhydrous conditions, which often leads to unstable intermediates and difficult purification processes. These factors collectively result in lower yields, inconsistent product quality, and a manufacturing footprint that is increasingly difficult to justify in a cost-sensitive global market.
The Novel Approach
In stark contrast, the methodology disclosed in CN102267944A reconstructs the synthetic logic by employing 4-methylphenylacetic acid and cyclohexene ethylamine as the primary building blocks, both of which are readily accessible commodity chemicals with stable supply chains. This novel route bypasses the need for the expensive tetrahydroisoquinoline scaffold entirely, constructing the core ring system through a strategic sequence of acylation, cyclodehydration, N-methylation, and reduction. By replacing the hazardous methyl bromide gas with safer liquid alkylating agents like methyl iodide or dimethyl sulfate, the process dramatically improves operational safety and reduces the regulatory burden associated with handling toxic gases. The reaction conditions are notably milder, avoiding the extreme constraints of Grignard chemistry, which translates to more stable intermediates that are easier to isolate and purify. This fundamental shift in synthetic strategy not only lowers the raw material cost base but also enhances the overall robustness of the manufacturing process, making it ideally suited for large-scale industrial application.
Mechanistic Insights into Cyclodehydration and Reduction Strategy
The core of this innovative synthesis lies in the efficient construction of the octahydroisoquinoline ring system via a Bischler-Napieralski type cyclodehydration. The process begins with the conversion of 4-methylphenylacetic acid into its corresponding acyl chloride using thionyl chloride, which is then condensed with cyclohexene ethylamine to form a stable amide intermediate. This amide serves as the precursor for the critical ring-closing step, where a dehydrating agent such as phosphorus oxychloride (POCl3) facilitates the intramolecular cyclization to generate the imine intermediate. The use of POCl3 in a solvent like toluene under reflux conditions ensures high conversion rates while maintaining a manageable reaction profile that minimizes side reactions. Following cyclization, the nitrogen atom is methylated to form a quaternary ammonium salt, which activates the system for the final reduction step. This sequential activation allows for precise control over the introduction of the methyl group, ensuring the correct substitution pattern required for the biological activity of the final API.
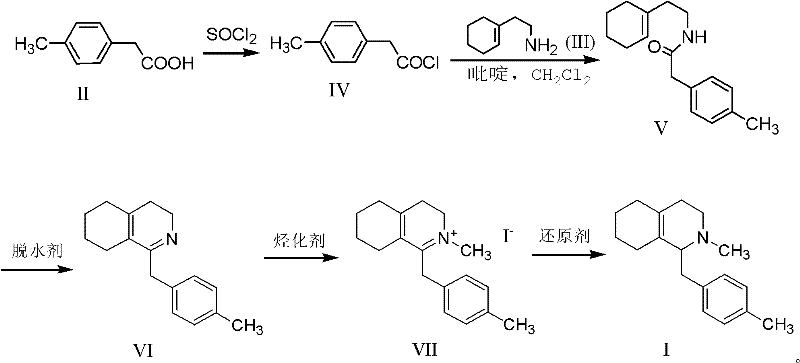
Impurity control is inherently superior in this new pathway due to the stability of the intermediates and the selectivity of the reduction step. The final transformation involves the reduction of the quaternary ammonium salt using sodium borohydride, a mild and selective reducing agent that effectively converts the iminium species to the desired amine without affecting other sensitive functional groups on the aromatic ring. Unlike the older methods where unstable intermediates could degrade into complex mixtures requiring difficult chromatographic separations, this route produces intermediates that can be purified through standard crystallization or extraction techniques. The avoidance of transition metal catalysts or harsh organometallic reagents means there is no risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates. Consequently, the final product exhibits high chemical purity and a clean impurity profile, reducing the burden on downstream processing and ensuring consistent quality for the final drug substance.
How to Synthesize dl-(4-methylphenyl)-2-methyl-1,2,3,4,5,6,7,8-octahydroisoquinoline Efficiently
The synthesis of this critical intermediate is achieved through a streamlined four-step sequence that prioritizes operational simplicity and high yield. Starting from commercially available 4-methylphenylacetic acid, the process involves acylation to form an amide, followed by cyclodehydration to close the ring, N-methylation to introduce the methyl group, and finally reduction to saturate the system. This logical progression allows for the isolation of stable intermediates at each stage, facilitating rigorous quality control before proceeding to the next step. For detailed standardized operating procedures and specific reaction parameters, please refer to the technical guide below.
- Convert 4-methylphenylacetic acid to acyl chloride using thionyl chloride, then react with cyclohexene ethylamine to form the amide intermediate.
- Perform cyclodehydration on the amide using phosphorus oxychloride in toluene to generate the imine intermediate.
- Execute N-methylation followed by sodium borohydride reduction to yield the final octahydroisoquinoline intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route represents a significant opportunity to optimize the cost structure and reliability of the dimemorfan supply chain. By eliminating the dependency on imported, high-cost specialty starting materials like 5,6,7,8-tetrahydroisoquinoline, manufacturers can drastically reduce their raw material expenditure and mitigate the risks associated with single-source suppliers. The substitution of toxic methyl bromide gas with safer liquid reagents not only lowers the cost of safety compliance and waste disposal but also simplifies the logistics of reagent storage and handling. Furthermore, the use of common industrial solvents and reagents ensures that the process can be easily scaled up in existing multipurpose facilities without requiring significant capital investment in new infrastructure. These factors combine to create a more resilient and cost-effective supply chain capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The shift to commodity raw materials such as 4-methylphenylacetic acid fundamentally alters the cost basis of the intermediate, removing the premium associated with imported specialty chemicals. The elimination of expensive and hazardous reagents like methyl bromide further reduces operational costs related to safety monitoring, specialized equipment maintenance, and hazardous waste treatment. Additionally, the improved stability of intermediates leads to higher overall yields and reduced material loss during purification, contributing to substantial cost savings throughout the production cycle. This economic efficiency makes the final API more competitive in the global market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly strengthened by the use of widely available starting materials that are produced by multiple suppliers globally, reducing the risk of supply disruptions. The removal of toxic gases from the process simplifies regulatory compliance and transportation logistics, ensuring smoother operations across international borders. The robustness of the synthetic route means that production schedules are less likely to be impacted by batch failures or quality deviations, providing a more predictable and reliable supply of the intermediate for downstream API synthesis. This reliability is crucial for maintaining continuous production lines and meeting delivery commitments to pharmaceutical partners.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal catalysts make this process inherently scalable from pilot plant to commercial tonnage production without encountering significant engineering hurdles. The use of standard organic solvents and reagents simplifies waste stream management, allowing for more efficient recycling and disposal protocols that align with modern environmental standards. The process generates fewer hazardous byproducts compared to traditional methods, reducing the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of dimemorfan phosphate intermediates, based on the specific advancements detailed in the patent literature. These insights are designed to clarify the operational benefits and quality advantages of the new methodology for stakeholders involved in process development and sourcing. Understanding these distinctions is key to evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary advantages of the new synthesis route over conventional methods?
A: The new route eliminates the need for expensive, imported 5,6,7,8-tetrahydroisoquinoline and toxic methyl bromide gas, utilizing readily available commodity chemicals instead.
Q: How does this process improve impurity control?
A: By avoiding harsh Grignard conditions and unstable intermediates, the new method ensures stable product quality and simplifies purification, leading to higher overall purity.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild, reagents are common industrial chemicals, and the process avoids hazardous gases, making it highly scalable and safe for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimemorfan Phosphate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the supply of essential pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in process optimization allows us to adapt the patented methodology to maximize yield and minimize impurities, providing our partners with a consistent and reliable source of material.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these technological advancements for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of dimemorfan intermediates can drive value and efficiency in your operations.
