Advanced Chiral Pyridine N,B-Ligands for Scalable Iridium-Catalyzed Asymmetric Synthesis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for high-purity single enantiomers in pharmaceutical and fine chemical manufacturing. A significant breakthrough in this domain is documented in patent CN112724168B, which discloses a novel chiral pyridine-derived N,B ligand and its preparation method. This technology addresses long-standing challenges in ligand design by introducing a rigid fused ring structure that offers superior stability and tunability compared to existing solutions. For R&D directors and procurement specialists, this innovation represents a pivotal shift towards more reliable and efficient catalytic systems for iridium-catalyzed asymmetric boronation. The ligand's unique architecture not only enhances catalytic activity but also ensures robust enantioselectivity, making it a critical component for the synthesis of complex chiral aryl boron compounds used in drug discovery.
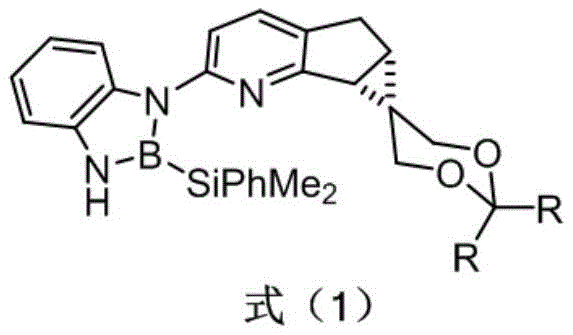
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral pyridine ligands has been hindered by structural rigidity and a lack of universality. Prominent examples include the chiral bipyridine ligand PINDY, synthesized from natural monoterpene pinene, and the planar chiral pyridine ligand BPY containing a ferrocene skeleton. While PINDY has found applications in cyclopropanation and allylic oxidation, its structural modification is notoriously difficult, restricting further optimization of its catalytic properties. Similarly, the introduction of chiral elements onto planar structures like BPY presents significant synthetic challenges, often leading to contradictions between catalytic activity and selectivity due to steric hindrance. These conventional ligands often suffer from poor modifiability and difficulties in modular synthesis, creating a bottleneck for the design of next-generation catalysts required for modern asymmetric transformations.
The Novel Approach
In contrast, the technology outlined in patent CN112724168B introduces a paradigm shift through the design of a chiral pyridine derivative featuring a rigid fused ring structure framework. This novel approach leverages an N and B bidentate coordination mode that significantly improves the catalytic activity of the central metal iridium. The entire ligand possesses a relatively rigid and stable chiral space, ensuring that the chiral environment remains unchanged during the reaction process. Crucially, the introduction of five-membered and three-membered rings manages steric hindrance effectively without obstructing the ortho position of the pyridine, thereby preserving the metal's catalytic activity. This design allows for the derivation of a series of ligands with adjustable structural height, offering a versatile platform for optimizing enantioselectivity in diverse chemical environments.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Boronation
The efficacy of this chiral N,B ligand stems from its ability to form a stable complex with metal iridium that resists conformational changes under reaction conditions. The rigid fused ring skeleton acts as a robust scaffold, locking the chiral information in place and transferring it efficiently to the substrate. During the iridium-catalyzed asymmetric boronation, the ligand coordinates with the metal center to create a well-defined chiral pocket. This pocket discriminates between enantiotopic faces of the substrate, such as diaryl pyridines, facilitating the selective insertion of the boron species. The stability of the complex prevents the degradation of chiral integrity, which is a common failure mode in less rigid ligand systems, thereby ensuring consistent high yields and enantiomeric excess across multiple batches.
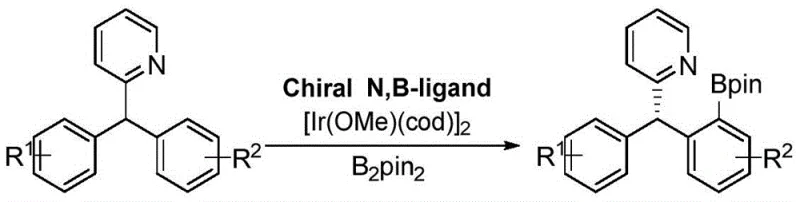
Furthermore, the impurity control mechanism is inherently built into the ligand's structural design. The specific spatial arrangement of the N and B atoms, combined with the steric bulk of the fused rings, disfavors the formation of side products that typically arise from non-selective pathways. By maintaining a tight coordination sphere around the iridium center, the ligand minimizes the opportunity for off-cycle reactions or catalyst decomposition. This results in a cleaner reaction profile with fewer by-products, simplifying downstream purification processes. The ability to tune the R groups on the ligand structure further allows chemists to fine-tune the electronic and steric properties, optimizing the system for specific substrates and minimizing the formation of regioisomers or other impurities that could compromise the purity of the final pharmaceutical intermediate.
How to Synthesize Chiral Pyridine Derived N,B Ligand Efficiently
The synthesis of this advanced ligand follows a logical, multi-step sequence that transforms readily available starting materials into a high-value chiral catalyst. The process begins with the construction of the chiral chloropyridine core, followed by functional group manipulations to install the necessary N,B-coordination motifs. Each step is optimized for yield and stereochemical integrity, utilizing standard laboratory equipment and reagents. The route is designed to be robust, allowing for the isolation of key intermediates like the chiral diol and the protected pyridine derivative. For detailed operational parameters and specific stoichiometric ratios required for reproducible results, please refer to the standardized synthesis protocol provided below.
- Perform cyclopropanation of bromo-cyclopentenone with diethyl malonate to generate the racemic ketone intermediate.
- Resolve the racemic ketone using (R)-tert-butyl sulfinamide and tetraethyl titanate to obtain chiral ketone enantiomers.
- Convert the chiral ketone to an oxime, reduce to an enamide, and cyclize with POCl3 to form the chiral chloropyridine derivative.
- Reduce the ester groups to diols, protect with ketal, and couple with o-phenylenediamine to form the chiral pyridine precursor.
- React the chiral pyridine derivative with PhMe2Si-B(NiPr2)2 in toluene at 125-135°C to yield the final chiral N,B ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel ligand technology offers substantial benefits for supply chain reliability and cost management in fine chemical manufacturing. The synthesis route relies on commodity chemicals such as bromo-cyclopentenone and diethyl malonate, which are widely available and cost-effective, reducing the risk of raw material shortages. The process avoids the use of exotic or prohibitively expensive chiral pools that often plague traditional ligand synthesis, thereby stabilizing the cost of goods sold. Additionally, the reaction conditions, such as refluxing in toluene or dichloroethane, are compatible with standard industrial reactors, eliminating the need for specialized high-pressure or cryogenic equipment.
- Cost Reduction in Manufacturing: The streamlined synthesis pathway eliminates the need for complex structural modifications often required by older ligand generations. By utilizing a modular approach where the chiral core is established early and then derivatized, manufacturers can achieve significant economies of scale. The high yields reported in the patent examples indicate efficient atom economy, reducing waste disposal costs. Furthermore, the elimination of transition metal catalysts in certain steps or the use of earth-abundant metals in the ligand synthesis itself contributes to a lower overall environmental footprint and reduced expenditure on precious metal recovery systems.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials ensures a consistent supply chain unaffected by the volatility of niche natural product markets. The robustness of the synthetic route means that production can be scaled up rapidly to meet fluctuating demand without compromising quality. The ligand's stability also implies a longer shelf life, reducing inventory write-offs and allowing for strategic stockpiling. This reliability is crucial for pharmaceutical companies that require uninterrupted supply of key intermediates to maintain their own production schedules and regulatory compliance.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction steps that translate well from gram-scale laboratory synthesis to multi-ton commercial production. The use of common solvents facilitates solvent recovery and recycling programs, aligning with green chemistry principles. The high selectivity of the resulting catalytic system reduces the generation of hazardous waste associated with separating racemic mixtures. This alignment with environmental regulations simplifies permitting processes and reduces the long-term liability associated with waste management, making it an attractive option for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They cover aspects ranging from structural properties to application scope, providing a quick reference for decision-makers evaluating this technology for their specific processes.
Q: What are the structural advantages of this new chiral N,B ligand over traditional pyridine ligands?
A: Unlike traditional ligands like PINDY which suffer from difficult structural modification, this novel ligand features a rigid fused ring framework with adjustable structural height. This design ensures a stable chiral environment during coordination with iridium, preventing conformational changes that could lower enantioselectivity, while avoiding excessive steric hindrance at the ortho position.
Q: How does this ligand improve the efficiency of asymmetric boronation reactions?
A: As an N and B bidentate ligand, it significantly enhances the catalytic activity of the central metal iridium. The rigid structure maintains a consistent chiral space throughout the reaction, leading to excellent reaction activity and high enantioselectivity (up to 96% ee in tested substrates) for chiral aryl boron compounds.
Q: Is the synthesis of this ligand scalable for industrial production?
A: Yes, the synthesis utilizes readily available raw materials such as bromo-cyclopentenone and diethyl malonate. The process involves standard organic transformations like refluxing in common solvents (toluene, DCE) and column chromatography purification, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral N,B-Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral pyridine-derived N,B ligand described in patent CN112724168B for the synthesis of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this synthesis, ensuring stringent purity specifications and rigorous QC labs to verify the enantiomeric excess and chemical purity of every batch. We are committed to delivering this advanced catalyst technology to support your R&D and manufacturing goals with unwavering quality.
We invite you to engage with our technical procurement team to discuss how this ligand can optimize your asymmetric boronation processes. By requesting a Customized Cost-Saving Analysis, you can gain insights into the specific economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this superior catalytic system is seamless, compliant, and commercially viable.
