Scalable Manufacturing of Emivirine Intermediates Using Novel Meldrum’s Acid Chemistry
Scalable Manufacturing of Emivirine Intermediates Using Novel Meldrum’s Acid Chemistry
The pharmaceutical industry continuously seeks robust synthetic pathways for antiretroviral agents, particularly non-nucleoside reverse transcriptase inhibitors (NNRTIs) like Emivirine. Patent CN102206187B discloses a groundbreaking synthetic methodology for 6-benzyl-1-ethoxymethyl-5-isopropyluracil and its analogues, addressing critical bottlenecks in existing manufacturing protocols. This innovation utilizes cycloisopropyl malonate, commonly known as Meldrum’s Acid, as a versatile starting material to construct the uracil core with high efficiency. By shifting away from sensitive organometallic reagents, this technology offers a streamlined approach that enhances both chemical yield and process safety. For R&D teams and procurement specialists, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently. The following analysis dissects the technical merits and commercial implications of this patented route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Emivirine has relied heavily on complex organometallic transformations that pose significant challenges for industrial scale-up. Traditional routes often employ Grignard reagents, such as isopropyl magnesium chloride, which necessitate strictly anhydrous and oxygen-free environments to prevent decomposition. As illustrated in prior art schemes, these reactions frequently require cryogenic conditions, often dropping temperatures to -30°C or lower, which drastically increases energy consumption and operational complexity. Furthermore, alternative pathways involving Reformatsky reactions demand large excesses of zinc powder, creating substantial solid waste disposal issues and complicating the purification process. The reliance on transition metal catalysts, including copper and palladium salts in cross-coupling steps, introduces the risk of heavy metal residues in the final API, necessitating expensive and time-consuming removal steps to meet regulatory standards.
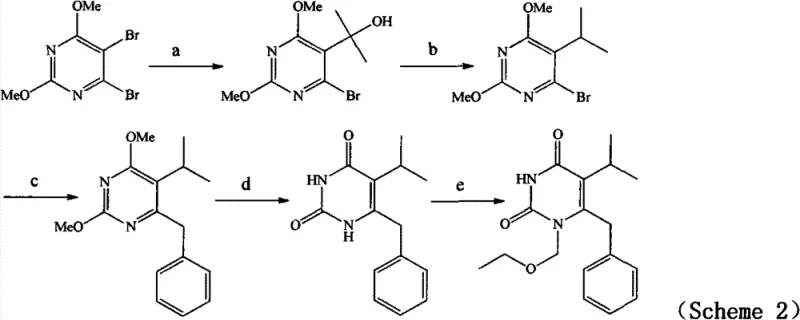
The Novel Approach
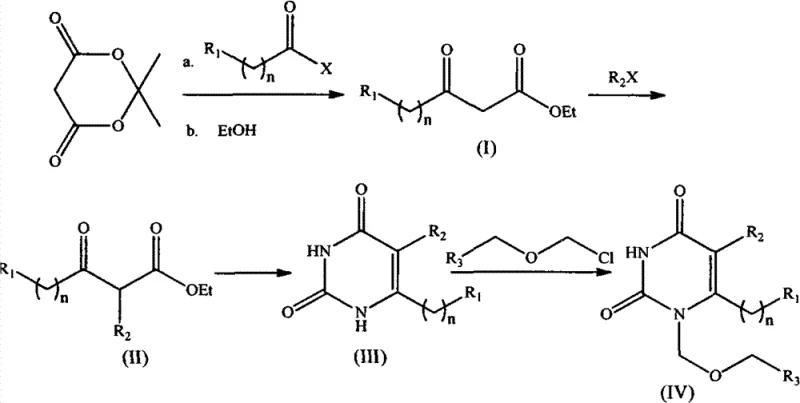
In stark contrast, the methodology described in CN102206187B leverages the unique reactivity of Meldrum’s Acid to bypass these hazardous steps entirely. This new strategy initiates with a mild nucleophilic substitution between Meldrum’s Acid and an acid chloride, proceeding smoothly at temperatures ranging from -10°C to 30°C. The subsequent alcoholysis step converts the intermediate directly into a beta-keto ester without requiring isolation, thereby telescoping the process and minimizing material loss. By avoiding pyrophoric reagents and cryogenic cooling, this approach significantly lowers the barrier to entry for manufacturing facilities. The route allows for facile modification at three distinct positions (R1, R2, and R3), providing medicinal chemists with a powerful platform for generating diverse analogue libraries. This flexibility is crucial for optimizing structure-activity relationships while maintaining a cost-effective production baseline.
Mechanistic Insights into Meldrum's Acid Mediated Cyclization
The core of this synthetic innovation lies in the efficient construction of the pyrimidine ring through a thiourea-mediated cyclization mechanism. After the initial formation of the substituted beta-keto ester (Compound II), the reaction with thiourea in the presence of sodium ethoxide triggers a condensation reaction. This step effectively closes the heterocyclic ring, forming the uracil scaffold with high regioselectivity. The mechanism proceeds through the nucleophilic attack of the sulfur atom on the carbonyl carbon, followed by elimination of ethanol and subsequent hydrolysis of the thio-intermediate. This specific pathway ensures that the substituents at the 5 and 6 positions are installed with precision, minimizing the formation of regioisomers that often plague traditional uracil syntheses. The use of chloroacetic acid in the hydrolysis step further drives the equilibrium towards the desired product, ensuring high conversion rates without the need for exotic catalysts.
Impurity control is inherently superior in this route due to the absence of radical side reactions common in organometallic chemistry. In Grignard-based syntheses, Wurtz-type coupling and protonation side reactions can generate difficult-to-remove byproducts. Here, the reaction intermediates are stable and well-defined, allowing for straightforward monitoring via standard analytical techniques like HPLC or TLC. The final N-1 alkylation step utilizes silylating agents like BSA (N,O-bistrimethylsilylacetamide) to activate the uracil nitrogen, facilitating nucleophilic attack by the chloromethyl ether. This mild activation strategy prevents degradation of the sensitive benzyl and isopropyl groups, resulting in a final product with exceptional purity profiles. Such control over the impurity spectrum is paramount for meeting the stringent quality specifications required by global regulatory bodies for HIV therapeutics.
How to Synthesize 6-benzyl-1-ethoxymethyl-5-isopropyluracil Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and solvent selection, although the conditions are notably forgiving compared to legacy methods. The process begins with the acylation of Meldrum's Acid, followed by in situ ethanolysis to generate the key beta-keto ester intermediate. Subsequent alkylation introduces the necessary side chains, setting the stage for ring closure. The cyclization and final N-alkylation steps are performed under reflux conditions, which are easily achievable in standard glass-lined reactors. Detailed standard operating procedures regarding specific molar ratios, addition rates, and workup protocols are essential for reproducibility. For a comprehensive guide on executing these transformations with maximum yield and safety, please refer to the standardized synthesis steps outlined below.
- React Meldrum's Acid with acid chloride in organic base to form ethyl 3-oxo-4-substituted butyrate (Compound I).
- Perform alkylation of Compound I with halides in aprotic solvent to generate Compound II.
- Cyclize Compound II with thiourea followed by hydrolysis to yield the uracil derivative (Compound III).
- Execute N-1 substitution using alkyl chloromethyl ether and silylating agents to obtain the final product (Compound IV).
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this Meldrum's Acid-based route offers profound strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. Meldrum's Acid and simple acid chlorides are commodity chemicals available from multiple global vendors, mitigating the risk of supply disruption associated with specialized organometallic reagents. This diversification of the supply base enhances negotiation leverage and stabilizes pricing structures. Moreover, the elimination of cryogenic requirements means that production can be scheduled more flexibly without competing for limited low-temperature reactor capacity. This operational agility translates directly into improved lead times and the ability to respond rapidly to fluctuating market demands for antiretroviral intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and hazardous reagents. By eliminating the need for transition metal catalysts like palladium or copper, the cost of goods sold is significantly reduced, as there is no requirement for costly metal scavenging resins or complex filtration systems. Furthermore, the ambient temperature conditions reduce energy consumption associated with heating and cooling cycles. The high yields reported in the patent examples, such as the 98.8% yield in the initial acylation step, indicate minimal raw material waste, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the chemical transformations. Since the reactions do not rely on moisture-sensitive reagents, the risk of batch failure due to minor environmental fluctuations is minimized. This stability ensures consistent output quality and quantity, which is critical for maintaining continuous API production lines. The simplicity of the workup procedures, often involving basic extraction and crystallization, reduces the turnaround time between batches. Consequently, manufacturers can maintain higher inventory levels of critical intermediates, buffering the supply chain against unforeseen logistical delays and ensuring uninterrupted delivery to downstream partners.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the lack of exothermic hazards typical of Grignard additions. The reaction profile is smooth and controllable, allowing for safe operation in large-volume reactors. Environmentally, the process generates less hazardous waste; the absence of heavy metals and excessive zinc sludge simplifies effluent treatment and reduces the environmental footprint. This alignment with green chemistry principles not only lowers waste disposal costs but also supports corporate sustainability goals, making the supply chain more resilient to evolving environmental regulations and enhancing the brand reputation of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The answers reflect the practical realities of scaling this chemistry while maintaining compliance with international quality standards.
Q: What are the primary advantages of the Meldrum's Acid route over Grignard methods?
A: The Meldrum's Acid route eliminates the need for harsh anhydrous and oxygen-free conditions required by Grignard reagents, significantly improving operational safety and reducing equipment costs associated with cryogenic cooling.
Q: Does this synthesis method avoid heavy metal contamination?
A: Yes, unlike prior art methods utilizing copper or palladium catalysts, this novel pathway relies on organic bases and simple inorganic salts, ensuring a cleaner impurity profile and easier downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The use of commercially available raw materials, ambient temperature reactions, and standard workup procedures makes this method highly scalable for industrial manufacturing without specialized infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-benzyl-1-ethoxymethyl-5-isopropyluracil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antiretroviral medications. Our technical team has extensively evaluated the Meldrum's Acid pathway described in CN102206187B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Emivirine intermediate meets the highest industry standards. We are committed to leveraging this advanced chemistry to deliver superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain for HIV therapeutics. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of this critical pharmaceutical intermediate.
