Scalable Modular Synthesis of (-)-Eburnamonine Intermediates for Commercial API Production
Introduction to Advanced Vasodilator Intermediate Synthesis
The pharmaceutical landscape for cerebrovascular treatments continues to demand high-purity intermediates for potent alkaloids like (-)-eburnamonine, also known as vinburnine. As detailed in the recent patent CN113402506A, a breakthrough modular synthesis strategy has been developed to address the historical challenges associated with producing this complex pentacyclic skeleton. This alkaloid, derived from Catharanthus roseus, exhibits remarkable vasodilation effects and is clinically utilized for treating ischemic stroke and enhancing cerebral blood flow. The structural complexity, featuring fused indole, piperidine, and lactam units alongside continuous chiral centers, has traditionally made its total synthesis a formidable task for organic chemists. However, this new methodology leverages distinct synthetic building blocks to achieve high efficiency and operational simplicity, marking a significant advancement for reliable pharmaceutical intermediate suppliers aiming to secure the supply chain for neuroprotective agents.
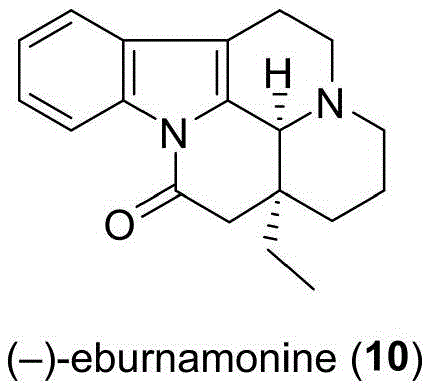
The significance of this technological leap cannot be overstated for the global supply of cerebrovascular medications. Traditional methods often struggled with low overall yields and cumbersome purification steps, which directly impacted the cost and availability of the final Active Pharmaceutical Ingredient (API). By introducing a route that allows for the simple conversion of advanced intermediates into the final target, the patent outlines a pathway that is not only scientifically elegant but also commercially viable. This report delves deep into the mechanistic nuances and commercial implications of this novel process, providing critical insights for R&D directors and procurement managers looking to optimize their manufacturing portfolios for high-value neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of vinburnine has been plagued by inefficiencies that hinder deep pharmacological research and commercial development. Early approaches, such as those described by Bartlett and Taylor or Grieco and Kaufman, often involved lengthy linear sequences with poor atom economy. These conventional routes frequently relied on racemic synthesis followed by difficult resolution steps, resulting in substantial material loss and increased waste generation. Furthermore, the construction of the quaternary carbon center at C20 and the fusion of the D-ring often required harsh reaction conditions or specialized catalysts that were difficult to source in bulk quantities. For a procurement manager, these factors translate into volatile pricing and unreliable lead times, as any bottleneck in a multi-step linear synthesis can halt the entire production line. The low college economy of these older methods simply does not align with the rigorous cost-control measures required in modern generic drug manufacturing.
The Novel Approach
In stark contrast, the novel approach disclosed in patent CN113402506A adopts a convergent modular synthesis strategy that fundamentally reshapes the production logic. Instead of building the molecule atom-by-atom from simple precursors, this method utilizes two advanced synthetic blocks: a compound possessing the D-ring structure and C20 quaternary carbon center (Compound 1), and a tryptophol derivative containing the indole ring (Compound 5). This convergence allows for the rapid assembly of the core skeleton through a simple condensation reaction, drastically reducing the number of steps required to reach the final target. The operational simplicity is further enhanced by the use of robust protecting group strategies, such as the acetal protection of the aldehyde moiety, which ensures stability during subsequent transformations. For stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing, this modularity means that different parts of the molecule can be synthesized in parallel, optimizing facility utilization and significantly shortening the overall campaign time.
Mechanistic Insights into Modular Assembly and Cyclization
The chemical elegance of this route is best understood through the lens of its key transformation steps, particularly the coupling and cyclization sequences. The process initiates with the preparation of the lactam fragment, where Compound 1 undergoes ozonolysis to cleave the terminal double bond, generating an aldehyde that is immediately protected as an acetal to form Compound 3. Following the removal of the benzoyl group, the resulting intermediate 4 serves as a nucleophile in a critical condensation reaction with the indole-based electrophile, Compound 5. This step, facilitated by strong bases like sodium hydride in tetrahydrofuran at elevated temperatures (60°C), forms the carbon-nitrogen bond that links the two major domains of the molecule. The success of this coupling is vital, as it establishes the connectivity required for the subsequent ring-closing events that define the eburnamonine architecture.
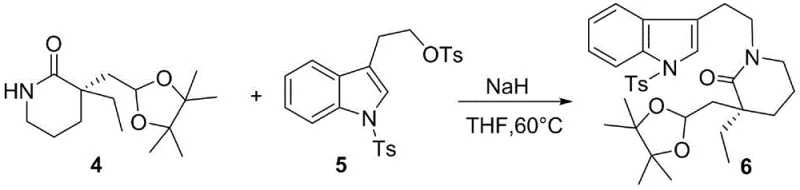
Following the coupling, the synthesis proceeds through a deprotection of the tosyl group using magnesium powder, yielding Compound 7, which is primed for the pivotal ring-closing reaction. The core of the mechanistic novelty lies in the Bischler-Napieralski reaction applied to Compound 7. Using trifluoromethanesulfonic anhydride and 2-fluoropyridine, the amide carbonyl is activated to form an iminium ion intermediate (Compound 8). This highly reactive species then undergoes an intramolecular electrophilic aromatic substitution to close the ring, forming the tetracyclic core. The subsequent stereoselective reduction of this iminium salt using lithium tri-tert-butoxyaluminum hydride at cryogenic temperatures (-78°C) is the masterstroke of the synthesis. It selectively delivers the hydride to the less hindered face of the iminium ion, establishing the crucial stereochemistry at the ring junction with a diastereomeric ratio of 5:1 favoring the desired product (Compound 9). This level of control eliminates the need for downstream chiral separations, a massive advantage for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize (-)-Eburnamonine Efficiently
The synthesis of (-)-eburnamonine via this patented route represents a paradigm shift from linear to modular construction, offering a streamlined path for process chemists. The protocol begins with the functionalization of the lactam precursor through ozonolysis and protection, followed by a base-mediated coupling with the indole fragment. The subsequent steps involve precise deprotection and a cascade of cyclization and reduction reactions to forge the rigid pentacyclic framework. Finally, the removal of the acetal protecting group triggers an intramolecular cyclization to form the alcohol, which is then oxidized to the final ketone functionality of the target molecule. For detailed standard operating procedures and specific stoichiometric ratios required to replicate this high-yielding process, please refer to the technical guide below.
- Perform ozonolysis on compound 1 followed by aldehyde protection to generate the key lactam intermediate 4.
- Condense intermediate 4 with tryptophol derivative 5 using sodium hydride to form the coupled product 6.
- Execute Bischler-Napieralski cyclization on compound 7 to form the iminium salt 8, followed by stereoselective reduction to compound 9.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this modular synthesis route offers profound benefits that extend beyond mere chemical yield. The strategic use of readily available starting materials and common reagents mitigates the risk of raw material shortages that often plague specialty chemical manufacturing. By avoiding the reliance on exotic transition metal catalysts or proprietary chiral ligands, the process ensures a more resilient supply chain that is less susceptible to geopolitical fluctuations in the mining of rare earth elements. Furthermore, the operational conditions, which largely operate between ambient and moderately elevated temperatures, reduce the energy burden on production facilities. This translates directly into a lower carbon footprint and reduced utility costs, aligning with the sustainability goals of modern multinational corporations seeking reliable pharmaceutical intermediate suppliers with green credentials.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive resolution steps and the high efficiency of the convergent strategy. By assembling the molecule from two advanced fragments, the overall step count is minimized, which inherently reduces the accumulation of yield losses typical in long linear syntheses. Additionally, the use of cost-effective reagents such as magnesium powder for deprotection and lithium hydroxide for saponification replaces more expensive alternatives, driving down the Bill of Materials (BOM) cost significantly. The high diastereoselectivity achieved during the reduction step further enhances cost efficiency by minimizing the formation of hard-to-separate impurities, thereby reducing the load on purification resources and solvent consumption.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable manufacturing timeline. Unlike sensitive organometallic reactions that require strict exclusion of moisture and oxygen, many steps in this sequence, such as the acetal formation and hydrolysis, are tolerant to minor variations in processing conditions. This tolerance reduces the likelihood of batch failures and reworks, ensuring a consistent flow of material to downstream API manufacturers. Moreover, the modular nature of the synthesis allows for the stocking of key intermediates (like Compound 4 and Compound 5), enabling a "just-in-time" assembly approach that can respond rapidly to spikes in market demand for vasodilator therapies without the need for long lead-time raw material procurement.
- Scalability and Environmental Compliance: Scaling this process from the laboratory to the pilot plant is facilitated by the use of standard unit operations such as filtration, extraction, and distillation. The avoidance of hazardous reagents like osmium tetroxide or large quantities of heavy metals simplifies the waste treatment protocols, making it easier to meet stringent environmental regulations in major manufacturing hubs. The final oxidation step utilizes AZADO, a stable nitroxyl radical catalyst, which offers a safer and more environmentally benign alternative to chromium-based oxidants. This commitment to safer chemistry not only protects the workforce but also streamlines the regulatory approval process for the manufacturing site, ensuring uninterrupted reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for technical teams evaluating the integration of this intermediate into their existing production pipelines.
Q: What are the key advantages of this modular synthesis route for (-)-eburnamonine?
A: The primary advantage lies in the high synthesis efficiency and operational simplicity. By utilizing pre-formed building blocks with established chirality (Compound 1 and Compound 5), the route avoids complex asymmetric inductions late in the synthesis, significantly streamlining the process for large-scale manufacturing.
Q: How is the stereochemistry controlled during the cyclization step?
A: Stereocontrol is achieved during the reduction of the iminium intermediate (Compound 8). The use of lithium tri-tert-butoxyaluminum hydride at low temperatures (-78°C) favors the formation of the desired diastereomer (Compound 9) over the isomer (Compound 12) with a ratio of approximately 5:1, ensuring high optical purity in the final product.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly highlights the ease of scale-up. The reaction conditions utilize common solvents like dichloromethane and tetrahydrofuran, and reagents such as magnesium powder and lithium hydroxide are cost-effective and readily available, making the transition from laboratory to commercial production feasible without exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Eburnamonine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cerebrovascular medications. Our team of expert process chemists has extensively analyzed the route disclosed in CN113402506A and possesses the technical capability to execute this complex modular synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of (-)-eburnamonine intermediate meets the highest international standards for pharmaceutical application.
We invite you to collaborate with us to leverage this innovative synthesis technology for your next project. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a competitive advantage in the vasodilator market through superior supply chain solutions and advanced chemical manufacturing expertise.
