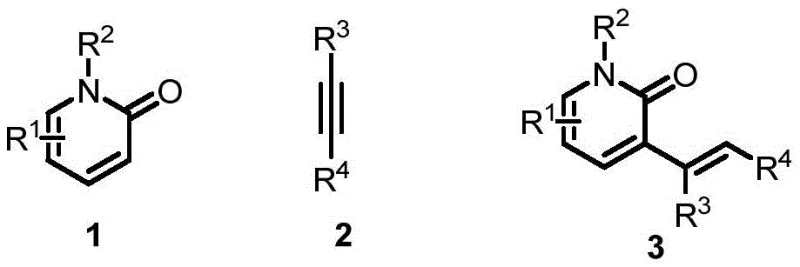
Revolutionizing Pharmaceutical Intermediate Production: Advanced Nickel-Catalyzed Synthesis of C3-Alkenylated 2-Pyridone Derivatives
Patent CN113024604B introduces a groundbreaking methodology for synthesizing C3-alkenylated 2-pyridone derivatives, representing a significant advancement in heterocyclic compound manufacturing for pharmaceutical applications. This innovative approach addresses longstanding challenges in the production of these biologically active molecules, which serve as critical building blocks in numerous therapeutic agents including anticancer drugs like camptothecin and cardiovascular medications such as milrinone. The patent demonstrates a novel catalytic system that achieves unprecedented site selectivity while maintaining commercial viability through simplified processing steps and reduced environmental impact. By eliminating the need for precious metal catalysts and complex multi-step procedures, this technology offers pharmaceutical manufacturers a more sustainable pathway to produce high-value intermediates with enhanced purity profiles and consistent quality metrics essential for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for C3-alkenylated 2-pyridone derivatives have been severely constrained by multiple technical and economic limitations that hinder commercial scalability. Existing methodologies predominantly rely on palladium-based catalytic systems requiring expensive precious metal catalysts and stoichiometric amounts of silver or copper oxidants, significantly increasing raw material costs while generating substantial metal-containing waste streams that require complex purification protocols. These conventional approaches suffer from poor site selectivity, frequently producing multiple regioisomers that necessitate costly separation processes, and are often incompatible with simple pyridone substrates, limiting their applicability across diverse molecular architectures. Furthermore, many established methods employ air- and moisture-sensitive reagents that demand specialized handling equipment and stringent operational conditions, creating significant barriers to large-scale manufacturing while increasing both capital expenditure and operational complexity for pharmaceutical producers seeking reliable supply chains for these critical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an innovative nickel-catalyzed system that operates under mild conditions with commercially available reagents, delivering exceptional site selectivity without generating unwanted by-products. This breakthrough process utilizes cost-effective nickel catalysts instead of precious palladium, eliminating expensive metal requirements while maintaining high reaction efficiency across diverse substrate classes including simple pyridones previously considered unreactive in conventional systems. The strategic combination of specialized phosphine ligands with Lewis acid promoters enables precise C3-site alkenylation with remarkable regiocontrol, avoiding multi-site reaction issues that plagued previous approaches and significantly reducing downstream purification costs. Operating under standard inert atmosphere conditions with readily accessible solvents, this method simplifies manufacturing requirements while achieving atom-economical transformations that align with modern green chemistry principles, making it ideally suited for commercial-scale production of pharmaceutical intermediates with stringent quality specifications.
Mechanistic Insights into Nickel-Catalyzed C3-Alkenylation
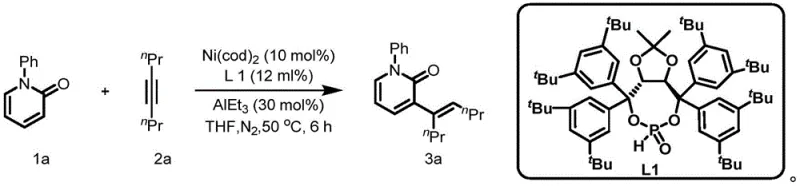
The catalytic cycle begins with the formation of an active nickel(0) species from Ni(cod)₂ in the presence of the phosphine ligand L1, which coordinates to create a highly reactive yet selective catalytic center. This nickel complex then undergoes oxidative addition with the alkyne substrate, followed by coordination with the 2-pyridone derivative through its nitrogen atom, positioning the molecule for selective C-H activation at the C3 position. The Lewis acid component (typically triethylaluminum) plays a crucial role by activating the pyridone carbonyl group, facilitating deprotonation and enabling the key migratory insertion step that forms the new carbon-carbon bond with precise regiocontrol at the C3 position. This synergistic interaction between nickel catalyst, specialized ligand architecture, and Lewis acid promoter creates a highly organized transition state that directs alkenylation exclusively to the C3 position while suppressing alternative reaction pathways that would lead to undesired regioisomers or by-products.
The exceptional site selectivity achieved in this process stems from the unique electronic and steric properties of the TIDOL-based ligand system (L1), which creates a well-defined chiral pocket around the nickel center that precisely controls substrate orientation during the catalytic cycle. This ligand design prevents undesired coordination modes that could lead to alternative reaction pathways while promoting optimal orbital alignment for selective C-H activation at the desired position. The Lewis acid component further enhances selectivity by modulating the electronic properties of the pyridone substrate, lowering the energy barrier for deprotonation at the desired position while simultaneously stabilizing key intermediates in the catalytic cycle. This sophisticated interplay between catalyst components results in a highly efficient transformation that produces single-site alkenylated products with minimal impurities, addressing critical quality concerns for pharmaceutical applications where strict purity specifications are mandatory for regulatory compliance.
How to Synthesize C3-Alkenylated 2-Pyridone Derivatives Efficiently
This innovative synthesis methodology represents a significant advancement in heterocyclic compound manufacturing, offering pharmaceutical developers a streamlined pathway to access structurally diverse C3-alkenylated 2-pyridone derivatives with exceptional purity profiles. The patented process eliminates multiple processing steps required by conventional methods while maintaining excellent yield and selectivity across a broad substrate scope, making it particularly valuable for producing complex intermediates needed in drug discovery and development programs. Detailed standardized synthesis procedures are provided below to facilitate seamless implementation in industrial manufacturing environments.
- Prepare the reaction system by adding the phosphine ligand (L1), nickel catalyst (Ni(cod)₂), and solvent (THF or toluene) to the reaction flask under nitrogen atmosphere.
- Introduce the 2-pyridone substrate (compound 1) and alkyne (compound 2) to the reaction mixture, maintaining the molar ratio between 1: 1 to 1:2.
- Add the Lewis acid (triethylaluminum) and heat the reaction to the specified temperature (50°C to 150°C) for 6 to 10 hours under inert conditions.
Commercial Advantages for Procurement and Supply Chain Teams
This patented manufacturing process delivers substantial value to procurement and supply chain operations by addressing critical pain points associated with traditional production methods for pharmaceutical intermediates. The elimination of precious metal catalysts and complex multi-step procedures significantly reduces raw material costs while improving process reliability and scalability, creating opportunities for more stable supply arrangements with predictable lead times that support just-in-time manufacturing strategies essential for modern pharmaceutical production networks.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with cost-effective nickel-based systems eliminates significant raw material expenses while removing the need for costly metal removal processes during purification. The atom-economical nature of this transformation reduces waste disposal costs and eliminates expenses associated with handling hazardous by-products common in conventional methods, creating substantial cost savings throughout the manufacturing value chain without compromising product quality or purity specifications required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilizing commercially available reagents with stable shelf lives under normal storage conditions ensures consistent raw material availability while eliminating supply chain vulnerabilities associated with specialized or sensitive components required by traditional methods. The simplified one-step process reduces production cycle times and minimizes potential failure points in manufacturing, enabling more predictable delivery schedules that support reliable inventory management practices essential for maintaining continuous production flows in pharmaceutical manufacturing operations.
- Scalability and Environmental Compliance: The robust reaction conditions that operate effectively from laboratory scale to commercial production volumes ensure seamless technology transfer without requiring significant process re-engineering, while the absence of hazardous waste streams simplifies environmental compliance management. This green chemistry approach aligns with increasingly stringent regulatory requirements for sustainable manufacturing practices while reducing both capital investment needs for specialized waste treatment equipment and ongoing operational costs associated with environmental compliance monitoring.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this innovative synthesis methodology in pharmaceutical manufacturing operations. These responses are based on detailed analysis of the patented technology's capabilities and performance characteristics as demonstrated through extensive experimental validation.
Q: How does the nickel-catalyzed method eliminate the need for expensive palladium catalysts in 2-pyridone alkenylation?
A: The patented process utilizes cost-effective nickel catalysts instead of precious palladium, eliminating the need for expensive transition metals while maintaining high reaction efficiency and selectivity.
Q: What makes this synthesis method atom-economical and environmentally friendly compared to conventional approaches?
A: This method achieves complete atom economy with no by-products generated during large-scale production, significantly reducing waste streams and aligning with green chemistry principles.
Q: How does the process achieve high site selectivity for C3-alkenylation of 2-pyridone derivatives?
A: The synergistic effect of the nickel catalyst, specialized phosphine ligand, and Lewis acid enables precise C3-site selectivity, eliminating multi-site reaction issues common in traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C3-Alkenylated 2-Pyridone Derivatives Supplier
Our patented nickel-catalyzed synthesis platform represents a transformative approach to producing high-value pharmaceutical intermediates with exceptional purity profiles and consistent quality metrics essential for drug development pipelines. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from laboratory validation to full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities for comprehensive quality assurance.
For companies seeking to optimize their supply chain for critical pharmaceutical intermediates, we invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your requirements. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative manufacturing approach can enhance your production economics while ensuring reliable supply of high-purity C3-alkenylated 2-pyridone derivatives for your drug development programs.
