Scalable Synthesis of Polysubstituted N-Arylpyrrole Compounds for Pharmaceutical Applications
Scalable Synthesis of Polysubstituted N-Arylpyrrole Compounds for Pharmaceutical Applications
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient and cost-effective routes to bioactive scaffolds. Patent CN111423353B introduces a groundbreaking methodology for the preparation of polysubstituted N-arylpyrrole compounds, addressing critical bottlenecks in current synthetic strategies. Pyrrole derivatives constitute the core skeleton of numerous natural products and pharmaceutical agents, ranging from antibiotics to anticancer drugs, making their accessible synthesis a priority for the global fine chemical industry. This patent details a robust three-component coupling reaction that leverages inexpensive Lewis acid catalysts to achieve high selectivity and yield under mild conditions. By eliminating the need for precious metal catalysts and harsh reaction environments, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chains for high-purity pharmaceutical intermediates.
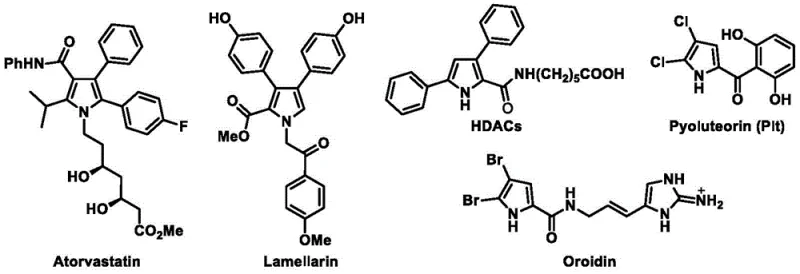
The biological significance of the pyrrole ring cannot be overstated, as evidenced by its presence in blockbuster drugs like Atorvastatin and complex natural products like Lamellarin. The ability to construct this heterocyclic core with diverse substitution patterns is essential for medicinal chemists exploring structure-activity relationships. The disclosed invention provides a generalized formula I that encompasses a vast chemical space, allowing for the incorporation of various aryl, heteroaryl, and alkyl groups at the N-1, C-2, and C-3 positions. This structural flexibility is paramount for developing next-generation therapeutics, particularly in the fields of antiviral and antibacterial research where polysubstituted pyrroles have shown immense promise. Consequently, access to a reliable supplier capable of delivering these complex intermediates with consistent quality is a strategic asset for any R&D organization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrroles has relied on classical name reactions such as the Knorr pyrrole synthesis, Hantzsch pyrrole synthesis, and Paal-Knorr synthesis. While these methods established the foundation of heterocyclic chemistry, they suffer from significant drawbacks when applied to modern industrial settings. Many of these traditional routes require pre-functionalized substrates, which increases the number of synthetic steps and overall material costs. Furthermore, they often necessitate the use of strong acids or bases, high temperatures, and high pressures, leading to safety concerns and difficult waste management. The reliance on transition metal catalysis in some modern variations introduces another layer of complexity, as removing trace heavy metals to meet stringent pharmaceutical purity specifications can be both technically challenging and economically burdensome. These limitations collectively hinder the rapid scale-up and cost reduction in API manufacturing that the industry desperately needs.
The Novel Approach
In stark contrast to conventional methodologies, the process described in CN111423353B utilizes a direct three-component tandem reaction involving aromatic amines, bromoacetaldehyde diethyl acetal, and 1,3-dicarbonyl compounds. This approach bypasses the need for substrate pre-functionalization, streamlining the synthetic pathway and reducing the overall step count. The reaction is catalyzed by abundant and inexpensive Lewis acids such as ferric trichloride, aluminum chloride, or cupric bromide, completely avoiding the use of costly noble metals like palladium or rhodium. Operating at moderate temperatures between 60°C and 80°C, the process ensures energy efficiency and minimizes thermal degradation of sensitive functional groups. This novel strategy not only simplifies the operational workflow but also significantly enhances the economic viability of producing high-purity OLED material precursors and pharmaceutical intermediates on a commercial scale.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovation lies in the efficient activation of reactants by the Lewis acid catalyst, which facilitates the formation of the pyrrole ring through a concerted mechanism. The aromatic amine first reacts with the bromoacetaldehyde diethyl acetal to form an intermediate imine or enamine species, which is subsequently attacked by the nucleophilic 1,3-dicarbonyl compound. The catalyst plays a crucial role in activating the carbonyl groups and stabilizing the transition states, thereby lowering the activation energy required for cyclization. This mechanistic pathway allows for excellent regioselectivity, ensuring that the substituents are installed at the desired positions on the pyrrole ring without the formation of unwanted isomers. The tolerance of the catalytic system towards various functional groups, including halogens, esters, and trifluoromethyl groups, underscores its versatility and robustness in complex molecule synthesis.

Impurity control is a critical aspect of this synthesis, particularly for applications in the pharmaceutical sector where regulatory standards are rigorous. The mild reaction conditions and the specific nature of the Lewis acid catalyst minimize side reactions such as polymerization or over-alkylation, which are common pitfalls in pyrrole chemistry. The use of stoichiometric ratios ranging from 1:1 to 1:2 for the reactants allows for precise control over the reaction kinetics, further suppressing the formation of by-products. Post-reaction purification is straightforward, typically involving silica gel column chromatography with standard eluent systems like petroleum ether and ethyl acetate. This simplicity in work-up procedures contributes to a cleaner impurity profile and higher isolated yields, often exceeding 80% for optimized substrates, thereby ensuring the delivery of high-purity pharmaceutical intermediates that meet the demanding specifications of global health authorities.
How to Synthesize Polysubstituted N-Arylpyrrole Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-plant production. The procedure involves dissolving the starting materials in a common organic solvent, adding the catalyst, and heating the mixture with magnetic stirring. The flexibility in solvent choice, including toluene, ethanol, and acetonitrile, allows process chemists to select the most appropriate medium based on solubility and environmental considerations. Detailed standardized synthetic steps see the guide below, which outlines the specific molar ratios and reaction times required to achieve optimal results for various derivatives.
- Dissolve aromatic amine, bromoacetaldehyde diethyl acetal, and 1,3-dicarbonyl compound in an organic solvent such as toluene or ethanol.
- Add a Lewis acid catalyst like ferric trichloride or aluminum chloride to the reaction mixture under magnetic stirring.
- Heat the reaction mixture to 60-80°C for 4-10 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift away from noble metal catalysts to earth-abundant Lewis acids represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for expensive catalysts and the associated metal scavenging processes, the overall cost of goods sold (COGS) can be drastically reduced. Furthermore, the use of commodity chemicals as raw materials ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized reagents. This stability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines in a volatile global market.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive alternatives like ferric trichloride or aluminum chloride removes a major cost driver from the production budget. Additionally, the mild reaction temperatures reduce energy consumption, while the high atom economy of the three-component reaction minimizes waste generation. These factors combine to create a highly economical process that offers substantial cost savings compared to traditional multi-step syntheses, allowing for more competitive pricing in the final API market.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including aromatic amines and 1,3-dicarbonyl compounds, are widely available from multiple global suppliers. This abundance reduces dependency on single-source vendors and shortens lead times for high-purity pharmaceutical intermediates. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by minor variations in raw material quality or environmental fluctuations, ensuring a consistent and reliable flow of materials to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with simple equipment and standard solvents. The absence of toxic heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. This eco-friendly profile not only reduces compliance costs but also enhances the corporate sustainability image of the manufacturing entity, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these polysubstituted N-arylpyrrole compounds. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing accurate and actionable insights for decision-makers.
Q: What are the advantages of this new pyrrole synthesis method over classical Knorr or Paal-Knorr reactions?
A: Unlike classical methods that often require harsh conditions, pre-functionalized substrates, or expensive noble metal catalysts, this novel approach utilizes cheap and readily available Lewis acid catalysts (e.g., FeCl3, AlCl3) and operates under mild temperatures (60-80°C), significantly reducing operational costs and safety risks.
Q: Can this synthesis route be scaled for commercial production of API intermediates?
A: Yes, the process demonstrates excellent scalability due to the use of common industrial solvents like toluene and ethanol, along with robust reaction conditions that tolerate a wide range of substituents, making it highly suitable for the commercial scale-up of complex pharmaceutical intermediates.
Q: What downstream applications do these polysubstituted pyrroles support?
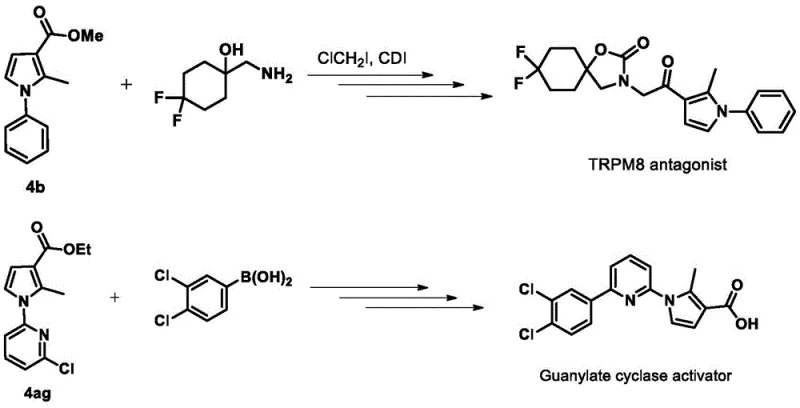
A: These compounds serve as versatile precursors for biologically active molecules, including TRPM8 antagonists and guanylate cyclase activators, offering active sites like ester and carboxyl groups for further structural optimization in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted N-Arylpyrrole Supplier
The technological advancements detailed in CN111423353B represent a significant leap forward in the field of heterocyclic synthesis, offering a pathway to valuable drug candidates that was previously hindered by cost and complexity. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these molecules to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of polysubstituted N-arylpyrrole delivered meets the highest international standards. We understand the critical nature of supply continuity in the pharmaceutical industry and are committed to being a partner you can trust for long-term success.
We invite you to explore the potential of this novel synthetic route for your specific drug development projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your unique requirements, demonstrating how this technology can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to accelerate your pipeline and bring life-saving therapies to patients faster and more efficiently.