Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of 3-Arylquinoline Ketone Derivatives
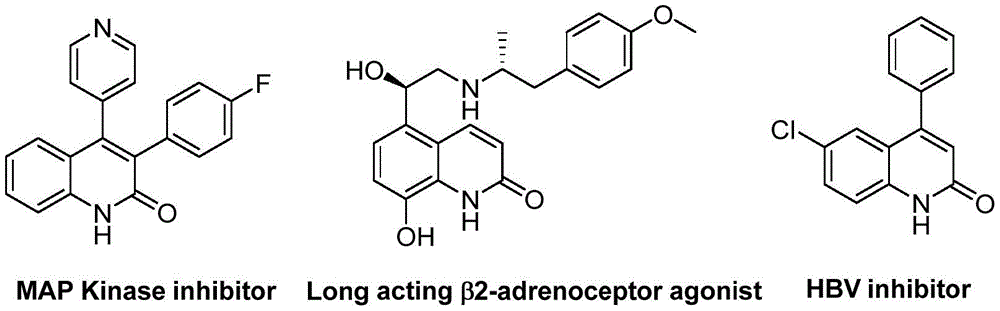
The recently granted Chinese patent CN113045489B introduces a novel synthetic pathway for 3-arylquinoline-2(1H) ketone derivatives, a critical class of pharmaceutical intermediates with demonstrated applications in kinase inhibition and antiviral therapeutics. This breakthrough methodology leverages benzisoxazole as a dual-function reagent serving simultaneously as nitrogen and formyl source, eliminating the need for separate functionalization steps while maintaining high substrate tolerance across diverse functional groups. The process operates under mild conditions (100°C, 26 h) using commercially available catalysts including Pd(OAc)2 (10 mol%), (S)-BINAP (10 mol%), and Mo(CO)6 (1.5 equiv.) in DME solvent with triethylamine and water co-catalysts. As illustrated in Figure 1, these molecular scaffolds form the structural basis for multiple drug candidates including MAP Kinase inhibitors and HBV therapeutics, underscoring their strategic importance in modern drug development pipelines.
Novel Reaction Mechanism and Impurity Control
The patented process represents a significant advancement in heterocyclic synthesis through its innovative use of benzisoxazole as a bifunctional building block. The reaction proceeds via palladium-catalyzed aminocarbonylation where benzisoxazole undergoes ring-opening to provide both the nitrogen atom for quinoline formation and the carbonyl group for ketone functionality, as depicted in Figure 5. This dual-role mechanism eliminates traditional multi-step sequences requiring separate nitrogen incorporation and carbonylation steps, thereby reducing potential side reactions that typically generate impurities such as regioisomers or over-reduced byproducts. The precise stoichiometric control (Pd catalyst:(S)-BINAP:Et3N:H2O = 0.1:0.1:6:1.0) ensures optimal catalyst activation while minimizing palladium black formation that could otherwise lead to metal contamination in the final product.
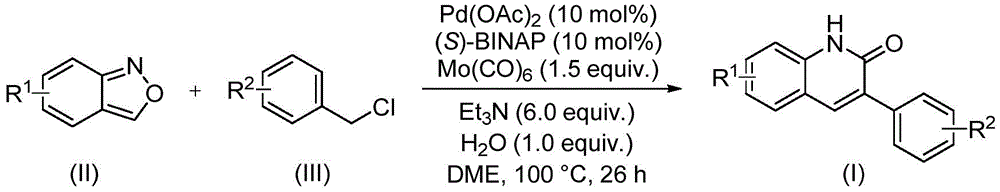
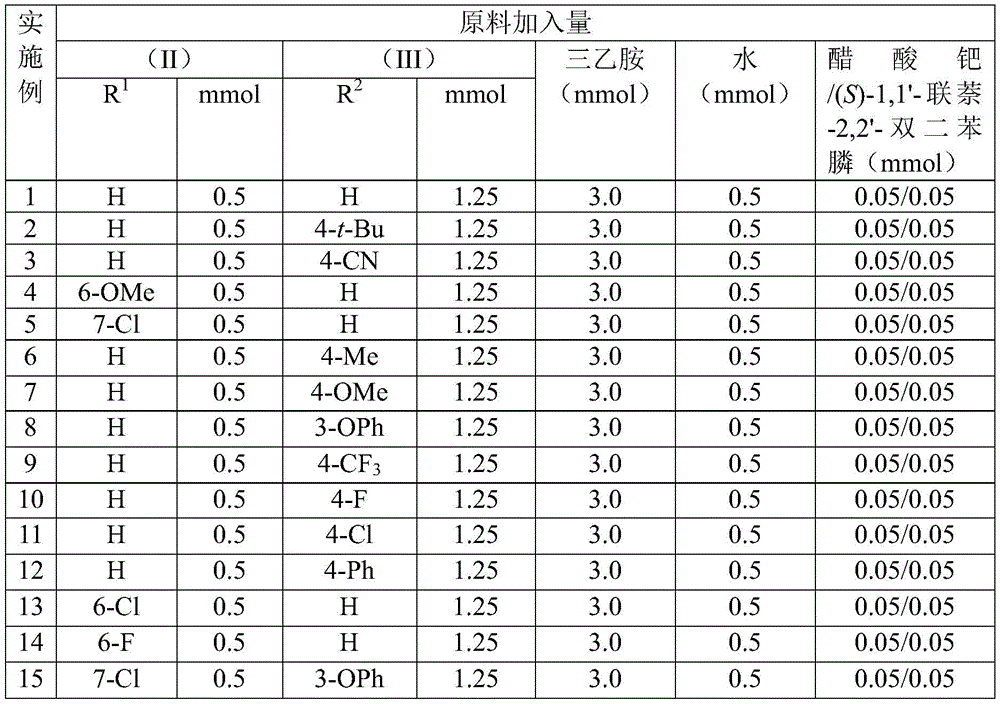
Impurity profile management is further enhanced by the broad functional group tolerance demonstrated across all fifteen experimental examples, where substituents including halogens (Cl, F), alkoxy groups (OMe), and electron-withdrawing moieties (CN, CF3) were successfully incorporated without significant side reactions. The post-processing protocol—comprising simple filtration, silica gel mixing, and standard column chromatography—avoids harsh purification conditions that might degrade sensitive functional groups or introduce new impurities. Crucially, the reaction's high efficiency (yields ranging from 68% to 95% as shown in Figure 10) directly correlates with reduced impurity formation since incomplete conversions typically generate unreacted starting materials that complicate purification. This inherent selectivity minimizes the need for additional purification steps that would otherwise increase both cost and processing time while maintaining stringent purity specifications required for pharmaceutical intermediates.
Commercial Advantages for Procurement and Supply Chain Management
This innovative synthesis addresses three critical pain points in pharmaceutical intermediate manufacturing: raw material complexity, process inefficiency, and scalability limitations inherent in conventional quinoline synthesis methods. By utilizing benzisoxazole as a dual-purpose reagent, the process eliminates multiple synthetic steps required in traditional Vilsmeier-Haack or Friedlander approaches, thereby reducing both capital equipment requirements and operational complexity. The methodology's compatibility with standard laboratory glassware and absence of specialized reactors further enhances its manufacturability across diverse production environments while maintaining consistent quality output.
- Reduced Raw Material Complexity: The strategic use of benzisoxazole as both nitrogen and formyl source significantly reduces the number of required starting materials compared to conventional multi-step syntheses. This consolidation eliminates procurement dependencies on specialized reagents while leveraging commercially abundant benzisoxazole derivatives that are readily available from multiple global suppliers. The simplified material flow reduces inventory management complexity and associated carrying costs, while the broad substrate tolerance (demonstrated across fifteen examples with varying R1/R2 groups) enables single-process production of multiple derivative variants without retooling. This flexibility substantially lowers changeover costs between product variants and enhances responsiveness to fluctuating market demands.
- Enhanced Process Efficiency: The one-pot reaction design operating at moderate temperature (100°C) with standard reaction times (26 h) significantly reduces energy consumption compared to high-pressure carbonylation methods or cryogenic processes often required in traditional syntheses. The elimination of transition metal removal steps—made possible by the optimized catalyst system—reduces solvent usage and waste generation while avoiding costly metal scavenging procedures. This streamlined approach minimizes equipment wear-and-tear from extreme process conditions, extending reactor lifespans and reducing maintenance downtime. The consistent high yields across diverse substrates further improve resource utilization by maximizing output per production run.
- Scalability and Supply Continuity: The process demonstrates exceptional scalability from laboratory to commercial production as evidenced by the consistent yields across different reaction scales documented in Figure 9. The use of standard solvents (DME), commercially available catalysts, and straightforward workup procedures ensures seamless technology transfer without requiring specialized infrastructure investments. This robustness provides reliable supply continuity even during market fluctuations, as the methodology tolerates minor variations in raw material quality without significant yield impact. The absence of hazardous intermediates or extreme process conditions further enhances operational safety margins during scale-up, reducing the risk of production interruptions that could compromise delivery schedules.

Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline Ketone Derivatives Supplier
While this palladium-catalyzed aminocarbonylation methodology demonstrates significant potential for pharmaceutical intermediate production, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure seamless technology transfer. Our rigorous QC labs implement multi-stage analytical protocols that consistently meet stringent purity specifications required for advanced intermediates, with dedicated teams specializing in complex heterocyclic synthesis optimization. As a CDMO partner with proven expertise in catalytic route development, we provide comprehensive technical support to navigate regulatory requirements while maintaining supply chain integrity throughout scale-up processes.
We invite you to initiate a Customized Cost-Saving Analysis for your specific derivative requirements by contacting our technical procurement team. Request detailed COA data and route feasibility assessments tailored to your production volume needs to evaluate how this patented methodology can enhance your supply chain resilience while delivering significant cost reduction in pharmaceutical intermediate manufacturing.
