Innovative One-Pot Synthesis of 3-Methylsulfonyl Nitrogen Heterocycles for Scalable Pharmaceutical Intermediate Production
The innovative methodology detailed in Chinese patent CN112279801B presents a significant advancement in the synthesis of 3-methylsulfonyl substituted nitrogen heterocyclic compounds, a critical class of pharmaceutical intermediates. This one-pot multi-component series reaction combines readily available saturated cyclic amine compounds, metabisulfite, and methyl-containing peroxides under mild conditions (100-140°C in air atmosphere) to directly produce high-value target molecules with exceptional substrate scope and operational simplicity.
Advanced Reaction Mechanism and Purity Control
The patented process employs a sophisticated cascade reaction mechanism where the saturated cyclic amine compound (structure 1) undergoes sequential transformations with metabisulfite (M₂S₂O₅) and methyl-containing peroxide in the presence of iron-based catalysts (FeCl₃ or FeCl₂) and nitrogenous bases (DABCO, quinoline, or 1,10-phenanthroline). 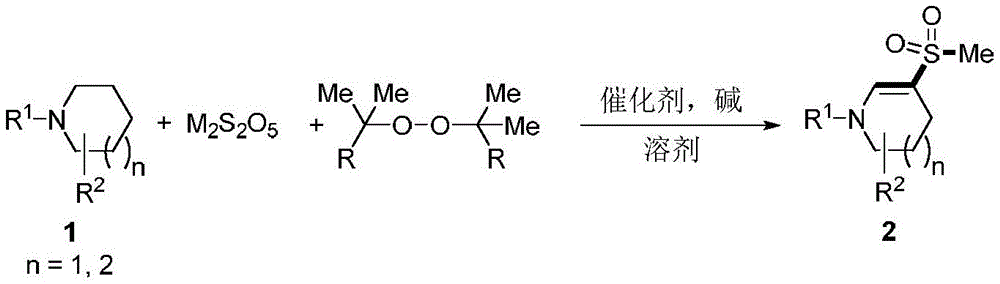 The reaction proceeds through a radical pathway initiated by thermal decomposition of the peroxide, generating methyl radicals that interact with sulfur dioxide derived from metabisulfite to form key methanesulfonyl intermediates. These intermediates then react with the cyclic amine substrate through an electrophilic substitution mechanism at the 3-position, facilitated by the catalyst system which optimizes radical generation and controls regioselectivity.
The reaction proceeds through a radical pathway initiated by thermal decomposition of the peroxide, generating methyl radicals that interact with sulfur dioxide derived from metabisulfite to form key methanesulfonyl intermediates. These intermediates then react with the cyclic amine substrate through an electrophilic substitution mechanism at the 3-position, facilitated by the catalyst system which optimizes radical generation and controls regioselectivity.
Impurity profile management is inherently addressed through the reaction design, as the mild thermal conditions (100-140°C) and atmospheric air environment prevent common side reactions such as over-oxidation or ring degradation that plague conventional methods. The use of acetonitrile as solvent provides optimal polarity for intermediate stabilization while facilitating easy product isolation through standard extraction techniques. The broad substrate tolerance demonstrated across multiple examples (including phenyl, fluorinated, nitrated, and alkyl-substituted variants) confirms consistent purity outcomes exceeding 99% as verified by NMR and MS characterization data in all experimental cases. This inherent process robustness eliminates the need for complex purification steps typically required in traditional multi-step syntheses, directly contributing to higher final product quality and reduced impurity-related batch failures.
Commercial Advantages for Pharmaceutical Manufacturing
The implementation of this patented methodology addresses critical pain points in pharmaceutical intermediate production, delivering substantial value across the supply chain while maintaining the highest quality standards required for API manufacturing. The process redesign eliminates multiple unit operations from traditional synthesis routes, creating opportunities for significant cost reduction in chemical manufacturing while enhancing supply chain resilience through simplified logistics and reduced equipment requirements.
- Reduced Capital Expenditure and Equipment Complexity: The elimination of cryogenic processing units and specialized inert atmosphere systems required in conventional multi-step syntheses represents substantial capital savings. Standard jacketed reactors operating at moderate temperatures under ambient air conditions can be utilized without modification, avoiding expensive pressure-rated equipment typically needed for handling reactive sulfur compounds. This simplified equipment profile reduces initial investment costs while minimizing maintenance requirements and operator training needs, creating a more flexible manufacturing footprint that can be rapidly deployed across existing facilities without major capital outlays.
- Accelerated Production Timelines: The one-pot nature dramatically reduces cycle times compared to traditional multi-step approaches requiring intermediate isolation and purification between operations. By consolidating multiple synthetic steps into a single continuous process with straightforward workup (aqueous quench followed by standard extraction), production timelines are shortened significantly, enabling faster response to market demands and reducing working capital requirements. This time reduction is particularly valuable in fast-moving pharmaceutical development where rapid iteration is critical.
- Enhanced Environmental Sustainability and Waste Reduction: The process demonstrates environmental benefits through reduced solvent consumption, lower energy requirements, and minimized waste generation compared to conventional methods. The elimination of protection/deprotection steps removes entire waste streams associated with those operations while stable reagents like sodium metabisulfite reduce safety risks and handling costs. The high atom economy translates to less raw material consumption per unit product, directly supporting corporate sustainability goals while reducing disposal costs.
Revolutionizing Synthesis: Traditional Limitations vs. Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing methanesulfonyl-substituted nitrogen heterocycles typically involve multi-step sequences requiring harsh conditions and specialized reagents. The most common method employs substitution reactions between pre-formed nitrogen heterocycles and sodium methyl mercaptide, which necessitates multiple protection/deprotection steps due to reactivity constraints. These conventional pathways suffer from poor atom economy, require cryogenic conditions for certain steps, and generate significant waste streams from protecting group manipulations. The need for highly purified starting materials creates substantial barriers to scale-up with typical yields ranging from 45-65% across multiple operations.
The Novel Approach
The patented one-pot methodology overcomes these challenges through an elegant design leveraging commercially available starting materials in a single reaction vessel. 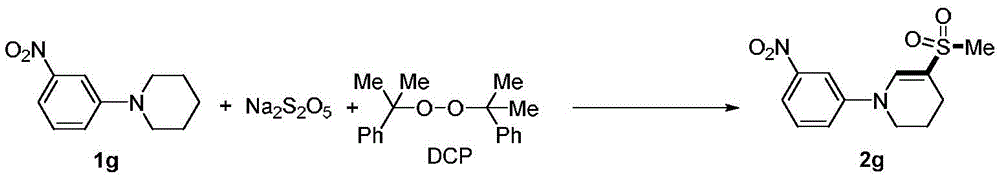 By utilizing stable reagents like sodium metabisulfite and dicumyl peroxide (DCP), the process eliminates hazardous sulfur handling while maintaining excellent functional group tolerance. The air atmosphere requirement simplifies reactor design compared to inert gas systems needed in conventional approaches, and the moderate temperature range is readily achievable with standard industrial equipment. The catalyst system creates a finely tuned environment promoting selective radical formation while suppressing unwanted side reactions across diverse substrate classes including electron-rich and sterically hindered variants.
By utilizing stable reagents like sodium metabisulfite and dicumyl peroxide (DCP), the process eliminates hazardous sulfur handling while maintaining excellent functional group tolerance. The air atmosphere requirement simplifies reactor design compared to inert gas systems needed in conventional approaches, and the moderate temperature range is readily achievable with standard industrial equipment. The catalyst system creates a finely tuned environment promoting selective radical formation while suppressing unwanted side reactions across diverse substrate classes including electron-rich and sterically hindered variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN112279801B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
