Innovative Metal-Free Synthesis for Scalable Production of High-Purity Quinolinyl-Triazole Pharmaceutical Intermediates
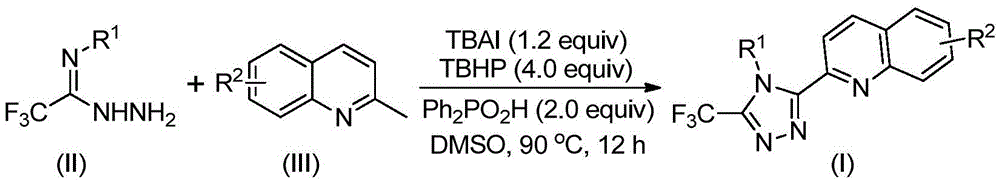
This patent (CN113307790B) introduces an innovative synthetic route for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical challenges in pharmaceutical intermediate manufacturing. The method employs tetrabutylammonium iodide as a catalyst with tert-butyl peroxide in DMSO solvent at 80–100°C, eliminating the need for anhydrous/anaerobic conditions and toxic heavy metal catalysts. This breakthrough enables scalable production of high-purity triazole derivatives with yields ranging from 51% to 97%, as demonstrated in the patent examples. The process design prioritizes operational simplicity while maintaining excellent functional group tolerance across diverse substrate combinations, making it particularly suitable for commercial-scale manufacturing of complex pharmaceutical intermediates.
Mechanistic Insights into Metal-Free Triazole Synthesis
The reaction mechanism begins with tetrabutylammonium iodide and tert-butyl peroxide promoting the oxidation of 2-methylquinoline to form 2-quinoline formaldehyde in situ, which then undergoes condensation with trifluoroethylimine hydrazide to generate a dehydrated hydrazone intermediate. This intermediate subsequently participates in oxidative iodination followed by intramolecular electrophilic substitution and aromatization to form the final triazole ring structure, with the possibility of radical pathway involvement as noted in the patent documentation. The absence of transition metal catalysts eliminates potential contamination pathways that typically require extensive purification steps in conventional methods, thereby simplifying the overall process flow and reducing production complexity while maintaining structural integrity of sensitive functional groups.
Impurity control is achieved through the careful selection of reaction conditions that favor the desired cyclization pathway while minimizing side reactions; the patent demonstrates that using diphenyl phosphoric acid as an additive significantly improves selectivity by stabilizing key intermediates during the cyclization step. The mild reaction conditions (80–100°C) prevent thermal degradation of sensitive functional groups, while the use of DMSO as solvent provides optimal polarity for intermediate stabilization without promoting unwanted side reactions. Post-reaction purification via column chromatography on silica gel effectively removes minor impurities, yielding products with high purity suitable for pharmaceutical applications as evidenced by the NMR and HRMS data provided in the patent examples.
Overcoming Traditional Limitations in Triazole Manufacturing
The Limitations of Conventional Methods
Traditional synthesis routes for quinolyl-substituted triazoles typically require quinoline-2-formic acid as starting material and involve multiple reaction steps under harsh conditions, resulting in low overall yields around 17% as cited in the patent background documentation. These conventional methods often necessitate strict anhydrous and anaerobic environments that increase operational complexity and equipment requirements while introducing significant process validation challenges for regulatory compliance. The multi-step nature creates substantial scalability barriers for commercial production, with each additional step introducing potential yield losses and quality control complications that make large-scale manufacturing economically unviable for many pharmaceutical intermediates.
The Novel Approach
The patented method overcomes these limitations by utilizing readily available 2-methylquinoline and trifluoroethylimine hydrazide as starting materials in a single-pot reaction that operates under ambient atmospheric conditions without requiring specialized equipment for moisture or oxygen exclusion. The tetrabutylammonium iodide catalyzed system enables direct oxidative cyclization at moderate temperatures (80–100°C), eliminating the need for transition metal catalysts while maintaining excellent functional group tolerance across diverse substrate combinations as demonstrated in the patent examples. This streamlined approach not only improves overall yield (51–97%) but also simplifies process validation and scale-up procedures by reducing the number of unit operations and eliminating critical quality attributes associated with metal catalyst removal.
Commercial Advantages for Supply Chain and Procurement Teams
This innovative synthesis methodology addresses critical pain points in pharmaceutical intermediate manufacturing by eliminating multiple cost drivers and supply chain vulnerabilities present in conventional production routes. The simplified process design reduces capital expenditure requirements while improving operational flexibility, enabling manufacturers to respond more effectively to fluctuating market demands without significant retooling investments. By removing the need for specialized handling equipment and complex purification systems, this approach creates substantial opportunities for cost reduction in chemical manufacturing while simultaneously enhancing supply chain resilience through process simplification.
- Reduced Capital and Operational Costs: The elimination of transition metal catalysts removes the need for expensive metal removal systems and associated validation procedures that typically account for significant portions of production costs in pharmaceutical manufacturing. Without requiring anhydrous or anaerobic reaction conditions, facilities can utilize standard processing equipment without costly modifications or specialized infrastructure investments. The simplified purification process using standard column chromatography on silica gel further reduces operational complexity and associated labor costs while maintaining high product quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Resilience: The use of commercially available starting materials with broad supplier options mitigates single-source dependency risks that often plague specialized pharmaceutical intermediate production. The robust reaction conditions tolerate minor variations in raw material quality, providing greater flexibility in supplier selection while maintaining consistent product quality. This process stability translates to more reliable delivery schedules and reduced vulnerability to supply chain disruptions that could otherwise impact critical drug manufacturing timelines.
- Scalable Production Pathway: The demonstrated gram-scale feasibility with straightforward process parameters creates a clear pathway for commercial scale-up to multi-kilogram or ton-scale production without requiring fundamental process re-engineering. The moderate temperature requirements (80–100°C) are easily achievable with standard industrial equipment, while the consistent yields across diverse substrates ensure predictable production planning and inventory management. This scalability directly supports reducing lead time for high-purity intermediates by enabling manufacturers to rapidly adjust production volumes based on customer demand.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While this tetrabutylammonium iodide catalyzed methodology demonstrates significant potential for triazole synthesis, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production with stringent purity specifications through our rigorous QC labs. Our technical team has successfully implemented similar metal-free catalytic systems across various pharmaceutical intermediate portfolios, ensuring seamless transition from laboratory discovery to commercial manufacturing while maintaining the highest quality standards required by global regulatory authorities.
We invite you to initiate a Customized Cost-Saving Analysis for your specific triazole-based compound requirements by contacting our technical procurement team, who will provide detailed route feasibility assessments and specific COA data to support your manufacturing decisions and supply chain optimization strategies.
