Advanced FeCl3-Catalyzed Synthesis of Azaanthracenes for Commercial Pharmaceutical Manufacturing
Advanced FeCl3-Catalyzed Synthesis of Azaanthracenes for Commercial Pharmaceutical Manufacturing
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern pharmaceutical and material science research. Azaanthracene compounds, particularly acridine derivatives, represent a class of privileged structures known for their extensive biological activities, ranging from antibacterial and antitumor properties to anti-inflammatory effects. Furthermore, these compounds have garnered significant attention in the field of optoelectronics, specifically as luminescent materials for Organic Light Emitting Diodes (OLEDs) due to their ability to form triplet states upon complexation with iridium. A pivotal advancement in this domain is documented in patent CN102219739A, which discloses a robust and high-yielding synthesis method for these valuable scaffolds. This technology shifts the paradigm from traditional, harsh reduction protocols to a more sustainable oxidative cyclization strategy utilizing inexpensive iron catalysts. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this catalytic system is essential for securing a stable supply chain of high-purity OLED material and API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of acridine derivatives has been fraught with significant technical and economic challenges that hinder large-scale manufacturing. Conventional bibliographical information often cites the reduction of oxynitrides or amines as a primary pathway, a method exemplified by earlier literature such as the work of Alonso et al. These traditional routes typically necessitate the use of complex and difficult-to-source feedstock, which immediately inflates the raw material costs and complicates the supply chain logistics for any fine chemical manufacturer. Moreover, these reactions frequently demand stronger reductive agents that are not only hazardous to handle but also generate substantial amounts of toxic waste, posing severe environmental compliance issues. The post-treatment processes associated with these older methods are often cumbersome, requiring rigorous purification steps to remove metal residues and by-products, which inevitably drags down the overall process efficiency. Consequently, the pursuit of cost reduction in electronic chemical manufacturing has been stifled by these inherent inefficiencies in the legacy synthetic technologies.
The Novel Approach
In stark contrast to the limitations of the past, the methodology described in the referenced patent introduces a streamlined catalytic cycle that fundamentally simplifies the production landscape. By employing iron(III) chloride (FeCl3) as a Lewis acid catalyst in conjunction with morpholine in a toluene solvent system, this novel approach achieves high conversion rates under relatively mild thermal conditions. The reaction utilizes o-anilino substituted aromatic aldehydes as the primary building blocks, which are significantly easier to synthesize and procure compared to the precursors required for reduction-based routes. The process operates at a manageable temperature of 120°C under nitrogen protection, ensuring safety and reproducibility while delivering separation yields that consistently exceed 80%, with some examples reaching as high as 93%. This transition to an oxidative cyclization mechanism not only enhances the atom economy but also drastically simplifies the downstream processing, making it an ideal candidate for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
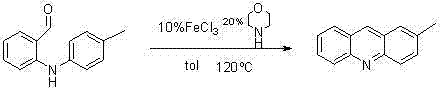
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The efficacy of this synthesis lies in the synergistic interaction between the iron catalyst and the organic substrates, facilitating a cascade of transformations that construct the tricyclic azaanthracene core with high precision. Mechanistically, the FeCl3 acts as a potent Lewis acid, coordinating with the carbonyl oxygen of the aromatic aldehyde to increase its electrophilicity, thereby promoting nucleophilic attack by the adjacent amine moiety. This initial condensation likely forms an imine intermediate, which subsequently undergoes an intramolecular electrophilic aromatic substitution or oxidative cyclization to close the central ring. The presence of morpholine plays a critical dual role, potentially acting as a base to neutralize generated protons and as a ligand that modulates the Lewis acidity of the iron center, preventing catalyst deactivation. This delicate balance ensures that the reaction proceeds with high selectivity, minimizing the formation of oligomeric by-products or incomplete cyclization species that often plague similar acid-catalyzed condensations. For technical teams, understanding this mechanistic pathway is vital for optimizing reaction parameters and ensuring the consistent quality of the high-purity OLED material produced.
Furthermore, the impurity profile of the resulting azaanthracene compounds is exceptionally clean, a direct result of the high chemoselectivity of the iron-catalyzed system. The reaction conditions are sufficiently robust to tolerate various electron-donating and electron-withdrawing groups on the aromatic rings, such as methyl, methoxy, and chloro substituents, without compromising the integrity of the final product. This tolerance suggests that the catalytic cycle is resilient against steric and electronic variations, allowing for the synthesis of a diverse library of derivatives from a single standardized protocol. The workup procedure, involving simple aqueous quenching and ether extraction, effectively removes the polar catalyst residues and inorganic salts, leaving the organic phase rich in the desired product. Such a clean reaction profile significantly reduces the burden on the purification team, as column chromatography can be performed with standard silica gel and common eluent systems like petroleum ether and ethyl acetate, ensuring that the final isolation yields remain commercially viable.
How to Synthesize Azaanthracene Efficiently
The operational simplicity of this patented method makes it highly accessible for laboratory replication and subsequent industrial adaptation. The protocol dictates a precise molar ratio of catalyst to substrate, typically utilizing a catalytic amount of FeCl3 relative to the aromatic aldehyde, which is dissolved in toluene to create a homogeneous reaction mixture. The addition of morpholine is critical and must be timed correctly to ensure the proper formation of the active catalytic species before the onset of heating. Once the reagents are combined in a Schlenk tube under an inert atmosphere, the mixture is heated to reflux, where the thermal energy drives the cyclization to completion over a period of approximately 26 hours. Detailed standardized synthesis steps, including specific quenching volumes and chromatography gradients, are provided in the technical documentation below to assist process chemists in replicating these high-yield results.
- Under nitrogen protection, sequentially add FeCl3, morpholine, o-anilino substituted aromatic aldehyde, and toluene into a Schlenk sealed tube.
- Stir the mixture and raise the temperature to 120°C, maintaining slight reflux for 26 hours to ensure complete cyclization.
- Cool to room temperature, quench with water, extract with ether, wash with saturated brine, dry over Na2SO4, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this FeCl3-catalyzed methodology offers profound benefits that directly address the pain points of modern chemical procurement and supply chain management. The shift away from exotic reductants and complex precursors to commodity chemicals like toluene, morpholine, and iron salts creates a much more resilient supply base that is less susceptible to market volatility. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines required by global pharmaceutical and electronics clients. Furthermore, the elimination of hazardous reducing agents simplifies the regulatory compliance landscape, reducing the administrative burden and safety risks associated with handling dangerous goods. For procurement managers, this translates into a more predictable cost structure and a lower total cost of ownership for the manufactured intermediates.
- Cost Reduction in Manufacturing: The economic implications of replacing expensive and specialized reagents with ubiquitous industrial chemicals cannot be overstated. Iron(III) chloride is one of the most cost-effective Lewis acids available globally, and its usage in catalytic quantities means that the raw material cost per kilogram of product is drastically minimized. Additionally, the simplified workup procedure eliminates the need for expensive scavengers or complex distillation setups often required to remove heavy metal contaminants from other catalytic systems. This streamlining of the process flow leads to substantial cost savings in both utility consumption and labor hours, allowing manufacturers to offer more competitive pricing without sacrificing margin. The high isolated yields reported in the patent further amplify these savings by maximizing the output from every batch of raw materials processed.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as o-anilino substituted aromatic aldehydes ensures that the supply chain remains robust even during periods of global logistical disruption. Unlike specialized reagents that may have single-source suppliers or long lead times, the key components for this synthesis are produced by multiple vendors worldwide, providing procurement teams with ample flexibility to negotiate terms and secure inventory. The stability of the catalyst and reagents in air also simplifies storage and transportation requirements, reducing the risk of degradation during transit. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream customers receive their materials exactly when needed for their own production cycles.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its use of standard solvents and moderate reaction temperatures, which are easily managed in large-scale reactors without requiring specialized high-pressure or cryogenic equipment. The waste stream generated is primarily composed of aqueous salts and organic solvents that can be treated using conventional wastewater treatment facilities, aligning with increasingly stringent environmental regulations. The absence of toxic heavy metals in the final product simplifies the disposal of solid waste and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for multinational corporations evaluating their supplier networks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is critical for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios. The responses cover aspects ranging from reaction optimization to product purity standards, ensuring a comprehensive overview of the technology's capabilities.
Q: What are the key advantages of the FeCl3-catalyzed method over traditional reduction routes?
A: Unlike traditional methods requiring complex feedstock and strong reducing agents, this novel approach utilizes inexpensive FeCl3 and morpholine, offering simpler post-treatment and significantly higher yields (up to 93%).
Q: Is this synthesis method scalable for industrial production of acridine derivatives?
A: Yes, the process uses common solvents like toluene and stable catalysts, with a straightforward workup procedure involving extraction and chromatography, making it highly suitable for commercial scale-up.
Q: What represents the substrate scope for this azaanthracene synthesis?
A: The method demonstrates excellent tolerance for various substituents including hydrogen, methyl, methoxy, and chloro groups on the aromatic ring, allowing for the diverse synthesis of functionalized acridine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaanthracene Supplier
The technological breakthroughs detailed in patent CN102219739A underscore the immense potential of iron-catalyzed cyclization for the efficient production of high-value heterocyclic compounds. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, industrial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of specific acridine derivatives or large-volume supply of standard intermediates, our capabilities are aligned to support your most demanding projects.
We invite you to engage with our technical sales team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Let us demonstrate how our commitment to innovation and quality can drive value for your organization and support your long-term strategic goals in the pharmaceutical and electronic materials sectors.
