Advanced Synthesis of DL-Alpha-Aminocaprolactam: Scalable Routes for Lysine Precursors
Advanced Synthesis of DL-Alpha-Aminocaprolactam: Scalable Routes for Lysine Precursors
The pharmaceutical and fine chemical industries are constantly seeking robust, safe, and cost-effective pathways for producing essential amino acid precursors. Patent CN102093292A introduces a significant technological advancement in the synthesis of DL-alpha-aminocaprolactam, a critical intermediate for lysine production. This innovation addresses long-standing safety and efficiency concerns associated with traditional synthetic routes by employing a two-step strategy involving nucleophilic substitution and catalytic hydrogenolysis. By shifting away from hazardous reagents like sodium azide, this method offers a streamlined approach that enhances both operator safety and process reliability. For R&D directors and procurement managers alike, understanding the nuances of this patent is crucial for optimizing supply chains and ensuring the consistent availability of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DL-alpha-aminocaprolactam has been plagued by significant safety hazards and operational complexities. Traditional literature describes methods utilizing liquid ammonia or the Gabriel synthesis, which often suffer from low yields and the formation of difficult-to-separate isomers such as piperidine-2-carboxamide. More critically, some established routes rely on sodium azide (NaN3) as a nucleophile. While chemically effective, sodium azide is highly toxic, shock-sensitive, and explosive, posing severe risks during storage, handling, and reaction scale-up. Furthermore, alternative multi-step routes starting from cyclohexene involve harsh conditions, including the use of liquid sulfur dioxide and chlorosulfonic acid, which complicate waste treatment and increase the environmental footprint. These factors collectively drive up production costs and create bottlenecks in the supply chain for reliable pharmaceutical intermediates suppliers.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift towards safer and more manageable chemistry. By utilizing alpha-halo caprolactam and benzylamine as starting materials, the process avoids the inherent dangers of azide chemistry entirely. The first step involves a controlled nucleophilic substitution to form an N-benzyl protected intermediate, which is stable and easy to isolate. The second step employs catalytic hydrogenolysis to remove the benzyl protecting group, yielding the desired DL-alpha-aminocaprolactam. This approach not only simplifies the purification process—often requiring only simple cold precipitation and solvent concentration—but also ensures higher overall yields. For manufacturers focused on cost reduction in pharmaceutical intermediates manufacturing, this reduction in processing steps and hazard mitigation translates directly into improved economic viability and operational continuity.
Mechanistic Insights into Nucleophilic Substitution and Catalytic Debenzylation
The core of this synthetic strategy lies in the efficient displacement of the halogen atom on the caprolactam ring. In the first stage, the nitrogen atom of benzylamine acts as a nucleophile, attacking the alpha-carbon of the alpha-halo caprolactam. This reaction is facilitated by the presence of a base, such as potassium carbonate or sodium carbonate, which neutralizes the hydrogen halide byproduct and drives the equilibrium forward. The choice of solvent, typically polar aprotic solvents like N,N-dimethylformamide (DMF) or 1,2-propanediol, is critical for solubilizing the reactants and stabilizing the transition state. The reaction temperature is carefully controlled between 80°C and 150°C to maximize conversion while minimizing side reactions. This precise control ensures that the resulting N-benzyl-DL-alpha-aminocaprolactam is formed with high selectivity, avoiding the isomeric impurities that plague ammonia-based substitutions.
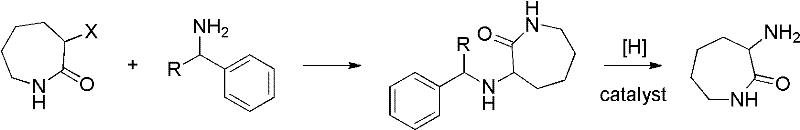
Following the formation of the protected intermediate, the second mechanistic phase involves catalytic hydrogenolysis. This step utilizes a transition metal catalyst, such as Palladium on Carbon (Pd/C) or Raney Nickel, in the presence of a hydrogen donor like formic acid or hydrogen gas. The catalyst facilitates the cleavage of the carbon-nitrogen bond between the benzyl group and the amine nitrogen. This debenzylation process is highly specific and occurs under relatively mild reflux conditions in solvents like methanol or ethanol. The mechanism ensures that the lactam ring remains intact while the protecting group is cleanly removed. This specificity is vital for maintaining the structural integrity of the final product, ensuring that the DL-alpha-aminocaprolactam produced meets the stringent purity specifications required for downstream amino acid synthesis.
How to Synthesize DL-Alpha-Aminocaprolactam Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and reaction conditions to achieve optimal results. The patent outlines a clear protocol where the molar ratios of reactants are tuned to balance cost and conversion efficiency. Typically, a slight excess of benzylamine and base is used to ensure complete consumption of the more valuable alpha-halo caprolactam. The subsequent hydrogenolysis step requires precise catalyst loading to maintain reaction rates without excessive metal usage. Detailed standardized operating procedures are essential for reproducibility, especially when transitioning from laboratory bench scales to pilot plant operations. The following guide summarizes the critical operational parameters derived from the patent data to assist technical teams in process validation.
- Conduct nucleophilic substitution by reacting alpha-halo caprolactam with benzylamine in an organic solvent like DMF with a base such as potassium carbonate at 80-150°C.
- Isolate the intermediate N-benzyl-DL-alpha-aminocaprolactam through water precipitation and filtration.
- Perform catalytic hydrogenolysis using Pd/C and formic acid in methanol under reflux to remove the benzyl group and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The elimination of hazardous materials like sodium azide drastically reduces the regulatory burden and insurance costs associated with raw material storage and transport. Furthermore, the simplified workup procedures, which often involve simple filtration and crystallization rather than complex chromatographic separations, lead to significant reductions in processing time and solvent consumption. These efficiencies contribute to substantial cost savings in pharmaceutical intermediates manufacturing, allowing for more competitive pricing structures in the global market. Additionally, the robustness of the reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining long-term contracts with downstream API manufacturers.
- Cost Reduction in Manufacturing: The removal of expensive and dangerous reagents like sodium azide eliminates the need for specialized containment equipment and costly waste disposal protocols. By utilizing commodity chemicals such as benzylamine and formic acid, the raw material costs are stabilized, and the reliance on volatile specialty chemical markets is reduced. The high yield and simple isolation steps further minimize material loss, ensuring that the theoretical output is closely matched by actual production volumes, thereby optimizing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The starting materials for this process, including alpha-halo caprolactams and benzylamine, are widely available from multiple global sources, reducing the risk of supply disruptions. Unlike routes dependent on niche reagents or complex multi-step sequences, this two-step process shortens the manufacturing lead time significantly. This agility allows suppliers to respond more rapidly to fluctuations in market demand for lysine precursors, ensuring a steady flow of high-purity pharmaceutical intermediates to clients without prolonged delays.
- Scalability and Environmental Compliance: The reaction conditions are compatible with standard stainless steel reactors and do not require extreme pressures or cryogenic temperatures, facilitating easy scale-up from kilograms to metric tons. The avoidance of heavy metal contaminants in the final product, thanks to efficient filtration of the heterogeneous catalyst, simplifies the purification process and ensures compliance with strict environmental and safety regulations. This green chemistry approach aligns with modern sustainability goals, making the supply chain more resilient against future regulatory tightening regarding hazardous waste.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is key to evaluating its potential for your specific application. The following questions address common concerns regarding safety, purity, and scalability based on the patent disclosures. These insights are designed to help technical decision-makers assess the feasibility of integrating this route into their existing production frameworks or sourcing strategies.
Q: Why is the benzylamine route preferred over sodium azide methods for DL-alpha-aminocaprolactam?
A: The benzylamine route eliminates the use of highly toxic and explosive sodium azide (NaN3), significantly improving operational safety and reducing hazardous waste disposal costs compared to traditional azide-based substitutions.
Q: What are the critical purity challenges in synthesizing amino caprolactams?
A: Traditional methods often generate isomeric impurities like piperidine-2-carboxamide which are difficult to separate. The novel two-step method described minimizes isomer formation, allowing for simpler purification via recrystallization.
Q: Can this synthesis method be scaled for industrial lysine production?
A: Yes, the process utilizes common solvents like DMF and methanol and standard catalysts like Pd/C, making it highly suitable for large-scale industrial mass production without requiring specialized high-pressure equipment typical of other routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DL-Alpha-Aminocaprolactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of safe and efficient synthetic routes for high-value intermediates like DL-alpha-aminocaprolactam. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab concept to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to process safety and environmental responsibility mirrors the advantages offered by the patented technology discussed herein.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your production efficiency and reduce your overall operational risks.
