Scalable Metal-Free Photocatalytic Synthesis of Fluorescent Indolizine-1,2-Diones
Scalable Metal-Free Photocatalytic Synthesis of Fluorescent Indolizine-1,2-Diones
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies to construct complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN111269228A introduces a groundbreaking advancement in this domain by detailing a novel, metal-free photocatalytic oxidation method for the preparation of indolizine ring-1,2-diones and their derivatives. These compounds are not merely academic curiosities; they represent a class of highly valuable structures with significant fluorescence activity, making them indispensable for applications ranging from drug discovery to advanced optical materials. The core innovation lies in the utilization of visible light-induced intermolecular cross-dehydrogenative coupling (CDC), which bypasses the traditional reliance on harsh oxidants or precious metal catalysts. By leveraging commercially available starting materials such as indolizines and 2-oxo-2-phenylacetaldehyde in the presence of an organic photocatalyst like Rose Bengal, this technology offers a pathway to high-purity intermediates that aligns perfectly with modern green chemistry principles. For R&D directors and procurement strategists, this patent signals a shift towards more economical and environmentally benign manufacturing processes that do not compromise on yield or structural diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized indolizine derivatives has heavily relied on transition metal-catalyzed C-H functionalization strategies involving elements such as Palladium, Copper, Silver, Rhodium, or Ruthenium. While these methods have undeniably propelled organic synthesis forward, they carry inherent drawbacks that pose significant challenges for large-scale industrial adoption. The primary concern is the toxicity and high cost associated with these heavy metals, which necessitates rigorous purification steps to ensure residual metal levels meet stringent regulatory standards for pharmaceutical intermediates. Furthermore, many conventional protocols require elevated temperatures, strong oxidants, or inert atmospheres, all of which increase energy consumption and operational complexity. The disposal of metal-contaminated waste streams adds another layer of environmental burden and cost, creating a bottleneck for companies aiming to reduce their carbon footprint. Additionally, the substrate scope in some traditional methods can be limited by the sensitivity of the metal catalysts to specific functional groups, restricting the chemical diversity accessible to medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN111269228A utilizes a visible-light-driven organophotocatalytic system that operates under exceptionally mild conditions. By employing organic dyes such as Rose Bengal, Eosin Y, or Fluorescein as photocatalysts, the process completely eliminates the need for transition metals, thereby removing the risk of heavy metal contamination from the outset. The reaction is driven by low-energy blue LED irradiation at room temperature and atmospheric pressure, utilizing molecular oxygen from the air as the terminal oxidant, which is both cost-effective and environmentally benign. This approach not only simplifies the reaction setup but also dramatically improves the atom economy and safety profile of the synthesis. The versatility of this method is demonstrated by its ability to tolerate a wide range of substituents, including alkyl, electron-withdrawing, and electron-donating groups, allowing for the efficient construction of diverse indolizine-1,2-dione libraries. 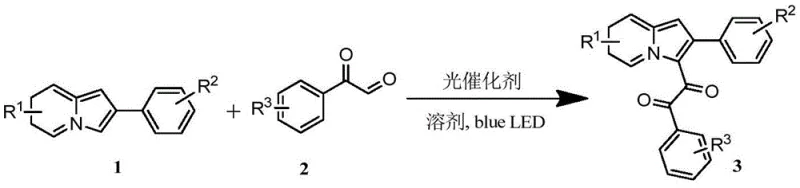
Mechanistic Insights into Visible-Light Photocatalytic Oxidation
The mechanistic foundation of this transformation rests on the principles of photo-induced electron transfer and radical chemistry. Upon irradiation with blue light, the organic photocatalyst absorbs photons and transitions to an excited state, possessing sufficient redox potential to initiate single-electron transfer processes with the substrates. This excitation facilitates the activation of the C-H bond at the C-3 position of the indolizine ring, generating a reactive radical intermediate. Simultaneously, the 2-oxo-2-phenylacetaldehyde undergoes activation, likely forming an acyl radical species through hydrogen atom abstraction or oxidative pathways mediated by the excited catalyst and molecular oxygen. The subsequent cross-coupling of these radical intermediates leads to the formation of the new carbon-carbon bond, constructing the 1,2-dicarbonyl motif characteristic of the target products. The regeneration of the ground-state photocatalyst by molecular oxygen closes the catalytic cycle, producing water or hydrogen peroxide as the only byproducts. This elegant mechanism ensures high regioselectivity for the C-3 position of the indolizine ring, minimizing the formation of unwanted isomers and simplifying the downstream purification process significantly.
From an impurity control perspective, the absence of metal catalysts inherently reduces the complexity of the impurity profile, as there are no metal-ligand complexes or metal-mediated side reactions to manage. The mild reaction conditions further suppress thermal degradation pathways that often plague high-temperature syntheses, resulting in cleaner crude reaction mixtures. The use of polar aprotic solvents like DMSO or DMF ensures good solubility for both the polar intermediates and the final diketone products, facilitating homogeneous reaction kinetics. Understanding this mechanism allows process chemists to fine-tune parameters such as light intensity, catalyst loading, and oxygen flow to maximize conversion rates. The robustness of the radical pathway means that electronic variations on the aromatic rings of the substrates have a manageable impact on reaction efficiency, providing the flexibility needed for synthesizing analogs with specific electronic properties for fluorescence tuning or biological activity modulation.
How to Synthesize Indolizine-1,2-dione Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires attention to light penetration and mixing efficiency to ensure uniform irradiation of the reaction mixture. The standard procedure involves dissolving the indolizine substrate and the organic photocatalyst in a suitable solvent such as dimethyl sulfoxide, followed by the addition of the alpha-keto aldehyde coupling partner. The reaction vessel is then subjected to blue LED irradiation while stirring under an aerobic atmosphere for a defined period, typically around 12 hours at room temperature. Workup generally involves simple solvent removal and chromatographic purification, although crystallization may be feasible for certain derivatives on a larger scale. The detailed standardized synthesis steps for replicating this high-yield transformation are outlined below.
- Combine 2-phenylindolizine and Rose Bengal photocatalyst in DMSO solvent within a reaction vessel equipped with a stirrer.
- Add 2-oxo-2-phenylacetaldehyde to the mixture and irradiate with 20W blue LED light at room temperature for 12 hours under air.
- Remove the solvent via rotary evaporation and purify the crude residue using flash column chromatography to isolate the target indolizine-1,2-dione.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free photocatalytic technology presents a compelling value proposition centered on cost reduction and supply security. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like Palladium and Rhodium are subject to significant market fluctuations and geopolitical supply risks. Furthermore, the simplified downstream processing required to remove metal residues translates directly into reduced manufacturing time and lower consumption of specialized scavenging resins or filtration media. This streamlining of the production workflow enhances overall throughput and allows facilities to allocate resources more efficiently across their product portfolios. The reliance on commodity chemicals and standard lighting equipment also means that the barrier to entry for manufacturing these intermediates is lower, fostering a more competitive and resilient supply base.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the substitution of expensive transition metal catalysts with inexpensive organic dyes like Rose Bengal, which are available in bulk quantities at a fraction of the cost. Additionally, the removal of heavy metal clearance steps eliminates the need for costly analytical testing and specialized purification columns, leading to substantial savings in both material and labor costs. The energy efficiency of using low-power LED lights compared to heating mantles or high-pressure reactors further contributes to a reduced operational expenditure profile. These cumulative savings allow for more competitive pricing strategies when supplying high-purity pharmaceutical intermediates to global clients.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials such as substituted indolizines and phenylglyoxals, manufacturers can mitigate the risk of supply disruptions often associated with specialized reagents. The robustness of the reaction conditions, which tolerate ambient temperature and pressure, reduces the dependency on complex infrastructure and specialized high-pressure equipment that can be prone to maintenance downtime. This operational simplicity ensures consistent production schedules and reliable delivery timelines, which are critical for maintaining the continuity of downstream drug development programs. The ability to source catalysts and solvents from multiple global suppliers further diversifies the supply chain, insulating the manufacturing process from regional shortages.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with increasingly stringent environmental regulations, reducing the liability associated with hazardous waste disposal. The use of molecular oxygen as the oxidant generates minimal waste compared to stoichiometric chemical oxidants, simplifying effluent treatment and lowering environmental compliance costs. The mild conditions facilitate easier scale-up from gram to kilogram scales without the need for extensive re-optimization of safety parameters, accelerating the timeline from process development to commercial production. This scalability ensures that the technology can meet the growing demand for fluorescent materials and bioactive intermediates without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing manufacturing frameworks.
Q: What are the advantages of this metal-free photocatalytic method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metals like Palladium or Copper, significantly reducing raw material costs and removing the complex downstream processing required for heavy metal residue removal, thereby enhancing environmental compliance.
Q: Can this synthesis protocol accommodate diverse substrate substituents?
A: Yes, the patent demonstrates excellent substrate tolerance, successfully synthesizing derivatives with various electron-withdrawing and electron-donating groups on both the indolizine ring and the aldehyde component, ensuring broad applicability for library synthesis.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction proceeds under mild conditions at room temperature and atmospheric pressure using common organic solvents like DMSO or DMF, driven by low-energy 20W blue LED irradiation, which simplifies equipment requirements and improves operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine-1,2-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free photocatalysis in the production of high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative patent can be realized at an industrial level. Our state-of-the-art facilities are equipped with advanced photoreactors capable of delivering uniform light distribution for large-batch synthesis, guaranteeing consistent quality and yield. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of indolizine-1,2-dione meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to process excellence ensures that we can deliver complex fluorescent intermediates with the reliability and speed your projects demand.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug discovery or material science program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you secure a sustainable and cost-effective supply chain for your critical indolizine derivatives.
