Scalable Metal-Free Hydroamination for High-Purity Pharmaceutical Intermediates Manufacturing
Scalable Metal-Free Hydroamination for High-Purity Pharmaceutical Intermediates Manufacturing
The construction of carbon-nitrogen bonds remains a cornerstone of modern organic synthesis, particularly within the realm of fine chemicals and pharmaceutical intermediates where structural complexity and purity are paramount. Patent CN107382741B introduces a transformative methodology for the intermolecular hydroamination of alkynes and amines, leveraging the potent Lewis acidity of tris(pentafluorophenyl)borane, commonly known as B(C6F5)3. This technology represents a significant departure from traditional transition-metal-catalyzed processes, offering a pathway that is not only atom-economical but also operationally robust for industrial scale-up. By utilizing a metal-free catalyst system, this approach directly addresses the stringent regulatory requirements regarding residual heavy metals in active pharmaceutical ingredients (APIs), positioning it as a highly attractive route for reliable pharmaceutical intermediates supplier networks seeking to optimize their manufacturing portfolios.
The core innovation lies in the ability to couple terminal alkynes with primary aromatic amines under relatively mild thermal conditions, ranging from 25°C to 100°C, within an aprotic polar solvent environment. This flexibility allows for the synthesis of a diverse array of secondary aromatic amines, which serve as critical building blocks in medicinal chemistry. The reaction mechanism avoids the use of expensive and potentially toxic transition metals such as palladium, rhodium, or lanthanides, which often necessitate complex ligand systems and rigorous purification protocols. Instead, the boron-based catalyst activates the alkyne moiety through polarization, facilitating a direct nucleophilic attack by the amine nitrogen. This streamlined process results in high yields, with specific embodiments demonstrating conversion efficiencies reaching up to 99%, thereby maximizing material throughput and minimizing waste generation in cost reduction in pharmaceutical intermediates manufacturing.
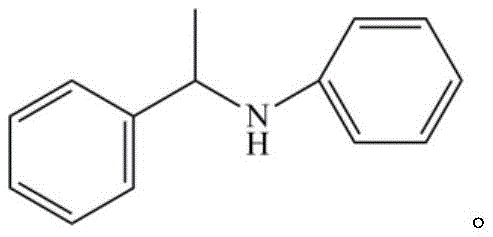
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of secondary amines via hydroamination has relied heavily on transition metal catalysis, which presents several inherent challenges for large-scale commercial production. Traditional methods often employ catalysts based on late transition metals or lanthanides, which, while effective, suffer from issues related to stability, cost, and environmental impact. The synthesis of these metal complexes frequently requires multi-step ligand preparation, adding significant upstream costs and complexity to the supply chain. Furthermore, the presence of metal residues in the final product is a major concern for the pharmaceutical industry, where regulatory bodies impose strict limits on parts-per-million (ppm) levels of heavy metals. Removing these residues often requires additional downstream processing steps, such as treatment with scavengers or extensive chromatography, which can drastically reduce overall yield and increase production time. Additionally, many conventional hydroamination protocols require harsh reaction conditions, including high temperatures or pressures, which can limit the functional group tolerance and lead to the formation of unwanted by-products, complicating the purification of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology described in CN107382741B utilizes tris(pentafluorophenyl)borane, a commercially available and stable main-group Lewis acid, to drive the hydroamination reaction. This novel approach eliminates the need for transition metals entirely, thereby removing the risk of metal contamination and the associated costs of metal removal. The reaction conditions are remarkably mild, operating effectively between 25°C and 100°C in common solvents like chlorobenzene or tetrahydrofuran, which enhances safety and reduces energy consumption. The catalyst itself does not require pre-synthesis or complex activation, simplifying the operational workflow and reducing the lead time for process development. Moreover, the system exhibits excellent substrate scope, accommodating various substituents on both the alkyne and the amine components, including electron-withdrawing and electron-donating groups. This versatility enables the commercial scale-up of complex pharmaceutical intermediates with diverse structural motifs, providing a robust platform for the rapid synthesis of compound libraries and key drug precursors without the baggage of traditional metal catalysis.
Mechanistic Insights into B(C6F5)3-Catalyzed Hydroamination
The mechanistic foundation of this reaction relies on the strong Lewis acidic nature of the boron center in B(C6F5)3, which interacts with the pi-electron system of the terminal alkyne. Upon coordination, the alkyne undergoes significant polarization, rendering the beta-carbon highly electrophilic and susceptible to nucleophilic attack by the lone pair of electrons on the nitrogen atom of the primary aromatic amine. This activation lowers the energy barrier for the addition reaction, allowing it to proceed under mild thermal conditions without the need for external bases or harsh activators. The active hydrogen on the nitrogen atom plays a crucial role in the catalytic cycle by promoting the dissociation of the boron species after the initial addition, regenerating the catalyst and releasing the enamine intermediate. This cycle is highly efficient, as evidenced by the low catalyst loading required (typically 10-20 mol%) to achieve near-quantitative conversions. The subsequent reduction of the enamine intermediate to the saturated secondary amine is achieved using standard reducing agents such as lithium aluminum hydride or sodium borohydride, completing the transformation with high stereochemical integrity and minimal side reactions.
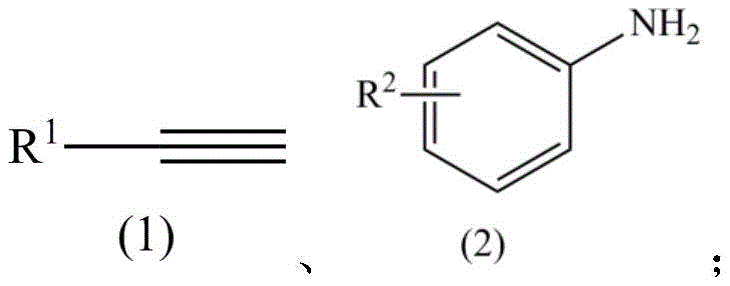
From an impurity control perspective, the metal-free nature of this catalytic system offers distinct advantages for the production of pharmaceutical-grade materials. In traditional metal-catalyzed processes, trace amounts of metal can coordinate with product amines, forming stable complexes that are difficult to remove and can catalyze degradation pathways during storage. By utilizing a boron-based catalyst, these risks are mitigated, as boron residues are generally easier to manage and less toxic than heavy metals. Furthermore, the mild reaction conditions minimize thermal degradation of sensitive functional groups, such as halogens or nitro groups, which are often present in bioactive molecules. The reaction's high selectivity ensures that the primary amine reacts specifically with the alkyne triple bond, avoiding oligomerization or polymerization of the alkyne, which are common side reactions in less controlled systems. This precision in bond formation translates directly to a cleaner crude reaction profile, reducing lead time for high-purity pharmaceutical intermediates by simplifying the isolation and purification stages required to meet stringent quality specifications.
How to Synthesize Secondary Aromatic Amines Efficiently
The synthesis protocol outlined in the patent provides a straightforward and reproducible method for accessing valuable secondary amine scaffolds. The process begins with the dissolution of the B(C6F5)3 catalyst in an anhydrous aprotic solvent, followed by the addition of the terminal alkyne and primary aromatic amine under an inert atmosphere to prevent moisture interference. The mixture is then heated to the desired temperature, typically between 25°C and 100°C, and maintained for a period of 6 to 24 hours to ensure complete conversion to the enamine intermediate. Following the hydroamination step, the reaction mixture is cooled, and a reducing agent is introduced to convert the unstable enamine into the stable secondary amine product. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are provided in the guide below to facilitate immediate implementation in R&D settings.
- Combine terminal alkyne and primary aromatic amine in an aprotic polar solvent under a protective atmosphere.
- Add tris(pentafluorophenyl)borane catalyst and react at 25-100°C for 6-24 hours to form the enamine intermediate.
- Reduce the intermediate with a reducing agent like LiAlH4 at 70-90°C, then quench and purify to obtain the secondary amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free hydroamination technology offers compelling economic and logistical benefits that extend beyond simple reaction yields. The elimination of precious metal catalysts removes a significant variable cost component, as prices for metals like palladium and rhodium are subject to high market volatility and geopolitical supply risks. By switching to a boron-based system, manufacturers can stabilize their raw material costs and secure a more predictable pricing structure for long-term production contracts. Additionally, the simplicity of the catalyst, which is commercially available off-the-shelf without the need for custom synthesis, drastically shortens the procurement lead time. This agility allows supply chain teams to respond more quickly to fluctuating demand signals from downstream pharmaceutical clients, ensuring continuity of supply even in tight market conditions. The operational simplicity also reduces the burden on quality control laboratories, as fewer tests are required to certify the absence of heavy metal contaminants.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of expensive transition metal catalysts and the associated ligands, which often account for a substantial portion of the bill of materials in fine chemical synthesis. Furthermore, the removal of heavy metal scavenging steps and the simplified purification workflow significantly reduce the consumption of auxiliary materials such as silica gel, solvents, and specialized filtration media. The high atom economy of the hydroamination reaction ensures that the majority of the starting mass is incorporated into the final product, minimizing waste disposal costs and maximizing the efficiency of raw material utilization. These factors combine to deliver substantial cost savings per kilogram of produced intermediate, enhancing the overall margin profile for commercial manufacturing operations without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like B(C6F5)3 and common solvents such as chlorobenzene or THF insulates the production process from the supply chain fragility associated with specialized organometallic reagents. These starting materials are produced by multiple global suppliers, reducing the risk of single-source bottlenecks and ensuring consistent availability. The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require high-pressure equipment, allows for flexible manufacturing across different facilities, further diversifying supply risk. This reliability is critical for maintaining the production schedules of downstream API manufacturers, who depend on the timely delivery of key intermediates to meet their own regulatory filing and market launch deadlines, thereby strengthening the strategic partnership between chemical suppliers and pharmaceutical innovators.
- Scalability and Environmental Compliance: The transition to a metal-free process aligns perfectly with the increasing global emphasis on green chemistry and sustainable manufacturing practices. By eliminating heavy metals, the process generates waste streams that are easier to treat and dispose of, reducing the environmental footprint and the regulatory burden associated with hazardous waste management. The mild reaction conditions also contribute to lower energy consumption, as there is no need for extreme heating or pressurization, supporting corporate sustainability goals. From a scalability perspective, the exothermic nature of the reaction is manageable, and the use of standard glass-lined or stainless-steel reactors facilitates seamless transfer from laboratory bench scale to multi-ton commercial production. This ease of scale-up ensures that the technology can meet the growing demand for complex amine intermediates without requiring significant capital investment in new infrastructure or specialized containment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroamination technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN107382741B, ensuring accuracy and relevance for process chemists and project managers evaluating this route. Understanding these details is essential for assessing the feasibility of integrating this method into existing manufacturing workflows and for optimizing the process parameters to suit specific substrate requirements. The insights provided here aim to clarify the operational advantages and technical capabilities of the B(C6F5)3 catalytic system.
Q: What are the advantages of using B(C6F5)3 over transition metal catalysts for hydroamination?
A: B(C6F5)3 is a metal-free Lewis acid that eliminates the risk of heavy metal contamination in the final product, which is critical for pharmaceutical compliance. It is also commercially available, stable, and does not require complex ligand synthesis, simplifying the supply chain and reducing costs.
Q: What is the substrate scope for this hydroamination reaction?
A: The method supports a wide range of substrates. R1 can be C1-C10 alkyl or aryl groups (including phenyl, benzyl, p-fluorophenyl), and R2 on the aniline ring can be hydrogen, alkyl, alkoxy, cyano, trifluoromethyl, nitro, halogen, or amino groups, allowing for diverse chemical space exploration.
Q: How does this process ensure high purity for API intermediates?
A: By avoiding transition metals, the process removes the need for rigorous and often yield-lowering metal scavenging steps. The reaction proceeds under mild conditions (25-100°C) with high conversion rates (up to 99% in examples), minimizing side reactions and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secondary Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the metal-free hydroamination route described in CN107382741B and confirmed its potential for delivering high-quality secondary aromatic amines at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch meets stringent purity specifications required by top-tier pharmaceutical clients. We are committed to leveraging this advanced catalytic technology to provide our partners with superior intermediates that accelerate their drug development timelines.
We invite you to collaborate with us to explore how this cost-effective and sustainable synthesis route can benefit your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating the tangible economic advantages of switching to this metal-free protocol. Please contact us to request specific COA data for similar compounds and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the production of complex fine chemical intermediates.
