Advanced Copper-Catalyzed Synthesis of Substituted Allene Thioethers for Commercial Scale Production
Introduction to Next-Generation Allene Sulfide Technology
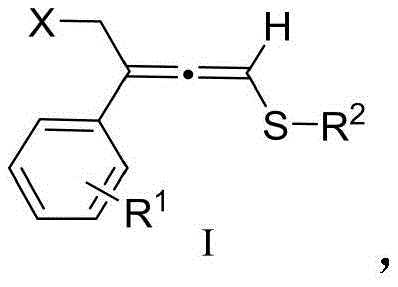
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, where efficiency and safety are paramount. A significant breakthrough in this domain is detailed in patent CN113292462B, which discloses a novel class of substituted allene thioether compounds and a highly efficient preparation method thereof. These compounds, characterized by the unique allene sulfide motif, serve as versatile building blocks for constructing complex molecular architectures found in bioactive molecules, including carbapenem antibiotics and various heterocyclic systems. The disclosed technology represents a paradigm shift from traditional, harsh synthetic protocols to a more sustainable and operationally simple copper-catalyzed approach. By leveraging mild reaction conditions and readily available starting materials, this innovation addresses critical bottlenecks in the supply chain of high-value fine chemical intermediates. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a competitive edge in the development of next-generation therapeutics and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allene sulfides has been plagued by significant operational challenges and safety concerns that hinder their widespread adoption in large-scale manufacturing. Traditional methodologies predominantly rely on the use of extremely strong bases, such as butyl lithium or potassium tert-butoxide, which necessitate stringent low-temperature conditions and an inert atmosphere to prevent decomposition or uncontrolled side reactions. These苛刻 conditions not only drive up energy costs but also introduce substantial safety risks associated with handling pyrophoric reagents on an industrial scale. Furthermore, the lack of selectivity in these conventional pathways often leads to a complex mixture of byproducts, including undesirable alkynyl sulfides, polymeric materials, and sulfide oxides. This poor selectivity complicates downstream purification processes, resulting in lower overall yields and increased waste generation, which is antithetical to the principles of green chemistry and cost-effective production required by modern supply chains.
The Novel Approach
In stark contrast to the legacy methods, the invention described in CN113292462B introduces a streamlined catalytic system that operates under remarkably mild conditions, typically between 20°C and 40°C. This novel approach utilizes a copper salt catalyst, specifically cuprous iodide, in conjunction with a mild organic base like diisopropylethylamine, effectively eliminating the need for cryogenic cooling or hazardous strong bases. The reaction proceeds with exceptional selectivity, favoring the formation of the desired allene thioether structure while suppressing the formation of polymeric side products and other impurities. This improvement in chemoselectivity translates directly into simplified workup procedures and higher isolated yields, often reaching up to 90% or higher for various substrates. The versatility of this method is further demonstrated by its tolerance to a wide range of functional groups, allowing for the synthesis of diverse derivatives suitable for different applications in medicinal chemistry.
Mechanistic Insights into Copper-Catalyzed Nucleophilic Addition
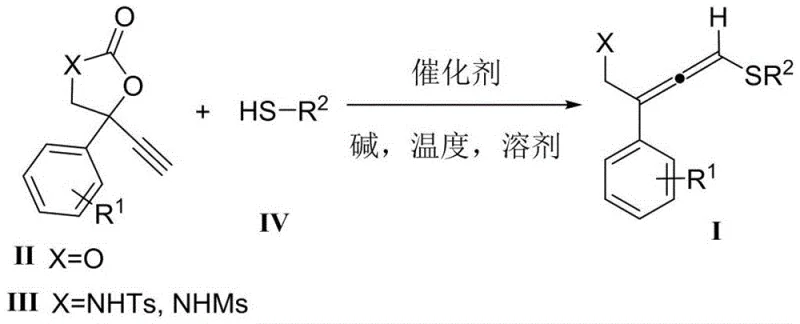
The success of this synthetic methodology lies in the precise orchestration of the copper catalytic cycle, which facilitates the nucleophilic attack of the thiol onto the activated alkyne species. The mechanism likely involves the coordination of the copper center to the alkyne moiety of the cyclic carbonate or carbamate precursor, thereby increasing its electrophilicity and rendering it susceptible to attack by the soft sulfur nucleophile. The choice of ligand and base plays a critical role in stabilizing the active catalytic species and promoting the subsequent ring-opening or rearrangement steps that lead to the allenic system. Experimental data indicates that cuprous iodide outperforms other copper salts such as copper acetate or copper triflate, suggesting that the specific electronic properties of the iodide ligand are crucial for optimal turnover. Additionally, the use of diisopropylethylamine as the base provides the necessary deprotonation of the thiol without inducing unwanted elimination reactions that could degrade the sensitive allene product. This delicate balance of reactivity ensures that the reaction proceeds cleanly to the target molecule with minimal formation of regioisomers or decomposition products.
From an impurity control perspective, the mild nature of this catalytic system is instrumental in maintaining a clean reaction profile. In traditional strong-base mediated reactions, the high basicity can lead to the deprotonation of the product itself or the starting material at multiple sites, leading to oligomerization or polymerization. By operating at near-neutral pH conditions facilitated by the organic base and copper catalyst, the new method effectively suppresses these degradation pathways. The result is a crude reaction mixture that is significantly cleaner, reducing the burden on purification columns and crystallization steps. This high level of purity is particularly vital for pharmaceutical intermediates, where strict limits on genotoxic impurities and heavy metals must be adhered to. The ability to achieve high selectivity without the need for exotic ligands or complex additive packages makes this process robust and reproducible, key attributes for any technology intended for commercial deployment.
How to Synthesize Substituted Allene Thioether Efficiently
The practical implementation of this synthesis route is designed to be straightforward and accessible for standard laboratory and pilot plant setups. The process begins with the dissolution of the catalyst, base, and alkyne precursor in a common organic solvent such as dichloromethane, followed by the controlled addition of the thiol component. The reaction is then allowed to proceed at a moderate temperature, typically around 40°C, for a duration sufficient to reach full conversion, usually around 15 hours. Detailed standardized synthesis steps for specific derivatives are provided in the structured guide below, which outlines the precise stoichiometry and workup procedures required to achieve optimal results. Adhering to these parameters ensures consistent quality and yield across different batches, facilitating the reliable production of these valuable building blocks.
- Prepare the reaction mixture by dissolving cuprous iodide catalyst (10 mol%), diisopropylethylamine base (1.0 equivalent), and 4-ethynyl-4-phenylcyclocarbonate substrate in dichloromethane solvent within a dry reaction vessel.
- Heat the solution to 40°C and slowly add benzyl mercaptan (2.0 equivalents) dropwise while maintaining stirring to ensure homogeneous mixing and controlled exotherm.
- Stir the reaction mixture for 15 hours at the specified temperature, then remove the solvent under reduced pressure and purify the crude residue via column chromatography to isolate the final white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel copper-catalyzed process offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of cryogenic requirements and hazardous strong bases fundamentally alters the cost structure of manufacturing these intermediates. By removing the need for specialized low-temperature reactors and the rigorous safety protocols associated with pyrophoric reagents, the capital expenditure and operational overhead are significantly reduced. Furthermore, the use of inexpensive and abundant copper salts as catalysts, as opposed to precious metals like palladium or platinum, contributes to a drastic reduction in raw material costs. This economic efficiency is compounded by the high yields and selectivity of the process, which minimize raw material waste and maximize the output per batch, thereby enhancing the overall cost-effectiveness of the supply chain.
- Cost Reduction in Manufacturing: The shift to mild reaction conditions eliminates the substantial energy costs associated with maintaining cryogenic temperatures, leading to significant operational savings. Additionally, the replacement of expensive and hazardous reagents with cost-effective copper catalysts and organic bases reduces the direct material cost per kilogram of product. The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that a greater proportion of input resources are converted into saleable inventory. These factors combined create a leaner manufacturing process that is highly resilient to fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents, such as dichloromethane and diisopropylethylamine, ensures a robust and uninterrupted supply of inputs. Unlike processes dependent on specialized organometallic reagents that may have long lead times or limited suppliers, this method utilizes commodity chemicals that are readily sourced from multiple vendors. This diversification of the supply base mitigates the risk of shortages and allows for greater flexibility in procurement planning. The simplicity of the process also reduces the likelihood of batch failures due to operational errors, further stabilizing the delivery schedule for downstream customers.
- Scalability and Environmental Compliance: The mild conditions and absence of hazardous waste streams make this process inherently safer and easier to scale from laboratory to industrial production. The reduced generation of polymeric byproducts and the use of recyclable solvents align with increasingly stringent environmental regulations, simplifying the permitting and compliance process for manufacturing facilities. This environmental compatibility not only reduces disposal costs but also enhances the sustainability profile of the final product, a key consideration for modern pharmaceutical companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these substituted allene thioether compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable basis for decision-making. Understanding these details is crucial for evaluating the fit of this technology within your specific development pipeline.
Q: What are the primary advantages of this copper-catalyzed method over traditional strong base methods?
A: Unlike traditional methods requiring hazardous strong bases like butyl lithium and cryogenic temperatures, this novel protocol operates under mild conditions (20-40°C) using inexpensive copper salts, significantly improving operational safety and reducing energy costs while minimizing side reactions such as polymerization.
Q: What is the typical yield and purity profile for these allene thioether intermediates?
A: The optimized process utilizing cuprous iodide and diisopropylethylamine consistently achieves high isolated yields, often exceeding 90% for model substrates, with excellent selectivity that avoids the formation of alkynyl sulfide byproducts common in less controlled synthetic routes.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the reaction utilizes commercially available solvents like dichloromethane and stable reagents, avoiding sensitive organometallic reagents, which makes the process highly amenable to kilogram-to-ton scale manufacturing for pharmaceutical and agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Allene Thioether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new drugs and advanced materials. Our team of expert chemists has thoroughly analyzed the potential of this copper-catalyzed synthesis route and is fully equipped to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions to optimize your manufacturing process.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient protocol. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to accelerate your R&D timeline and secure a sustainable supply chain for your critical allene thioether intermediates.
