Revolutionizing Pharmaceutical Intermediate Synthesis: Metal-Free Diphenyl Sulfide Production for Commercial Scale-Up
The groundbreaking patent CN113651738A introduces a novel metal-free methodology for synthesizing diphenyl sulfide compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach utilizes thiophenol and o-diiodobenzene as substrates, reacting them in a solvent system under the influence of metal hydride to produce iodine-containing diphenyl sulfides with exceptional efficiency. The process operates at room temperature without requiring transition metal catalysts, thereby eliminating the persistent challenges of metal residue contamination that have long plagued conventional synthetic routes. Crucially, the iodine-containing intermediates generated through this method serve as versatile building blocks for further molecular transformations, enabling the creation of complex structures essential for modern drug discovery and materials science applications. This patent addresses fundamental limitations in current manufacturing practices by providing a streamlined pathway that enhances both product purity and process sustainability while maintaining compatibility with large-scale production requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing thioether compounds have relied heavily on transition metal-catalyzed C-S coupling reactions, which present multiple critical challenges for pharmaceutical manufacturers. These methods typically employ rare and expensive metals such as palladium or copper, creating significant cost pressures while introducing persistent contamination risks that necessitate complex and costly purification steps to meet regulatory standards for pharmaceutical intermediates. The residual metal content often requires additional processing stages like chelation or extensive chromatography, substantially increasing production timelines and operational complexity. Furthermore, conventional techniques frequently demand harsh reaction conditions including strong alkalis, high temperatures, or hazardous oxidants, which compromise process safety and limit scalability. The formation of disubstituted byproducts during transition metal-catalyzed reactions with o-diiodobenzene creates additional purification hurdles, while alternative approaches using Grignard reagents or diazonium salts involve multi-step sequences with poor atom economy and safety concerns. These cumulative limitations have constrained the commercial viability of thioether-based pharmaceutical intermediates despite their significant therapeutic potential.
The Novel Approach
The patented methodology described in CN113651738A overcomes these limitations through an elegant metal-free coupling strategy that utilizes sodium hydride as the key reagent. This innovative process enables the direct reaction between thiophenol and o-diiodobenzene at ambient temperature (25°C) in a mixed THF/DMA solvent system, producing iodine-containing diphenyl sulfides as single products with high yields ranging from 82% to 93% across diverse substrates. The elimination of transition metals completely removes concerns about metallic impurities, significantly simplifying downstream purification and enhancing product purity to meet stringent pharmaceutical requirements. The room-temperature operation not only improves energy efficiency but also enhances process safety by avoiding high-pressure or high-temperature conditions. Critically, the iodine functionality in the resulting intermediates serves as a strategic handle for subsequent transformations, allowing manufacturers to access a wide range of complex structures through well-established Pd-catalyzed couplings without requiring additional synthetic development. This approach demonstrates superior atom economy and operational simplicity compared to conventional methods, making it particularly suitable for commercial-scale production of high-value pharmaceutical intermediates.
Mechanistic Insights into NaH-Mediated C-S Coupling
The core innovation lies in the sodium hydride-mediated deprotonation mechanism that facilitates direct C-S bond formation without transition metal involvement. In this process, NaH first deprotonates the thiophenol substrate to generate a thiolate anion, which then undergoes nucleophilic aromatic substitution with o-diiodobenzene through an addition-elimination pathway. The ortho-positioned iodine atoms in the benzene ring activate the system toward nucleophilic attack while simultaneously serving as excellent leaving groups. This dual functionality enables the reaction to proceed efficiently at room temperature without requiring additional catalysts or harsh conditions. The solvent system comprising THF and DMA in a 5:1 ratio creates an optimal polarity environment that stabilizes the reactive intermediates while facilitating smooth progression through the transition state. The mechanism avoids radical pathways or unstable intermediates that could lead to side products, explaining the consistent formation of single products across various substituted thiophenols as demonstrated in the patent examples. This mechanistic understanding provides manufacturers with precise control parameters for optimizing yield and purity in commercial production settings.
Impurity control is inherently engineered into this synthetic pathway through multiple self-regulating mechanisms. The absence of transition metals eliminates the primary source of metallic contaminants that typically require extensive post-reaction purification. The room-temperature conditions prevent thermal degradation pathways that could generate byproducts in conventional high-temperature processes. The selective nucleophilic substitution mechanism ensures that only mono-substituted products form, avoiding the disubstituted impurities commonly observed in transition metal-catalyzed reactions with dihalogenated substrates. The patent demonstrates consistent product purity across diverse substrates (as evidenced by NMR characterization data), with no detectable metal residues or significant side products requiring additional purification steps. This inherent selectivity translates directly to higher quality intermediates that meet pharmaceutical industry standards without costly remediation processes, significantly reducing quality control burdens during scale-up.

How to Synthesize Diphenyl Sulfide Compounds Efficiently
This patented methodology provides a robust framework for manufacturing high-purity diphenyl sulfide compounds essential for advanced pharmaceutical applications. The process begins with careful preparation of anhydrous reaction conditions under nitrogen atmosphere to ensure optimal performance of the sodium hydride reagent. Manufacturers should note that precise stoichiometric control (thiophenol:NaH:o-diiodobenzene at 1:3:2 molar ratio) is critical for achieving maximum yield and purity as demonstrated in the patent examples. The solvent mixture composition (THF/DMA at 5:1 v/v) creates the ideal polarity environment for intermediate stabilization while maintaining sufficient solubility for all components. Reaction monitoring via TLC provides real-time progress assessment, with completion typically achieved within 1-10 hours depending on substrate substitution patterns. The subsequent workup procedure involving ice water quenching followed by ethyl acetate extraction and silica gel chromatography delivers consistently pure products ready for further transformation or direct use in pharmaceutical synthesis. Detailed standardized synthesis steps are provided below to ensure seamless implementation across manufacturing facilities.
- Prepare reaction mixture with thiophenol and o-diiodobenzene in THF/DMA solvent system under nitrogen atmosphere
- Add sodium hydride (3.0 equiv) at room temperature and monitor reaction progression via TLC
- Purify iodine-containing diphenyl sulfide intermediate through ethyl acetate extraction and silica gel chromatography
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate production. The elimination of transition metal catalysts removes a major cost driver associated with expensive palladium or copper complexes while simultaneously eliminating downstream processing requirements for metal removal. The ambient temperature operation reduces energy consumption and equipment demands compared to conventional high-temperature processes, creating significant operational efficiencies that translate directly to cost savings without requiring capital investment in specialized reactors. The use of commercially available starting materials with straightforward handling characteristics enhances supply chain resilience by reducing dependency on specialized or restricted reagents. Most importantly, the inherent scalability of this room-temperature process enables seamless transition from laboratory development to commercial production volumes while maintaining consistent product quality.
- Cost Reduction in Manufacturing: The complete removal of transition metal catalysts eliminates both the raw material cost of expensive palladium complexes and the substantial processing expenses associated with metal residue removal. Without requiring specialized equipment for high-temperature operations or complex purification systems for metal contamination, manufacturers achieve significant cost savings through simplified process design and reduced operational complexity. The elimination of multiple purification steps required in conventional methods further contributes to economic efficiency while maintaining high product quality standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilizing readily available commercial reagents including sodium hydride and standard solvents creates greater supply chain flexibility compared to processes dependent on scarce transition metals or specialized catalysts. The room-temperature operation reduces equipment failure risks associated with high-pressure or high-temperature systems, minimizing production interruptions. The consistent performance across diverse substrates demonstrated in the patent ensures reliable output quality regardless of minor variations in starting material specifications, providing procurement teams with greater confidence in supply continuity and quality assurance.
- Scalability and Environmental Compliance: The ambient temperature process enables straightforward scale-up from laboratory to commercial production without requiring specialized equipment modifications or safety protocols associated with high-energy processes. The simplified reaction profile generates fewer byproducts and eliminates toxic metal waste streams, significantly reducing environmental impact and waste treatment costs. This green chemistry approach aligns with increasingly stringent environmental regulations while providing manufacturers with a sustainable production pathway that maintains regulatory compliance across global markets.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113651738A's methodology and implementation data. These insights reflect practical experience from scaling similar metal-free processes in pharmaceutical intermediate manufacturing and provide actionable guidance for procurement and R&D teams evaluating this technology for commercial adoption.
Q: How does this metal-free process improve impurity profiles compared to traditional transition metal catalysis?
A: The elimination of transition metal catalysts prevents heavy metal residues that complicate purification. Patent CN113651738A demonstrates that the NaH-mediated coupling produces single-product iodine-containing diphenyl sulfides without disubstituted byproducts, significantly enhancing purity for pharmaceutical applications where metal contaminants require costly removal processes.
Q: What scalability advantages does the room-temperature reaction condition provide for commercial manufacturing?
A: Operating at ambient temperature (25°C) eliminates energy-intensive heating/cooling cycles required in conventional methods. The patent shows consistent yields (82-93%) across diverse substrates without specialized equipment, enabling seamless scale-up from laboratory to industrial production while maintaining stringent quality control standards.
Q: How does the iodine-containing intermediate enable flexible route design for complex pharmaceuticals?
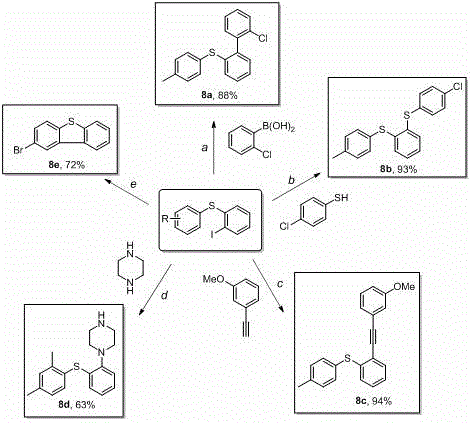
A: The ortho-iodine functionality serves as a versatile handle for Pd-catalyzed transformations. As documented in the patent, this intermediate undergoes Suzuki, Sonogashira, and Buchwald-Hartwig couplings to generate diverse structures including vortioxetine precursors, providing manufacturers with adaptable pathways for high-value pharmaceutical intermediates without redeveloping synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Sulfide Compound Supplier
Our company leverages this patented technology to deliver exceptional value in complex intermediate manufacturing, combining deep expertise in metal-free synthesis with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through advanced analytical capabilities in our rigorous QC labs, ensuring consistent product quality that meets global pharmaceutical standards. Our technical team specializes in adapting this innovative methodology to specific client requirements while optimizing cost structures through continuous process refinement. As a trusted partner in pharmaceutical intermediate supply, we provide comprehensive support from route validation through full-scale manufacturing, ensuring seamless integration of this technology into your production ecosystem.
Request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this metal-free synthesis can optimize your specific manufacturing pathway. We will provide detailed route feasibility assessments along with specific COA data demonstrating purity profiles and scalability metrics tailored to your production requirements.
