Industrial Scale Synthesis of Chiral Ferrocene Bisphosphine Ligands via Chromatography-Free Routes
The development of efficient synthetic routes for chiral ligands is a cornerstone of modern asymmetric catalysis, particularly for the pharmaceutical industry where enantiomeric purity is paramount. Patent CN101570550A introduces a robust methodology for the synthesis of chiral ferrocene bisphosphine ligands, addressing critical bottlenecks in traditional manufacturing. Unlike conventional laboratory procedures that depend heavily on tedious column chromatography for purification, this invention leverages a sequence of acetylation, reduction, esterification, and amination that can be performed with minimal intermediate isolation. The process begins with the Friedel-Crafts acylation of ferrocene, a ubiquitous and cost-effective starting material, to yield acetylferrocene. This intermediate is subsequently reduced and esterified to form acetoxyethyl ferrocene, which serves as the precursor for the introduction of the chiral amine functionality. The strategic design of this pathway ensures that high-purity intermediates are obtained through simple work-up procedures such as washing, distillation, and crystallization, rather than expensive chromatographic separation.

This technological advancement is particularly significant for the production of high-purity pharmaceutical intermediates where trace metal contamination from silica gel or excessive solvent usage must be minimized. The patent outlines a comprehensive approach where the ferrocene backbone is functionalized step-by-step, maintaining the integrity of the organometallic structure while introducing the necessary steric and electronic properties required for effective catalysis. By optimizing reaction conditions such as temperature ranges between 0°C and 100°C and utilizing standard Lewis acid catalysts, the method ensures reproducibility and safety. The elimination of chromatographic steps not only reduces the environmental footprint but also drastically shortens the production cycle time, making it an attractive option for reliable agrochemical intermediate and pharmaceutical suppliers seeking to optimize their manufacturing overheads.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral ferrocenyl diphosphine ligands has been plagued by operational complexities that hinder large-scale adoption. Prior art, such as the methods reported in literature like Bull. Chem. Soc. Jpn., typically involves multi-step syntheses where each intermediate requires purification via column chromatography. This reliance on chromatography presents severe limitations for industrial application, including high consumption of silica gel and organic solvents, which escalates both material costs and waste disposal expenses. Furthermore, chromatographic separation is inherently batch-limited and difficult to automate, creating bottlenecks in supply chain continuity. The exposure of sensitive organometallic intermediates to silica surfaces can also lead to decomposition or lower yields, compromising the overall efficiency of the process. For procurement managers, these factors translate into volatile pricing and extended lead times, as the purification stage often becomes the rate-limiting step in the entire manufacturing workflow.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by engineering a synthesis pathway that relies on phase separation and crystallization for purification. By converting the ferrocene derivative into an acetoxy intermediate and subsequently displacing the acetate group with dimethylamine, the process generates a tertiary amine that is amenable to chiral resolution via salt formation. This strategy allows for the isolation of optically pure intermediates using chiral acids like tartaric acid or camphorsulfonic acid, followed by simple filtration and recrystallization. The subsequent lithiation and phosphination steps are conducted under controlled inert atmospheres, ensuring high regioselectivity without the need for further chromatographic cleanup. This streamlined workflow significantly enhances the commercial scale-up of complex polymer additives and fine chemicals by reducing the number of unit operations. The ability to perform multiple reaction steps in a single reactor or with simple solvent swaps demonstrates a profound understanding of process chemistry, offering a distinct competitive advantage in cost reduction in electronic chemical manufacturing and related high-value sectors.
Mechanistic Insights into Ferrocene Functionalization and Chiral Resolution
The core of this synthesis lies in the precise manipulation of the ferrocene electronic system to achieve regioselective functionalization. The initial acetylation activates one cyclopentadienyl ring, directing subsequent reductions and substitutions to the alpha-position. The conversion of the ketone to an alcohol and then to an ester creates a good leaving group for the nucleophilic attack by dimethylamine. This amination step is crucial as it installs the directing group necessary for the later ortho-lithiation. The mechanism relies on the coordination of the nitrogen lone pair to the lithium cation, which directs the deprotonation to the adjacent position on the ferrocene ring. This directed ortho-metalation (DoM) is a powerful tool in organometallic chemistry, allowing for the installation of the first phosphine group with high stereochemical fidelity. The use of strong organolithium bases at low temperatures (-15°C to -25°C) ensures that the lithiation occurs exclusively at the desired position, preventing side reactions that could compromise the ligand's performance in asymmetric hydrogenation.
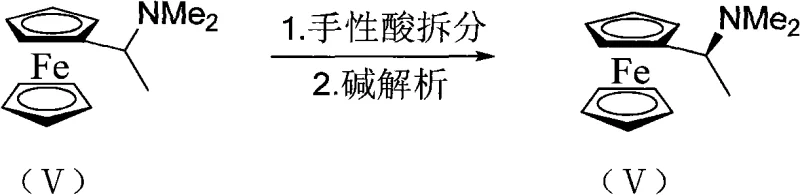
Following the installation of the first phosphine moiety, the chiral integrity of the molecule is preserved and enhanced through the final substitution reaction. The displacement of the dimethylamino group by a second phosphine species proceeds via a mechanism that retains the configuration at the chiral center, driven by the steric bulk of the ferrocene backbone. The patent specifies the use of various dialkyl or diaryl phosphines, allowing for the tuning of the ligand's steric and electronic properties to suit specific catalytic applications. Impurity control is managed through the rigorous purification of the chiral amine salt prior to lithiation; any racemic material remaining at this stage would lead to a mixture of diastereomers in the final product, which are difficult to separate. By ensuring high optical purity early in the sequence through recrystallization of the chiral salt, the process guarantees that the final bisphosphine ligand meets the stringent purity specifications required for catalytic applications in the synthesis of active pharmaceutical ingredients.
How to Synthesize Chiral Ferrocene Bisphosphine Ligand Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable ligands with high efficiency and minimal waste. The process integrates several classical organic transformations into a cohesive flow that maximizes yield while minimizing handling. Operators begin by preparing the acetylated ferrocene derivative, which is then reduced and esterified in situ or with minimal workup. The subsequent amination with dimethylamine yields the racemic amine, which is the pivotal branch point for introducing chirality. Detailed standardized synthesis steps see the guide below for specific molar ratios and temperature controls that ensure optimal outcomes.
- Perform Friedel-Crafts acetylation of ferrocene followed by reduction and esterification to generate acetoxyethyl ferrocene without intermediate purification.
- Execute nucleophilic substitution with dimethylamine to form the racemic amine, followed by chiral acid resolution to isolate the optically pure R-enantiomer.
- Conduct directed ortho-lithiation using organolithium bases, followed by sequential reaction with chlorophosphines to install the two phosphine groups with high stereocontrol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain professionals, the adoption of this chromatography-free synthesis route offers transformative benefits in terms of cost structure and operational reliability. The elimination of silica gel and the associated large volumes of elution solvents directly translates to substantial cost savings in raw material procurement and waste management. Traditional methods often incur hidden costs related to the disposal of hazardous chromatographic waste, which this new method effectively mitigates by relying on aqueous workups and crystallization. Furthermore, the use of ferrocene as a starting material leverages a widely available and inexpensive commodity chemical, insulating the production cost from the volatility associated with exotic precursors. This stability in raw material sourcing is critical for maintaining consistent pricing models in long-term supply contracts.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of column chromatography, which is notoriously resource-intensive. By replacing this with crystallization and distillation, manufacturers can significantly reduce the consumption of high-purity solvents and stationary phases. This simplification of the purification train lowers the capital expenditure required for equipment, as large chromatography columns and solvent recovery systems are no longer the bottleneck. Additionally, the higher throughput achievable with batch crystallization compared to column loading allows for better utilization of reactor volume, effectively increasing plant capacity without additional infrastructure investment. These efficiencies compound to deliver a more competitive cost base for high-value chiral ligands.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route enhances supply chain resilience by reducing the number of critical failure points. Chromatography is often a source of variability due to column packing inconsistencies or solvent quality issues; removing this step stabilizes the production timeline. The reagents used, such as acetyl chloride, borohydrides, and common chiral acids, are commercially available in bulk quantities from multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base ensures that production schedules can be maintained even if one vendor faces disruptions. Consequently, lead times for high-purity pharmaceutical intermediates become more predictable, allowing downstream customers to plan their inventory levels with greater confidence.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns well with green chemistry principles by minimizing solvent waste and avoiding solid waste generation from silica gel. The process operates under relatively mild conditions, reducing energy consumption for heating and cooling compared to more extreme synthetic protocols. The ability to recycle solvents from distillation steps further enhances the sustainability profile of the manufacturing process. For facilities operating under strict environmental regulations, this reduced waste load simplifies compliance and lowers the cost of environmental permits. The scalability is proven by the patent's explicit mention of industrial applicability, indicating that the chemistry holds up when moving from gram-scale laboratory experiments to kilogram or ton-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity on how this method compares to existing standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: How does this synthesis method improve upon traditional laboratory routes for ferrocene ligands?
A: Traditional methods often rely on multiple column chromatography separations which are costly and difficult to scale. This patent describes a route that eliminates column chromatography entirely, utilizing crystallization and distillation for purification, making it highly suitable for industrial production.
Q: What represents the key step for establishing chirality in this ligand synthesis?
A: Chirality is established through the resolution of the racemic N,N-dimethylaminoethyl ferrocene intermediate using chiral acids such as D-tartaric acid or D-camphorsulfonic acid. This forms a diastereomeric salt that can be purified via recrystallization to achieve high optical purity before the final phosphination steps.
Q: Is this process compatible with large-scale manufacturing of asymmetric hydrogenation catalysts?
A: Yes, the process is explicitly designed for industrialization. It uses inexpensive raw materials like ferrocene and avoids complex purification techniques. The reactions operate under standard conditions (0-100°C) and utilize common solvents, ensuring robust scalability for commercial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation pharmaceuticals and fine chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our facility is equipped to handle the sensitive organometallic chemistry required for ferrocene derivatives, maintaining the inert atmospheres and precise temperature controls necessary for success.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific applications. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this chromatography-free method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to enhance your supply chain efficiency and drive innovation in asymmetric catalysis together.
