Advanced Synthesis of N,N-di-N-propyl-2-propoxyethylamine for High-Efficiency Agrochemical Production
Advanced Synthesis of N,N-di-N-propyl-2-propoxyethylamine for High-Efficiency Agrochemical Production
The development of efficient synthetic routes for agrochemical intermediates remains a critical priority for modern chemical manufacturing, particularly for high-volume herbicides like pretilachlor. A groundbreaking approach detailed in patent CN111100019A introduces a novel auxiliary agent, N,N-di-N-propyl-2-propoxyethylamine, which addresses longstanding challenges in reaction kinetics and product selectivity. This technical insight report analyzes the preparation and application of this specialized amine, highlighting its potential to revolutionize the production of 2,6-diethyl-N-(2-propoxyethyl)aniline. By leveraging a straightforward thermal nucleophilic substitution strategy, manufacturers can overcome the limitations of traditional acid-binding agents, paving the way for more robust and reliable agrochemical intermediate supplier networks globally.
The core innovation lies in the specific molecular architecture of the auxiliary, which optimizes the physical properties of the reaction medium. Unlike conventional methods that struggle with viscosity and phase transfer issues, this new compound ensures superior system fluidity. For R&D directors and process engineers, understanding the mechanistic advantages of this additive is essential for implementing cost reduction in agrochemical manufacturing. The following sections provide a deep dive into the synthetic pathway, the comparative advantages over legacy technologies, and the commercial implications for supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key pretilachlor intermediates via N-alkylation of 2,6-diethylaniline has been plagued by significant operational inefficiencies. Traditional protocols often rely on inorganic bases such as sodium hydroxide or metal oxides like magnesium oxide to act as acid scavengers. While chemically feasible, these solid or heterogeneous systems frequently result in poor reaction mixture fluidity, leading to inadequate heat dissipation and uneven mixing. Patent literature indicates that under these conventional conditions, the selectivity for the desired mono-substituted product is often compromised, with reported contents in the feed liquid reaching a maximum of only 63%. Furthermore, the presence of solid residues complicates downstream processing, requiring extensive filtration and washing steps that increase waste generation and reduce overall throughput.
The Novel Approach
In stark contrast, the introduction of N,N-di-N-propyl-2-propoxyethylamine as a liquid auxiliary agent fundamentally alters the reaction environment. This organic additive acts not merely as a base but as a solvating medium that maintains low viscosity even at elevated temperatures. The patent data demonstrates that when this auxiliary is employed, the reaction system exhibits excellent fluidity, allowing for precise temperature control and homogeneous reagent distribution. Consequently, the progression of undesirable di-substitution side reactions is effectively inhibited. This strategic shift from heterogeneous solid bases to a tailored liquid amine auxiliary results in a dramatic improvement in target product selectivity, with experimental data showing values exceeding 95% in optimized runs. This transition represents a significant leap forward for any entity seeking a reliable agrochemical intermediate supplier capable of delivering high-purity materials.
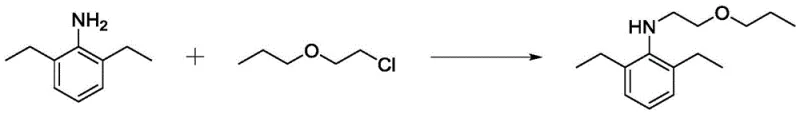
Mechanistic Insights into Nucleophilic Substitution and Fluidity Enhancement
The synthesis of the auxiliary agent itself relies on a classic yet highly optimized nucleophilic substitution mechanism. The process involves the reaction of chloroethyl propyl ether with di-n-propylamine under thermal conditions. The nitrogen atom of the secondary amine acts as a nucleophile, attacking the electrophilic carbon adjacent to the chlorine leaving group on the ether chain. This SN2-type displacement is driven by the elevated reaction temperatures ranging from 100°C to 180°C, which provide the necessary activation energy to overcome steric hindrance. The choice of reactants is crucial; the propyl groups on the amine provide a balance of nucleophilicity and steric bulk that prevents over-alkylation of the auxiliary itself, ensuring the final product retains the specific structural motif required for its function.
Regarding impurity control, the mechanism inherently favors the formation of the tertiary amine structure due to the stoichiometry and reaction conditions employed. By carefully controlling the molar ratio of di-n-propylamine to chloroethyl propyl ether between 1:1 and 4:1, the process minimizes the formation of quaternary ammonium salts or polymeric byproducts. Post-reaction workup involves a simple pH adjustment to 8-10 using an aqueous alkali solution, which neutralizes any unreacted amine hydrochloride salts formed during the process. This allows for a clean phase separation where the organic layer containing the high-purity auxiliary can be isolated. Subsequent vacuum distillation further refines the product, removing trace volatiles and ensuring a GC purity of 99%, which is vital for preventing the introduction of new impurities into the downstream herbicide synthesis.
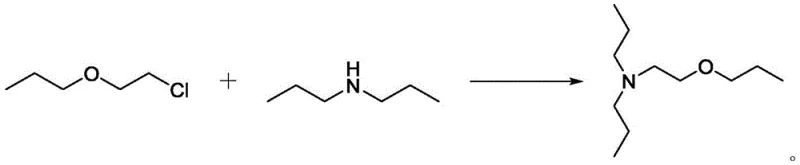
How to Synthesize N,N-di-N-propyl-2-propoxyethylamine Efficiently
The preparation of this high-value auxiliary is designed for operational simplicity and scalability, making it an attractive candidate for commercial adoption. The process utilizes readily available commodity chemicals, eliminating the need for exotic catalysts or complex multi-step sequences. The reaction is conducted in a standard stirred tank reactor, where thermal energy drives the conversion. Following the reaction, the workup procedure leverages basic liquid-liquid extraction principles, avoiding the need for chromatographic purification which is cost-prohibitive at scale. The detailed standardized synthesis steps, including specific temperature ramps and distillation cuts, are outlined in the technical guide below to ensure reproducibility across different manufacturing sites.
- Heat chloroethyl propyl ether to a temperature range of 100-180°C in a reaction vessel equipped with stirring capabilities.
- Add di-n-propylamine dropwise while maintaining the temperature, ensuring a molar ratio between 1: 1 and 4:1 relative to the ether.
- Maintain the reaction mixture for 8 to 20 hours, then cool, adjust pH to 8-10 with alkali, separate the organic layer, and purify via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel auxiliary offers compelling economic and logistical benefits. The shift away from solid metal oxide bases simplifies the material handling requirements, reducing the risk of dust exposure and equipment abrasion. Moreover, the liquid nature of the auxiliary facilitates automated dosing systems, enhancing process safety and consistency. From a cost perspective, the elimination of solid waste filtration and the reduction in solvent usage for washing steps contribute to substantial cost savings in agrochemical manufacturing. The ability to recover and recycle unreacted starting materials through distillation further enhances the atom economy of the process, aligning with modern green chemistry principles and reducing raw material expenditure.
- Cost Reduction in Manufacturing: The implementation of this liquid auxiliary system eliminates the need for expensive and difficult-to-handle solid acid binders like magnesium oxide. By streamlining the workup procedure to a simple phase separation and distillation, manufacturers can significantly reduce labor costs and energy consumption associated with filtration and drying operations. The high selectivity achieved minimizes the formation of byproducts, thereby reducing the load on purification columns and extending the lifecycle of catalysts or packing materials used in downstream processing. These cumulative efficiencies translate into a lower cost of goods sold (COGS) without compromising on the quality of the final agrochemical active ingredient.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically chloroethyl propyl ether and di-n-propylamine, are established commodity chemicals with robust global supply chains. This reduces the risk of supply disruptions that are often associated with specialized reagents or imported catalysts. Furthermore, the process tolerance allows for flexibility in sourcing, as the reaction conditions are not overly sensitive to minor variations in feedstock quality. This resilience ensures a consistent supply of the auxiliary agent, which in turn stabilizes the production schedule for the critical pretilachlor intermediate, safeguarding against market volatility and delivery delays.
- Scalability and Environmental Compliance: The process has been successfully demonstrated at a 5-liter scale with excellent results, indicating a clear path for commercial scale-up to multi-ton production. The absence of heavy metal catalysts or toxic reagents simplifies wastewater treatment and waste disposal compliance. The primary waste streams are aqueous salt solutions and organic distillation residues, both of which are manageable within standard industrial waste treatment facilities. This environmental profile supports sustainable manufacturing goals and reduces the regulatory burden on production sites, facilitating faster permitting and long-term operational continuity for high-purity agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. They serve to clarify the operational parameters and expected outcomes for teams evaluating this route for potential licensing or procurement.
Q: How does this auxiliary agent improve the synthesis of pretilachlor intermediates?
A: The addition of N,N-di-N-propyl-2-propoxyethylamine significantly enhances the fluidity of the reaction system during the N-alkylation of 2,6-diethylaniline. This improved rheology facilitates better heat and mass transfer, which inhibits di-substitution side reactions and boosts the selectivity of the target mono-substituted product to over 95%.
Q: What are the purity specifications achievable with this method?
A: Through optimized vacuum distillation protocols described in the patent, the auxiliary agent can be obtained with a GC purity of 99%. This high level of purity is critical for preventing downstream contamination in sensitive agrochemical manufacturing processes.
Q: Is this process scalable for industrial production?
A: Yes, the methodology has been successfully demonstrated at a 5-liter scale, achieving a yield of 94.9% for the target intermediate. The use of standard unit operations like distillation and liquid-liquid separation ensures that the process is readily adaptable for commercial scale-up without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-di-N-propyl-2-propoxyethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic route for the agrochemical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this new auxiliary is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N,N-di-N-propyl-2-propoxyethylamine meets the highest industry standards. We are committed to supporting your R&D and production teams with the technical expertise needed to optimize this process for your specific manufacturing environment.
We invite you to explore how this innovation can drive value in your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us collaborate to enhance the efficiency and sustainability of your agrochemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
