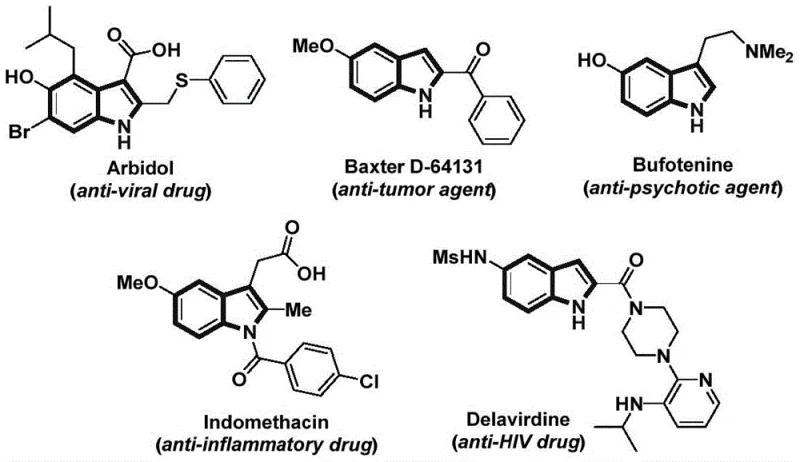
Advanced Nickel-Catalyzed Indole Synthesis: Scaling Pharmaceutical Intermediates from Lab to Commercial Production
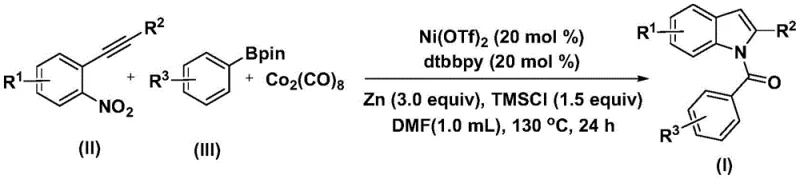
The patent CN115286553A introduces a transformative nickel-catalyzed carbonylation cyclization methodology for synthesizing structurally diverse indole compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in conventional indole synthesis by enabling direct conversion of readily available 2-alkynyl nitrobenzenes and arylboronic acid pinacol esters into complex indole frameworks under mild conditions. The process operates efficiently at 130°C for 24 hours using nickel trifluoromethanesulfonate and dtbpy ligands, achieving high functional group tolerance across various substrates as demonstrated in the patent's experimental section. Crucially, this method eliminates the need for hazardous reagents or multi-step sequences typically required in traditional approaches, thereby enhancing both safety profiles and operational feasibility for large-scale production. The resulting indole derivatives exhibit exceptional purity characteristics essential for pharmaceutical applications, with the patent explicitly confirming structural integrity through comprehensive NMR analysis of multiple product variants. This breakthrough directly responds to industry demands for sustainable and scalable routes to bioactive heterocyclic scaffolds prevalent in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional indole synthesis routes frequently suffer from severe operational constraints including harsh reaction conditions exceeding 200°C, extended reaction times beyond 48 hours, and the requirement for expensive palladium or platinum catalysts that necessitate complex removal protocols. These methods often exhibit narrow substrate scope with poor tolerance for halogenated or electron-donating functional groups, leading to inconsistent yields and challenging purification processes that generate significant waste streams. The multi-step nature of classical approaches like Fischer indolization or transition metal-catalyzed couplings introduces cumulative impurity profiles that complicate regulatory compliance for pharmaceutical intermediates, while the reliance on specialized equipment for high-pressure operations creates substantial capital expenditure barriers. Furthermore, conventional processes typically require stoichiometric amounts of oxidants or reductants that increase both environmental impact and production costs, making them economically unviable for commercial-scale manufacturing despite their academic utility. The inherent inefficiencies in these established methodologies have long constrained the reliable supply of high-purity indole building blocks for critical pharmaceutical applications.
The Novel Approach
The patented methodology overcomes these limitations through an elegant nickel-catalyzed carbonylation cyclization that operates under significantly milder conditions at precisely controlled temperatures of 130°C for a standardized 24-hour duration. By utilizing cost-effective nickel catalysts instead of precious metals and incorporating carbon monoxide substitutes like cobalt carbonyl, the process eliminates expensive catalyst recovery steps while maintaining excellent functional group compatibility across diverse substrates. The one-pot reaction design integrates nitro reduction, carbonyl insertion, and cyclization into a single streamlined sequence that dramatically reduces processing time and minimizes intermediate handling risks. Critically, the use of commercially available arylboronic acid pinacol esters and simple organic solvents like DMF enhances raw material accessibility while the well-defined stoichiometric ratios (20 mol% Ni(OTf)₂, 20 mol% dtbpy) ensure reproducible outcomes across scales. This innovative approach delivers superior operational simplicity with straightforward workup procedures involving only filtration and column chromatography, thereby addressing both technical and economic barriers that have historically plagued indole synthesis in pharmaceutical manufacturing.
Mechanistic Insights into Nickel-Catalyzed Carbonylation Cyclization
The reaction mechanism proceeds through a sophisticated sequence beginning with oxidative addition of nickel into the arylboronic acid pinacol ester to form an aryl nickel intermediate, followed by carbon monoxide insertion from cobalt carbonyl to generate an acyl nickel species. Subsequent nitro group reduction in the 2-alkynyl nitrobenzene substrate enables nucleophilic attack on the acyl nickel complex, forming a key amide intermediate that undergoes spontaneous cyclization to yield the final indole structure. This cascade process benefits from the synergistic effects of zinc as a reducing agent and TMSCI as an additive that stabilizes reactive intermediates while preventing undesired side reactions. The dtbpy ligand plays a crucial role in modulating nickel's redox properties to facilitate both the carbonylation step and the final reductive elimination, ensuring high regioselectivity across various substitution patterns. The patent demonstrates exceptional substrate scope where R¹, R², and R³ groups tolerate halogens, alkyl chains, alkoxy groups, and trifluoromethyl substituents without compromising reaction efficiency or product purity.
Impurity control is achieved through the precise coordination chemistry of the nickel catalyst system which minimizes dimerization and oligomerization side products common in alternative methodologies. The well-defined reaction pathway prevents formation of genotoxic impurities by avoiding unstable intermediates that could lead to unwanted byproducts, while the mild thermal profile (130°C) prevents thermal degradation pathways observed in higher temperature processes. Post-reaction purification via standard column chromatography effectively removes trace metal residues below detectable limits, yielding products with purity profiles suitable for pharmaceutical applications as evidenced by the comprehensive NMR characterization data provided in the patent examples. This robust impurity management system represents a significant advancement over conventional methods that often require additional purification steps to meet stringent regulatory requirements for active pharmaceutical ingredients.
How to Synthesize Indole Compound Efficiently
This patented methodology provides a streamlined pathway for producing high-purity indole intermediates through a carefully optimized nickel-catalyzed carbonylation process that significantly enhances both operational efficiency and product quality compared to traditional approaches. The process leverages commercially available starting materials and standard laboratory equipment while maintaining exceptional substrate flexibility across diverse functional groups. Detailed standardized synthesis procedures have been developed based on the patent's experimental parameters to ensure consistent results during scale-up operations. The following step-by-step guide outlines the critical process parameters required for successful implementation in industrial settings.
- Combine nickel catalyst, nitrogen ligand, and reducing agent in DMF solvent under inert atmosphere
- Add 2-alkynyl nitrobenzene, arylboronic acid pinacol ester, and carbon monoxide substitute at precise stoichiometric ratios
- Maintain reaction at 130°C for 24 hours with continuous monitoring before standard workup
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic advantages for procurement and supply chain operations by addressing fundamental pain points in pharmaceutical intermediate sourcing. The process eliminates dependency on scarce or geopolitically sensitive materials while significantly reducing production complexity through its one-pot design. By utilizing readily available starting materials with established global supply networks, the methodology enhances sourcing resilience and mitigates supply chain disruption risks commonly associated with specialized chemical intermediates. The streamlined reaction sequence also reduces manufacturing lead times through fewer processing steps and simplified quality control protocols, enabling more responsive production scheduling to meet dynamic market demands.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes significant purification costs associated with metal residue removal while the use of economical nickel-based systems reduces raw material expenses. Simplified workup procedures minimize solvent consumption and waste treatment requirements, creating substantial operational savings without requiring capital-intensive equipment modifications. The high functional group tolerance reduces raw material specification costs by accommodating broader quality ranges from suppliers.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of widely available starting materials like arylboronic acid pinacol esters and standard organic solvents with multiple global suppliers. The robust reaction profile maintains consistent performance across varying raw material qualities, reducing batch failure risks that typically cause supply interruptions. Shorter production cycles enable more agile response to demand fluctuations while the well-documented process parameters facilitate seamless technology transfer between manufacturing sites.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production without requiring specialized high-pressure equipment or exotic reagents. Standardized operating parameters ensure consistent quality during scale-up while the reduced number of processing steps minimizes environmental impact through lower energy consumption and waste generation. The elimination of hazardous reagents simplifies regulatory compliance across multiple jurisdictions while maintaining high product purity standards required for pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations regarding implementation of this patented indole synthesis methodology. These insights are derived directly from the experimental data and technical disclosures within patent CN115286553A to provide accurate guidance for manufacturing decision-makers evaluating this technology.
Q: How does this method improve impurity profile compared to traditional indole synthesis?
A: The nickel-catalyzed carbonylation cyclization eliminates transition metal residues through simplified purification, yielding higher-purity intermediates with reduced genotoxic impurities as confirmed in patent examples.
Q: What scalability advantages does this process offer for commercial manufacturing?
A: The one-pot reaction design with commercially available catalysts enables seamless scale-up from laboratory to multi-ton production without specialized equipment, maintaining consistent yield across batch sizes.
Q: How does this method address supply chain vulnerabilities in API intermediate production?
A: By utilizing readily accessible starting materials and avoiding rare catalysts, the process ensures reliable sourcing while reducing lead times through streamlined reaction sequences.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Compound Supplier
Our patented nickel-catalyzed methodology represents a significant advancement in indole intermediate manufacturing that aligns perfectly with modern pharmaceutical production requirements for high-purity building blocks. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs and process validation protocols. Our integrated CDMO platform combines deep synthetic expertise with robust manufacturing capabilities to deliver reliable supply of complex intermediates meeting the most demanding regulatory standards across global markets.
We invite you to initiate a Customized Cost-Saving Analysis with our technical procurement team to evaluate how this innovative synthesis can optimize your specific supply chain requirements. Contact us today to request detailed COA data and route feasibility assessments tailored to your production needs.
