Advanced One-Pot Synthesis of Nitrogen-Doped Ovalene-2N for Commercial OLED Production
The landscape of organic optoelectronic materials is undergoing a significant transformation, driven by the urgent need for materials that combine high charge mobility with enhanced environmental stability. Patent CN113666928A introduces a groundbreaking preparation method for a novel nitrogen-doped polycyclic aromatic hydrocarbon, specifically Ovalene-2N, which features nitrogen atoms doped on serrated edges. This structural modification addresses the inherent limitations of traditional all-carbon graphene segments, which often suffer from poor solubility and uncontrollable electronic characteristics. By integrating nitrogen atoms into the six-membered zigzag edge fused ring system, the invention creates a conjugated molecular skeleton that offers superior assembly performance and stability. For R&D Directors and Procurement Managers seeking a reliable OLED material supplier, this technology represents a pivotal shift towards more robust and tunable organic semiconductors that can be manufactured with greater efficiency and lower environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for large polycyclic aromatic hydrocarbons often involve multi-step processes that are fraught with inefficiencies and high production costs. Conventional methods typically rely on harsh reaction conditions, including high temperatures and the use of expensive transition metal catalysts, which complicate the purification process and introduce potential heavy metal contaminants. These impurities are detrimental to the performance of organic electronic devices, necessitating rigorous and costly post-synthesis cleaning procedures. Furthermore, the solubility of all-carbon PAHs is frequently poor, leading to processing difficulties during device fabrication and limiting their practical application in solution-processed OLEDs. The lack of functional handles on the molecular periphery also restricts the ability to fine-tune electronic properties, resulting in materials that are rigid and difficult to adapt for specific optoelectronic requirements.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a streamlined one-pot reaction strategy that dramatically simplifies the manufacturing workflow. By employing trifluoromethanesulfonic acid (TfOH) as a catalyst in trifluoroacetic acid (TFA), the process facilitates dehydration, denitrification, and electrophilic cyclization in a single operational step. This method eliminates the need for intermediate isolation and reduces the consumption of organic solvents, directly contributing to cost reduction in organic optoelectronic material manufacturing. The introduction of nitrogen atoms not only enhances the solubility of the final product but also provides active sites for further multifunctional modification, allowing for precise control over the material's electronic characteristics. This strategic shift from complex multi-step syntheses to a concise, acid-catalyzed cyclization offers a scalable pathway for producing high-purity organic semiconductors suitable for industrial applications.
Mechanistic Insights into TfOH-Catalyzed Electrophilic Cyclization
The core of this synthetic breakthrough lies in the efficient activation of the Bisanthene-2CHO precursor through a sophisticated acid-catalyzed mechanism. The reaction initiates with the interaction between the aldehyde groups of Bisanthene-2CHO and azidotrimethylsilane (TMSN3) under the strong acidic conditions provided by TfOH. This environment promotes a cascade of transformations, including dehydration and the generation of reactive nitrene or imine intermediates, which subsequently undergo Friedel-Crafts-type alkylation and electrophilic cyclization. The result is the seamless formation of the nitrogen-doped six-membered rings at the zigzag edges of the ovalene core. This mechanism is particularly advantageous because it avoids the use of stoichiometric oxidants or metal catalysts, thereby reducing the chemical waste associated with the production of complex polycyclic aromatic hydrocarbons.
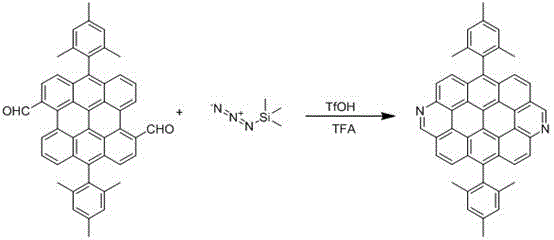
From an impurity control perspective, the one-pot nature of this reaction significantly minimizes the formation of side products that typically arise during multi-step sequences. The use of TFA as both solvent and reaction medium ensures a homogeneous environment that favors the desired cyclization pathway over competing polymerization or decomposition reactions. The resulting imine structure within the six-membered ring is inherently stable, yet reactive enough to allow for subsequent derivatization if needed. For quality assurance teams, this means that the crude product contains fewer structurally similar impurities, simplifying the chromatographic purification steps. The ability to achieve such high structural precision through a simple acid-catalyzed process underscores the robustness of this method for producing high-purity OLED materials that meet the stringent specifications required for commercial display technologies.
How to Synthesize Ovalene-2N Efficiently
The synthesis of this advanced optoelectronic material is designed to be operationally simple while maintaining high standards of chemical precision. The protocol involves mixing the precursor Bisanthene-2CHO with azidotrimethylsilane and trifluoromethanesulfonic acid in a specific mass ratio within a trifluoroacetic acid solvent system. The reaction mixture is then subjected to controlled heating at 60°C for a duration of 12 hours to ensure complete conversion. Following the reaction, a straightforward workup procedure involving aqueous quenching and organic extraction allows for the isolation of the crude product, which is then purified via column chromatography. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations.
- Mix Bisanthene-2CHO, azidotrimethylsilane, and trifluoromethanesulfonic acid in trifluoroacetic acid at a specific mass ratio.
- Stir the reaction mixture at 60°C for 12 hours to facilitate dehydration and electrophilic cyclization.
- Quench with aqueous NaOH, extract with ethyl acetate, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers substantial strategic benefits regarding cost efficiency and supply reliability. The elimination of transition metal catalysts removes a significant cost driver and supply chain bottleneck, as precious metals are often subject to price volatility and geopolitical supply risks. Furthermore, the simplified one-pot process reduces the overall processing time and labor requirements, leading to significant cost savings in manufacturing operations. The use of readily available reagents such as TFA and TfOH ensures that the supply chain remains robust and less susceptible to disruptions caused by specialized raw material shortages. This operational simplicity translates directly into enhanced supply chain reliability, allowing for more predictable production schedules and faster response times to market demands.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily by removing the need for expensive transition metal catalysts and complex purification steps associated with metal removal. By utilizing a simple acid-catalyzed system, the material costs are significantly reduced, and the waste treatment costs are lowered due to the absence of heavy metal contaminants. This streamlined approach allows for a more economical production model, making high-performance organic semiconductors more accessible for large-scale commercial applications without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: The reliance on common organic reagents and solvents ensures a stable supply chain that is not dependent on scarce or specialized resources. The robustness of the reaction conditions means that production can be scaled up with minimal risk of batch failure due to sensitive catalyst deactivation. This stability is crucial for maintaining continuous supply to downstream device manufacturers, reducing lead time for high-purity OLED materials and ensuring that production targets are met consistently even during periods of high market demand.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis facilitates easier scale-up from laboratory to commercial production, as there are fewer unit operations to manage and optimize. The reduction in solvent consumption and the absence of toxic heavy metals simplify waste management and ensure compliance with increasingly stringent environmental regulations. This environmental advantage not only reduces regulatory risk but also aligns with the sustainability goals of modern electronics manufacturers, enhancing the overall marketability of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this nitrogen-doped ovalene derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this material into their existing product lines or research pipelines.
Q: What are the primary advantages of nitrogen doping in Ovalene structures?
A: Nitrogen doping at the zigzag edges significantly improves the stability and solubility of the polycyclic aromatic hydrocarbon compared to all-carbon analogs, while allowing for tunable electronic properties essential for OLED applications.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes trifluoromethanesulfonic acid (TfOH) as an organocatalyst in trifluoroacetic acid, eliminating the need for costly transition metals and simplifying the purification process.
Q: What is the reported yield optimization for this reaction?
A: Experimental data indicates that extending the reaction time to 12 hours at 60°C optimizes the yield, achieving up to 53% conversion under the specified conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ovalene-2N Supplier
As the demand for advanced organic optoelectronic materials continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of Ovalene-2N meets the high standards required for next-generation OLED and OFET applications. We understand the critical nature of material consistency in electronic devices and have optimized our processes to deliver reliable performance every time.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our advanced synthesis capabilities. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your product development timeline and enhance your competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
