Advanced Asymmetric Catalysis for 5,11-Methanomorphanthridine Alkaloid Intermediates and Commercial Scale-Up
Advanced Asymmetric Catalysis for 5,11-Methanomorphanthridine Alkaloid Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient pathways to access complex natural product scaffolds, particularly those exhibiting potent biological activities such as anticancer and antiviral properties. Patent CN103012422A introduces a groundbreaking methodology for the preparation of optically pure 5,11-methanomorphanthridine compounds, which serve as pivotal intermediates for the Amaryllidaceae Montanine type alkaloids. This technology addresses the longstanding challenges associated with the scarcity of natural resources and the complexity of total synthesis. By leveraging a biomimetic strategy combined with modern asymmetric catalysis, the disclosed route enables the rapid construction of the characteristic 5,11-methanomorphanthridine nucleus. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity precursors for drug development programs targeting neurological and oncological indications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of Montanine type alkaloids has been plagued by inefficient methodologies that hinder commercial viability. Traditional approaches often rely on the use of stoichiometric chiral auxiliaries or the resolution of racemic mixtures, which inherently limit the maximum theoretical yield to 50% and generate substantial chemical waste. Furthermore, existing routes frequently involve tediously long synthetic sequences, sometimes exceeding fifteen steps, resulting in poor overall combined coefficients and escalating production costs. The structural complexity of the 5,11-methylene radical morphanthridine skeleton makes the introduction of functional groups at the C-2 and C-3 positions particularly difficult using prior art, often requiring harsh conditions that compromise stereochemical integrity. These limitations create significant bottlenecks for supply chain heads who require consistent, scalable sources of high-purity API intermediates without the volatility associated with extraction from rare plant sources.
The Novel Approach
The innovative process detailed in the patent overcomes these barriers through a concise four-step sequence starting from readily available aromatic nitro ethene derivatives. The core breakthrough lies in the application of a rhodium-catalyzed asymmetric conjugate addition followed by a unique oxidative dearomatization-azepine Michael addition cascade. This strategy effectively constructs the complex bridged ring system in a single operational step, drastically reducing the step count compared to classical methods. By employing a chiral sulfoxide phosphine ligand with a rhodium catalyst, the process achieves high enantiomeric excess early in the synthesis, which is further enhanced through a simple recrystallization of a key intermediate. This approach not only improves the overall yield but also introduces oxygen functionality at the C-2/C-3 positions in advance, solving the difficult problem of late-stage functionalization. 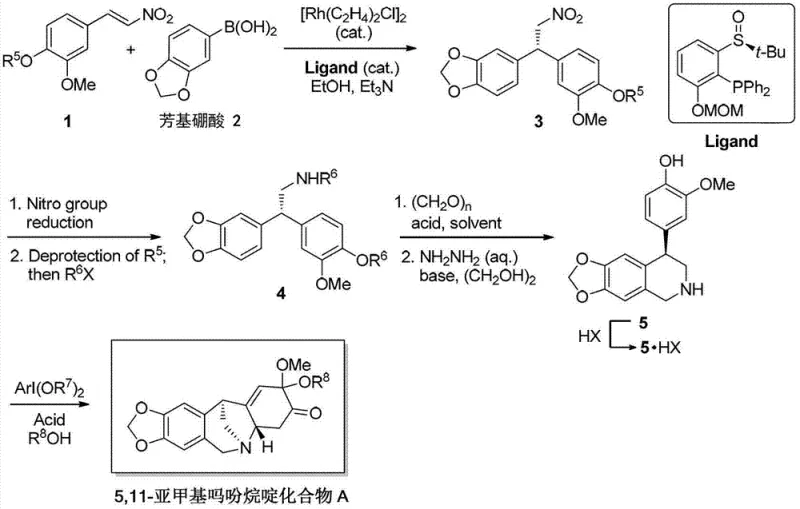
Mechanistic Insights into Rh-Catalyzed Asymmetric Conjugate Addition and Oxidative Cascade
The success of this synthesis hinges on the precise control of stereochemistry during the initial carbon-carbon bond formation. The reaction utilizes a cationic rhodium complex generated in situ from [Rh(C2H4)2Cl]2 and a chiral sulfoxide phosphine ligand. This catalyst system facilitates the enantioselective addition of aryl boronic acids to nitro ethene derivatives, establishing the first chiral center with high fidelity. The mechanism involves the transmetallation of the aryl group to the rhodium center, followed by migratory insertion into the nitro alkene double bond. The steric environment provided by the bulky tert-butyl sulfoxide moiety on the ligand dictates the facial selectivity of the attack, ensuring the formation of the desired enantiomer. Subsequent steps involve careful manipulation of protecting groups, where the simultaneous protection of the amine and phenol functionalities plays a crucial role in stabilizing the intermediate for the critical cyclization event.
Impurity control is meticulously managed through the design of the cascade reaction in the final step. The transformation of the tetrahydroquinoline intermediate into the target 5,11-methanomorphanthridine skeleton employs hypervalent iodine reagents, such as diacetoxyiodobenzene or bis(trifluoroacetoxy)iodobenzene, in an acidic medium. This promotes an oxidative dearomatization of the phenol ring, generating a reactive quinone monoketal species that immediately undergoes an intramolecular Michael addition with the tethered amine. This tandem process minimizes the accumulation of unstable intermediates and side products. Furthermore, the patent highlights that the optical purity of the key diarylamine intermediate can be upgraded to greater than 99% ee via recrystallization from ethyl acetate and n-hexane. This purification capability is vital for meeting the stringent purity specifications required for clinical grade materials, ensuring that downstream derivatives maintain high stereochemical integrity.
How to Synthesize 5,11-Methanomorphanthridine Compound A Efficiently
The synthesis of this valuable intermediate is designed for operational simplicity and robustness, making it highly attractive for process chemistry teams aiming for technology transfer. The route begins with the rhodium-catalyzed coupling in ethanol, followed by a reduction and protection sequence that yields a crystalline solid amenable to purification. The subsequent Pictet-Spengler cyclization and final oxidative cascade are performed under relatively mild conditions, avoiding the need for cryogenic temperatures in the key bond-forming steps. Detailed standard operating procedures for each transformation, including specific reagent ratios, solvent choices, and workup protocols, are essential for reproducing the high yields and selectivity reported in the patent literature. The following guide outlines the critical stages for implementing this chemistry in a pilot or production setting.
- Perform Rh-catalyzed asymmetric conjugate addition of aryl boronic acid to aromatic nitro ethene derivatives to generate chiral diaryl nitro-compound.
- Reduce the nitro group to primary amine, deprotect phenol, and simultaneously protect amine and phenol groups to obtain high optical purity diarylamine.
- Execute Pictet-Spengler reaction with paraformaldehyde followed by hydrazinolysis to form the tetrahydroquinoline intermediate.
- Conduct oxidative dearomatization-azepine Michael addition cascade using hypervalent iodine reagents to construct the final 5,11-methanomorphanthridine skeleton.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits over traditional extraction or longer synthetic pathways. The primary advantage stems from the drastic reduction in process mass intensity (PMI) achieved by shortening the synthetic sequence. By consolidating multiple transformations into a concise four-step linear route, the consumption of raw materials, solvents, and energy is significantly lowered, directly translating to cost reduction in pharmaceutical intermediates manufacturing. The use of conventional, commercially available reagents such as zinc powder, paraformaldehyde, and hypervalent iodine oxidants eliminates the dependency on exotic or custom-synthesized building blocks that often suffer from supply volatility. Additionally, the robustness of the reaction conditions, with many steps tolerating ambient atmosphere and moisture, reduces the capital expenditure required for specialized equipment like gloveboxes or extensive drying systems.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the high efficiency of the rhodium catalyst system substantially lower the cost of goods sold (COGS). The ability to upgrade optical purity through recrystallization rather than preparative chiral chromatography represents a massive saving in both time and consumable costs at scale. Furthermore, the high overall yield ensures that less starting material is required to produce a kilogram of the final active intermediate, optimizing the utilization of expensive aryl boronic acids and specialized ligands.
- Enhanced Supply Chain Reliability: Sourcing natural alkaloids from plant extracts is inherently risky due to seasonal variations, geopolitical instability in growing regions, and low natural abundance. This fully synthetic route decouples production from agricultural constraints, ensuring a consistent and predictable supply of high-purity intermediates. The use of stable intermediates that can be isolated and stored allows for flexible production scheduling, enabling manufacturers to build inventory buffers against market fluctuations. This reliability is critical for maintaining uninterrupted production lines for downstream API manufacturing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like ethanol, methanol, and dichloromethane which are well-understood in large-scale chemical engineering. The avoidance of heavy metal contaminants in the final product, thanks to efficient purification steps, simplifies regulatory compliance and reduces the burden on waste treatment facilities. The atom economy of the cascade reaction minimizes waste generation, aligning with green chemistry principles and reducing the environmental footprint associated with the disposal of hazardous byproducts.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the scalability, purity, and versatility of the 5,11-methanomorphanthridine platform. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the key advantage of this synthesis route for Montanine alkaloids?
A: The route utilizes a biomimetic 'de-aromatization/azepine Michael addition cascade' to construct the core skeleton in fewer steps with high enantioselectivity, avoiding tedious chiral auxiliary methods.
Q: How is high optical purity achieved in the intermediate stages?
A: High optical purity (>99% ee) is achieved through a combination of asymmetric rhodium catalysis and a critical recrystallization step of the protected diarylamine intermediate.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses conventional reagents, tolerates moisture and oxygen in many steps, and avoids extremely low temperatures or inert atmospheres for most transformations, facilitating scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,11-Methanomorphanthridine Compound A Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing high-quality intermediates for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in asymmetric catalysis and complex ring construction allows us to optimize this specific patent route for maximum efficiency and cost-effectiveness, providing our partners with a competitive edge in the marketplace.
We invite you to collaborate with us to leverage this advanced technology for your drug development projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term supply goals. Let us be your partner in transforming complex chemical challenges into commercial successes.
