Revolutionizing Triazole Synthesis: Scalable Metal-Free Manufacturing for Pharmaceutical Intermediates
The patent CN113121462A introduces a groundbreaking methodology for synthesizing 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical limitations in traditional heterocyclic chemistry. This innovation represents a significant advancement in the production of nitrogen-containing heterocycles that serve as essential building blocks for bioactive molecules across pharmaceutical and agrochemical sectors. The process eliminates hazardous azide intermediates while maintaining high reaction efficiency through a novel base-promoted mechanism. By utilizing readily accessible starting materials including cesium carbonate and molecular sieves, the method achieves exceptional operational simplicity without requiring specialized equipment or stringent environmental controls. This patent establishes a new paradigm for manufacturing triazole-based intermediates that meet the rigorous demands of modern drug development pipelines while offering substantial advantages in process safety and scalability for industrial implementation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
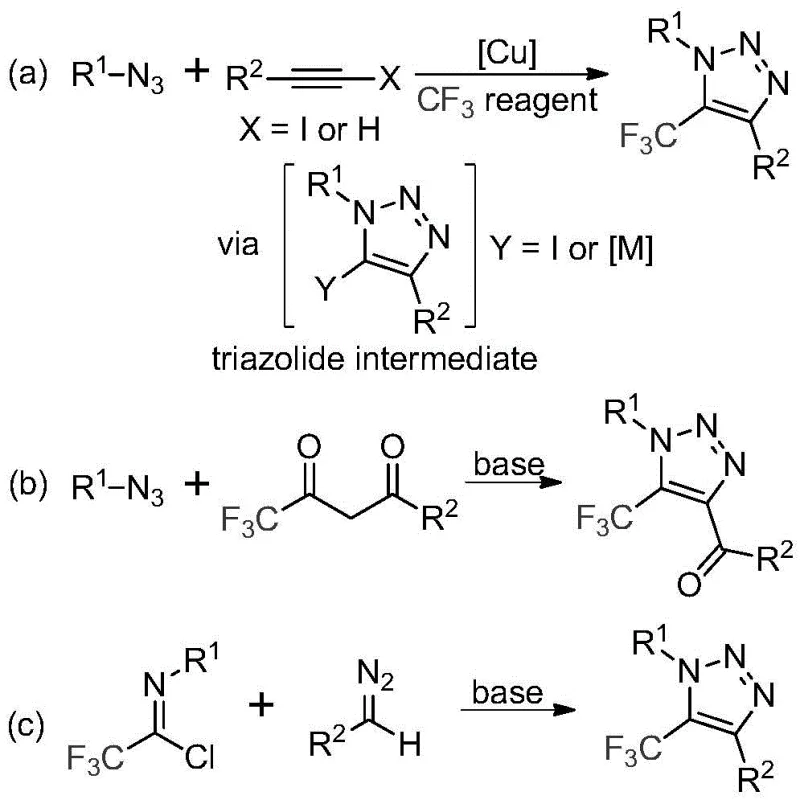
Traditional synthesis routes for trifluoromethyl triazoles predominantly rely on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, which present significant safety hazards due to the explosive nature of azide compounds. These methods require toxic transition metal catalysts that necessitate complex purification steps to remove residual metals below pharmacopeial limits, substantially increasing production costs and complicating regulatory compliance. The reaction conditions often demand cryogenic temperatures or high-pressure systems to control exothermic processes, creating substantial barriers to large-scale manufacturing. Furthermore, the narrow substrate scope and sensitivity to functional groups limit the structural diversity achievable through conventional approaches, restricting their applicability in developing novel therapeutic candidates. The multi-step nature of these processes also generates considerable waste streams that require specialized treatment before disposal, adding environmental compliance burdens to the manufacturing workflow.
The Novel Approach
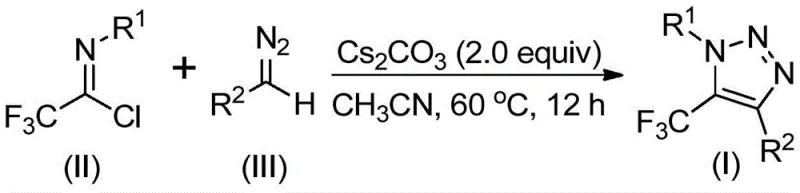
The patented methodology overcomes these challenges through an elegant base-promoted reaction between trifluoroethylimidoyl chloride and diazo compounds under mild thermal conditions (50–70°C). This metal-free process eliminates the need for hazardous azides while maintaining high functional group tolerance across diverse substrates as demonstrated in the patent examples. The reaction proceeds through a nucleophilic addition elimination mechanism followed by intramolecular cyclization, achieving excellent yields without generating toxic byproducts. Crucially, the use of cesium carbonate as a promoter enables simple workup procedures involving filtration and standard column chromatography purification, significantly reducing processing time compared to conventional metal removal protocols. The method's robustness is further evidenced by its successful application across various substituent patterns on both aryl and aroyl groups, providing unprecedented flexibility in designing triazole derivatives for specific pharmaceutical applications while maintaining operational simplicity at scale.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism involves a precisely orchestrated sequence where cesium carbonate facilitates deprotonation of the diazo compound, generating a nucleophilic species that attacks the electrophilic carbon of trifluoroethylimidoyl chloride. This intermolecular addition forms a key triazolide intermediate through nucleophilic substitution, followed by intramolecular cyclization via a 5-endo-dig process that establishes the triazole ring structure with perfect regioselectivity at the 5-position. The absence of transition metals prevents unwanted side reactions such as dimerization or reduction pathways commonly observed in copper-catalyzed systems, while the mild thermal conditions (60°C) maintain optimal kinetic control throughout the transformation. Molecular sieve addition effectively scavenges trace moisture that could hydrolyze sensitive intermediates, ensuring consistent reaction progression without requiring anhydrous conditions typically demanded by organometallic methodologies. This mechanistic pathway demonstrates exceptional substrate versatility across various aryl and aroyl substituents while maintaining high conversion efficiency through multiple reaction cycles.
Impurity control is achieved through the inherent selectivity of the base-promoted cyclization mechanism, which minimizes formation of regioisomers and dimeric byproducts that plague conventional approaches. The absence of transition metals eliminates concerns about residual catalyst contamination that would require additional purification steps to meet pharmaceutical quality standards. The well-defined reaction pathway produces minimal side products as evidenced by clean chromatographic profiles in patent examples, with impurities primarily limited to unreacted starting materials that are easily separated during standard column chromatography purification. This inherent selectivity translates directly to higher product purity without requiring specialized analytical monitoring or additional processing steps, making the method particularly suitable for manufacturing intermediates where stringent purity specifications are mandatory for subsequent synthetic steps in API production.
How to Synthesize 5-Trifluoromethyl Triazole Efficiently
This innovative synthesis route represents a significant advancement in manufacturing complex triazole intermediates through its elegant combination of operational simplicity and chemical efficiency. The patented methodology eliminates multiple pain points associated with traditional approaches while maintaining excellent functional group compatibility across diverse molecular architectures. By leveraging readily available starting materials and standard laboratory equipment, this process enables rapid implementation in existing manufacturing facilities without requiring substantial capital investment or specialized training. The following standardized procedure details the precise implementation steps for consistent production of high-purity triazole compounds at commercial scale.
- Combine cesium carbonate, molecular sieve, trifluoroethylimidoyl chloride, and diazo compound in acetonitrile solvent under nitrogen atmosphere
- Maintain reaction temperature at 60°C for 12 hours with continuous stirring to ensure complete conversion
- Perform post-processing through filtration, silica gel mixing, and column chromatography purification to obtain final product
Commercial Advantages for Procurement and Supply Chain Teams
This manufacturing innovation directly addresses critical pain points in pharmaceutical supply chains by transforming triazole intermediate production from a hazardous, complex operation into a streamlined, reliable process. The elimination of specialized handling requirements for dangerous reagents significantly reduces operational risks while creating new opportunities for cost optimization throughout the production lifecycle. By simplifying the synthetic pathway and reducing dependency on scarce catalysts, this method enhances supply chain resilience against market volatility while improving overall manufacturing economics for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated purification steps creates substantial cost savings through reduced raw material expenses and simplified processing workflows. Avoiding hazardous azide compounds removes significant safety compliance costs related to specialized handling equipment and personnel training while decreasing waste treatment expenditures through cleaner reaction profiles. The use of commercially available starting materials at optimal stoichiometric ratios minimizes material waste without requiring complex recycling systems, translating to more efficient resource utilization across the entire production cycle.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of widely available starting materials that can be procured from multiple global suppliers without dependency on specialized chemical manufacturers. The robust reaction conditions tolerate minor variations in raw material quality while maintaining consistent output specifications, reducing batch failure risks that typically disrupt supply continuity. Simplified logistics requirements eliminate the need for specialized transportation and storage conditions for hazardous intermediates, enabling more reliable just-in-time delivery schedules that align with modern lean manufacturing principles.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes without requiring fundamental modifications to reaction parameters or equipment configuration. Standard purification techniques using conventional chromatography systems ensure straightforward technology transfer between development and manufacturing facilities while maintaining consistent quality attributes. The elimination of toxic metal residues and reduction in hazardous waste streams significantly lowers environmental compliance burdens while supporting corporate sustainability initiatives through inherently greener chemistry principles that align with global regulatory expectations.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113121462A's experimental data and implementation requirements. These insights reflect practical experience from scaling similar methodologies across multiple pharmaceutical intermediate production platforms while maintaining strict adherence to quality and regulatory standards.
Q: How does this method eliminate safety risks associated with traditional triazole synthesis?
A: The process replaces toxic azide-based routes with stable diazo compounds and trifluoroethylimidoyl chloride precursors, removing explosion hazards while maintaining high functional group tolerance as demonstrated in patent examples.
Q: What enables the commercial scalability of this synthesis route?
A: Mild reaction conditions (50–70°C), readily available starting materials, and straightforward purification via column chromatography allow seamless transition from gram-scale lab production to industrial manufacturing volumes.
Q: How does the base-promoted mechanism improve product purity for pharmaceutical applications?
A: The cesium carbonate-mediated nucleophilic addition eliminates transition metal residues and reduces byproduct formation, yielding triazoles with stringent purity specifications suitable for API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex heterocyclic intermediates like this patented triazole compound. We maintain stringent purity specifications through advanced analytical capabilities and rigorous QC labs that ensure consistent product quality meeting global regulatory requirements across all manufacturing scales. Our technical team has successfully implemented similar base-promoted methodologies for numerous clients, delivering reliable supply chains for critical pharmaceutical building blocks while optimizing production economics through continuous process improvement initiatives.
Leverage our expertise with a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments for integrating this innovative synthesis into your supply chain operations.
