Advanced TEMPO-Mediated Oxidation for Commercial Production of 1-Benzyl-4-Piperidinealdehyde Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly those serving high-value therapeutic areas such as neurodegenerative disorders. Patent CN102079720B introduces a transformative methodology for the preparation of 1-benzylpiperidine-4-carboxaldehyde, a pivotal building block in the synthesis of Donepezil (E2020), a leading medication for Alzheimer's disease. This innovation addresses long-standing challenges in process chemistry by replacing hazardous, energy-intensive oxidation protocols with a mild, catalytic system based on 2,2,6,6-tetramethyl-1-piperidone (TEMPO). For R&D directors and procurement specialists, understanding this shift is crucial, as it directly impacts the cost structure and supply reliability of high-purity pharmaceutical intermediates. The structural integrity of the target molecule, essential for downstream coupling reactions, is preserved through this selective oxidation strategy.
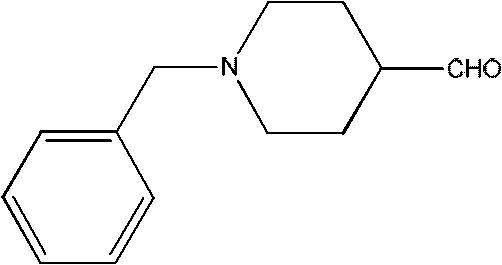
Traditionally, the synthesis of 1-benzyl-4-piperidinealdehyde relied heavily on activated DMSO oxidation methods, such as the Swern oxidation or variations utilizing oxalyl chloride. These conventional approaches, while chemically effective on a small scale, present severe limitations when evaluated through the lens of industrial manufacturing and cost reduction in API manufacturing. The requirement for cryogenic conditions, often necessitating temperatures as low as -50°C, imposes a massive energy burden on production facilities and requires specialized cooling infrastructure that increases capital expenditure. Furthermore, the use of oxalyl chloride generates stoichiometric amounts of corrosive byproducts and noxious gases, complicating waste treatment and posing significant safety risks to plant personnel. The workup procedures associated with these older methods are notoriously tedious, often requiring column chromatography to remove impurities, which drastically reduces overall throughput and increases solvent consumption.
In stark contrast, the novel approach detailed in the patent utilizes a biphasic oxidation system comprising TEMPO, sodium periodate, and sodium bromide. This method operates efficiently within a温和 temperature range of 0°C to 40°C, with optimal results observed at ambient conditions (20°C to 25°C). By eliminating the need for extreme cooling, the process inherently lowers utility costs and simplifies reactor design, making it an attractive option for a reliable pharmaceutical intermediate supplier aiming to optimize their production lines. The selectivity of the TEMPO catalyst ensures that the primary alcohol is oxidized to the aldehyde without significant over-oxidation to the corresponding carboxylic acid, a common side reaction in less controlled environments. This high chemoselectivity translates directly into simplified isolation procedures, where simple phase separation and washing suffice to achieve high purity, thereby enhancing the overall economic viability of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on oxalyl chloride-based oxidation protocols represents a significant bottleneck in the commercial scale-up of complex piperidine derivatives. Beyond the aforementioned thermal constraints, the handling of oxalyl chloride requires stringent safety measures due to its moisture sensitivity and the release of carbon monoxide and hydrogen chloride during the reaction. These factors contribute to extended batch cycles, as additional time is required for safe reagent addition and gas scrubbing. Moreover, the formation of sulfonium byproducts in DMSO-based oxidations can lead to difficult-to-remove impurities that persist through subsequent synthetic steps, potentially compromising the quality of the final Active Pharmaceutical Ingredient (API). For supply chain managers, the dependency on specialized reagents and the generation of hazardous waste streams create vulnerabilities in the supply chain, increasing the risk of regulatory delays and environmental compliance issues.
The Novel Approach
The TEMPO-mediated oxidation pathway offers a streamlined alternative that aligns with modern principles of green chemistry and process efficiency. By employing sodium periodate as the terminal oxidant and sodium bromide as a co-catalyst, the system regenerates the active oxoammonium species of TEMPO in situ, allowing for catalytic loading as low as 1:100 relative to the substrate. This drastic reduction in catalyst loading minimizes the cost of goods sold (COGS) associated with reagent procurement. The reaction proceeds in common organic solvents such as dichloromethane, which facilitates easy product isolation. The robustness of this method is evidenced by its tolerance to slight variations in temperature and stirring rates, providing a wider operating window that is forgiving in large-scale reactors. This reliability is paramount for ensuring consistent batch-to-batch quality, a key metric for any reliable pharmaceutical intermediate supplier serving regulated markets.
Mechanistic Insights into TEMPO-Catalyzed Oxidation
The core of this technological advancement lies in the catalytic cycle of the nitroxyl radical TEMPO. In the presence of sodium bromide and sodium periodate, TEMPO is oxidized to the reactive oxoammonium cation. This electrophilic species selectively abstracts a hydride from the benzylic alcohol substrate, 1-benzyl-4-piperidine carbinol, converting it into the desired aldehyde while being reduced back to the hydroxylamine form. The sodium periodate then re-oxidizes the hydroxylamine and bromide species to regenerate the active catalyst and bromine equivalents, closing the cycle. This mechanism avoids the formation of alkoxysulfonium salts typical of Swern oxidations, thereby bypassing the pathways that lead to Pummerer rearrangement byproducts. Understanding this mechanistic nuance is vital for R&D teams, as it explains the superior impurity profile observed in the final product.
Impurity control is further enhanced by the biphasic nature of the reaction system. The organic substrate and catalyst reside primarily in the organic phase, while the inorganic oxidants remain in the aqueous phase. This phase separation limits the exposure of the sensitive aldehyde product to excessive oxidative stress, preventing over-oxidation to 1-benzyl-4-piperidinecarboxylic acid. Additionally, the mild pH conditions maintained by the buffer action of the salt system prevent acid-catalyzed degradation of the piperidine ring. For quality assurance professionals, this means that the resulting crude material often meets stringent purity specifications (>99% by HPLC) without the need for recrystallization or chromatographic purification. Such high purity is critical when the intermediate is used for the synthesis of E2020, where trace impurities could affect the pharmacokinetic profile of the final drug.
How to Synthesize 1-Benzyl-4-Piperidinealdehyde Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and phase transfer dynamics to maximize yield and safety. The process begins with the dissolution of the alcohol substrate and the TEMPO catalyst in the chosen organic solvent, followed by the controlled addition of the aqueous oxidant solution. Maintaining vigorous stirring is essential to ensure sufficient interfacial area for the reaction to proceed at an optimal rate. While the general procedure is robust, specific parameters regarding stoichiometry and workup are critical for reproducibility. The detailed standardized synthesis steps, including precise molar ratios and quenching protocols, are outlined below to assist technical teams in replicating this high-efficiency process.
- Prepare the reaction mixture by dissolving 1-benzyl-4-piperidine carbinol and catalytic TEMPO in dichloromethane within a three-necked flask.
- Add the aqueous oxidant solution containing sodium periodate and sodium bromide slowly while maintaining the temperature between 0°C and 40°C.
- Monitor reaction progress via TLC, then separate layers, wash the organic phase with sodium thiosulfate, dry, and concentrate to obtain the pure aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this TEMPO-based oxidation technology offers compelling advantages that extend beyond mere chemical yield. For procurement managers tasked with cost reduction in pharmaceutical intermediate manufacturing, the elimination of cryogenic cooling represents a direct and substantial decrease in utility expenses. The ability to run reactions at ambient temperature removes the need for expensive glycol chillers and insulated reactors, lowering both capital investment and maintenance overheads. Furthermore, the use of commodity chemicals like sodium periodate and sodium bromide, as opposed to specialized and hazardous reagents like oxalyl chloride, stabilizes the raw material supply chain against market volatility. This stability ensures consistent pricing and availability, which is crucial for long-term contract planning.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the operational workflow. By removing the requirement for -50°C conditions, the energy footprint of the manufacturing process is drastically reduced, leading to significant operational cost savings. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the volume of solvents and adsorbents needed for purification. This efficiency translates to a lower cost per kilogram of the final intermediate, allowing suppliers to offer more competitive pricing without sacrificing margins. The avoidance of column chromatography in favor of simple extraction and distillation further accelerates production throughput, maximizing asset utilization.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available, non-restricted reagents. Unlike certain chlorinating agents that may face regulatory scrutiny or transportation restrictions, sodium salts and TEMPO are stable and easy to source globally. This reduces the risk of supply disruptions due to logistical bottlenecks or regulatory changes. Moreover, the robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to extreme temperature control systems. For supply chain heads, this translates to more predictable lead times and a higher degree of confidence in meeting delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot and commercial plant is straightforward due to the manageable exotherm and lack of hazardous gas evolution. The absence of carbon monoxide generation simplifies venting requirements and improves workplace safety standards. From an environmental standpoint, the aqueous waste stream contains primarily inorganic salts which are easier to treat than the complex organic waste generated by traditional methods. This alignment with green chemistry principles facilitates smoother environmental permitting and reduces the liability associated with hazardous waste disposal, making the process sustainable for long-term commercial scale-up of complex piperidine derivatives.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis method, we have compiled a set of questions based on the patent data and industry experience. These answers provide clarity on the practical implementation and benefits of the TEMPO oxidation route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply chains. The following section addresses specific concerns regarding purity, scalability, and comparative advantages.
Q: Why is the TEMPO oxidation method superior to traditional oxalyl chloride methods for this intermediate?
A: The TEMPO method operates at mild temperatures (0°C to 40°C) compared to the cryogenic -50°C required for oxalyl chloride methods, significantly reducing energy costs and operational complexity while minimizing side reactions like over-oxidation.
Q: What is the expected purity level achievable with this patented process?
A: According to patent data, this process consistently achieves purity levels exceeding 99% (HPLC) without the need for complex column chromatography, making it highly suitable for direct use in downstream API synthesis.
Q: Is this process scalable for industrial manufacturing of Alzheimer's drug intermediates?
A: Yes, the patent explicitly demonstrates successful scaling from gram-scale to multi-kilogram batches (e.g., 5.1kg substrate) with consistent yields above 90%, confirming its viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyl-4-Piperidinealdehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications like E2020. Our technical team has extensively analyzed the TEMPO-mediated oxidation pathway described in CN102079720B and possesses the expertise to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1-benzyl-4-piperidinealdehyde meets the exacting standards required for pharmaceutical synthesis.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for Alzheimer's disease therapeutics. By leveraging our manufacturing capabilities, you can secure a stable source of this critical intermediate while achieving significant cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can enhance your project's success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
