Advanced Iridium-Catalyzed Synthesis of Chiral Alpha-Amino Acid Esters for Industrial Scale-Up
Advanced Iridium-Catalyzed Synthesis of Chiral Alpha-Amino Acid Esters for Industrial Scale-Up
The pharmaceutical industry continuously demands more efficient and stereoselective routes for producing non-natural amino acids, which serve as critical building blocks for peptide drugs and active pharmaceutical ingredients (APIs). Patent CN112279743B introduces a groundbreaking methodology for the synthesis of chiral alpha-amino acid ester derivatives through the asymmetric hydrogenation of imido esters. This technology leverages a sophisticated catalytic system comprising a metal iridium precursor and a novel chiral ferrocene skeleton phosphine-phosphoramidite ligand. Unlike traditional methods that often struggle with limited substrate scope or harsh reaction conditions, this invention provides a robust pathway characterized by high atom economy and exceptional enantioselectivity. The process is designed to be operationally simple, utilizing mild temperatures and standard hydrogenation equipment, making it highly attractive for reliable pharmaceutical intermediate supplier networks aiming to streamline their production pipelines. By achieving enantiomeric excess (ee) values exceeding 96% in optimized cases, this technique sets a new benchmark for purity and efficiency in the manufacturing of complex chiral intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amino acid esters has relied heavily on methods such as the asymmetric hydrogenation of dehydroamino acid esters or early transition metal-catalyzed reductions of alpha-imino esters. While effective for specific substrates, these conventional approaches often suffer from significant drawbacks that hinder large-scale adoption. For instance, earlier palladium or rhodium-based systems frequently exhibited narrow substrate tolerance, failing to maintain high stereoselectivity when bulky or electron-deficient aromatic groups were present on the imine nitrogen or the alpha-carbon. Furthermore, many legacy processes require expensive chiral auxiliaries or resolving agents, which drastically increases the cost of goods sold (COGS) and generates substantial chemical waste. The need for cryogenic temperatures or extremely high pressures in some older protocols also poses safety risks and energy inefficiencies, complicating the commercial scale-up of complex pharmaceutical intermediates. These limitations create bottlenecks for procurement managers seeking cost-effective and scalable solutions for API manufacturing.
The Novel Approach
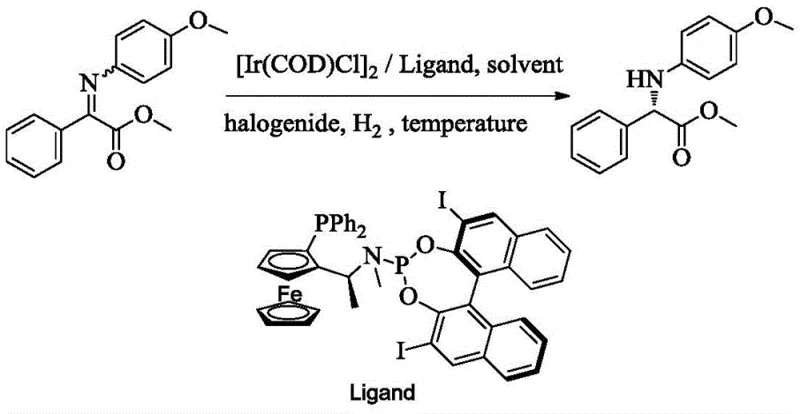
The methodology disclosed in CN112279743B represents a paradigm shift by utilizing an iridium complex coordinated with a specialized chiral ferrocene-phosphine-phosphoramidite ligand. This novel catalytic system overcomes the substrate limitations of previous generations, demonstrating remarkable versatility across a wide range of aromatic and aliphatic imido esters. The reaction conditions are notably mild, typically operating between -20°C and 50°C, which significantly reduces energy consumption compared to high-temperature alternatives. Moreover, the inclusion of specific additives like iodine or N-iodosuccinimide activates the catalyst in situ, ensuring rapid turnover frequencies and high yields often exceeding 90%. This approach not only simplifies the operational workflow but also enhances the overall green chemistry profile of the synthesis by minimizing waste and avoiding toxic heavy metal residues often associated with less selective catalysts. For supply chain heads, this translates to a more reliable and continuous production capability with reduced lead times for high-purity intermediates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the unique structure of the chiral ligand and its interaction with the iridium center. The ligand features a ferrocene backbone which provides a rigid, chiral environment essential for inducing high stereoselectivity during the hydrogenation event. The phosphine-phosphoramidite motif acts as a bidentate coordinator, stabilizing the iridium species while leaving open coordination sites for the substrate and hydrogen gas. 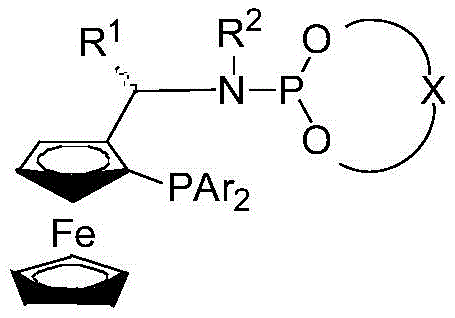 During the catalytic cycle, the iridium complex undergoes oxidative addition with molecular hydrogen, followed by the coordination of the alpha-imino ester substrate. The chiral pocket created by the ferrocene scaffold directs the hydride transfer to the prochiral carbon of the imine bond with extreme precision, favoring the formation of one enantiomer over the other. This precise spatial control is what allows the system to achieve ee values up to 96%, even for challenging substrates like 2-thienyl or cyclohexyl derivatives. The mechanistic robustness ensures that minor variations in substrate electronics do not compromise the stereochemical outcome, a critical factor for R&D directors focused on impurity control.
During the catalytic cycle, the iridium complex undergoes oxidative addition with molecular hydrogen, followed by the coordination of the alpha-imino ester substrate. The chiral pocket created by the ferrocene scaffold directs the hydride transfer to the prochiral carbon of the imine bond with extreme precision, favoring the formation of one enantiomer over the other. This precise spatial control is what allows the system to achieve ee values up to 96%, even for challenging substrates like 2-thienyl or cyclohexyl derivatives. The mechanistic robustness ensures that minor variations in substrate electronics do not compromise the stereochemical outcome, a critical factor for R&D directors focused on impurity control.
Impurity control is further enhanced by the specific choice of additives and reaction parameters. The presence of halogen additives, particularly iodine species, is believed to facilitate the formation of the active cationic iridium-hydride species, which is the true catalytic entity. This activation step prevents the formation of inactive iridium clusters that could lead to non-selective background reactions or catalyst decomposition. Additionally, the mild temperature range prevents thermal degradation of the sensitive imine functionality, thereby reducing the formation of side products such as hydrolysis byproducts or polymerization residues. The high chemoselectivity of the system ensures that other functional groups on the aromatic rings, such as methoxy, fluoro, or chloro substituents, remain intact during the reduction. This fidelity is paramount for downstream synthetic steps where functional group integrity is required for coupling reactions in peptide synthesis.
How to Synthesize Chiral Alpha-Amino Acid Ester Derivatives Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction monitoring to maximize yield and optical purity. The process begins with the in situ generation of the active catalyst, followed by the introduction of the substrate under a controlled hydrogen atmosphere. The flexibility of the system allows for adjustments in pressure and temperature to fine-tune the reaction kinetics for specific substrates. Detailed standardized synthetic steps see the guide below, which outlines the general procedure derived from the patent examples.
- Prepare the catalyst solution by dissolving the iridium-cyclooctadiene complex and the chiral ferrocene skeleton phosphine-phosphoramidite ligand in a solvent such as tetrahydrofuran under nitrogen protection, stirring at room temperature for 10 minutes to ensure complete coordination.
- Add the substrate alpha-imino ester dissolved in solvent along with an additive like iodine or N-iodosuccinimide to the catalyst mixture, then transfer the solution into a high-pressure autoclave.
- Replace the atmosphere with hydrogen three times, pressurize to 20-100 bar, and react at temperatures between -20°C and 50°C for 1 to 24 hours, followed by solvent removal and silica gel column purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this iridium-catalyzed hydrogenation technology offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage lies in the significant reduction of manufacturing complexity. By eliminating the need for stoichiometric chiral resolving agents or multi-step protection-deprotection sequences, the overall process mass intensity (PMI) is drastically lowered. This simplification directly correlates to cost reduction in API manufacturing, as fewer raw materials are consumed, and less waste requires disposal. Furthermore, the catalyst loading can be optimized to very low levels (S/C ratios up to 1000:1 in some embodiments), meaning that the expensive iridium metal and chiral ligand contribute minimally to the final product cost. This economic efficiency makes the process viable for large-volume production without compromising on the quality of the chiral intermediate.
- Cost Reduction in Manufacturing: The streamlined nature of this single-step hydrogenation process eliminates several unit operations typically found in classical amino acid synthesis. By avoiding resolution steps which inherently cap the maximum theoretical yield at 50%, this asymmetric method potentially doubles the material throughput from the same amount of starting imine. The use of common solvents like tetrahydrofuran and the ability to recover and recycle the catalyst system further drive down operational expenditures. Additionally, the high selectivity reduces the burden on downstream purification, allowing for simpler crystallization or distillation protocols rather than expensive preparative chromatography. These factors combine to create a highly cost-competitive manufacturing route suitable for generic drug production as well as innovative new chemical entities.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on scarce natural sources or complex biological fermentation processes for certain chiral amino acids. This chemical synthesis method provides a fully synthetic alternative that is independent of agricultural variables or biological contamination risks. The starting materials, alpha-imino esters, are readily accessible through simple condensation reactions from abundant ketones and amines. This ensures a stable and predictable supply of precursors, mitigating the risk of raw material shortages. Moreover, the robustness of the catalyst system means that production batches are highly reproducible, reducing the likelihood of failed runs that could disrupt inventory levels. This reliability is crucial for maintaining just-in-time delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often introduces unforeseen challenges, but this hydrogenation protocol is inherently scalable due to its reliance on standard high-pressure reactor technology widely available in the fine chemical industry. The reaction exhibits high atom economy, as the only reagent added to the substrate is hydrogen gas, which incorporates completely into the product without generating stoichiometric byproducts. This aligns perfectly with modern environmental regulations and sustainability goals, minimizing the carbon footprint of the manufacturing process. The absence of heavy metal waste streams, thanks to the low catalyst loading and efficient containment, simplifies wastewater treatment and regulatory compliance. Consequently, this method supports the commercial scale-up of complex intermediates with a clear path to green certification.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What is the optimal pressure range for this asymmetric hydrogenation reaction?
A: According to the patent data, the reaction operates effectively within a hydrogen pressure range of 20 to 100 bar, with 50 bar often yielding excellent results in terms of both conversion and enantioselectivity.
Q: Which additives are recommended to enhance catalytic activity?
A: The protocol suggests using halogen-based additives such as iodine, N-iodosuccinimide (NIS), N-bromosuccinimide, or tetrabutylammonium iodide, with iodine and NIS showing superior performance in optimizing yield and ee values.
Q: Can this method be applied to substrates with bulky aromatic groups?
A: Yes, the method demonstrates broad substrate tolerance, successfully processing various aryl groups including phenyl, naphthyl, and substituted phenyl rings (e.g., 4-methoxy, 4-chloro, 4-trifluoromethyl) with high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the iridium-ferrocene system described in CN112279743B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of chiral amino acid ester meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process chemistry team is ready to optimize this route specifically for your target molecule.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific catalytic route can reduce your overall production expenses. We encourage you to contact our technical procurement team today to request specific COA data for similar intermediates and discuss route feasibility assessments tailored to your unique development timeline. Let us help you secure a competitive edge in the market with high-quality, cost-effective chiral building blocks.
