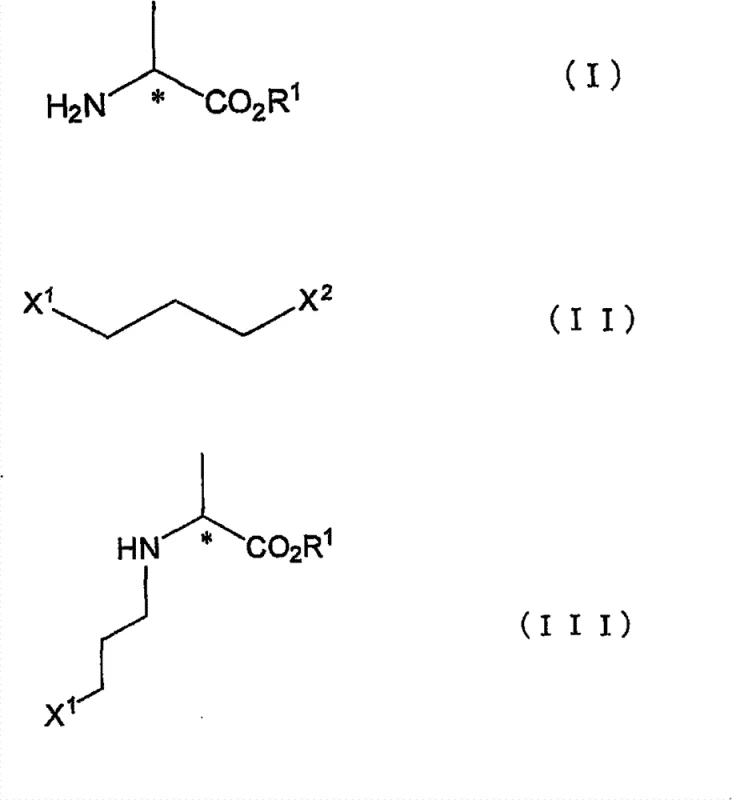
Efficient Commercial Production of Optically Active N-Halopropyl Alanine Derivatives
The pharmaceutical and agrochemical industries continuously demand more efficient pathways for synthesizing chiral building blocks, particularly those serving as precursors for cyclic amino acids. Patent CN101778815B introduces a significant technological advancement in the production of optically active N-(halopropyl)amino acid derivatives. This innovation addresses critical bottlenecks in traditional synthesis by utilizing a direct alkylation strategy that preserves optical integrity while drastically simplifying the operational workflow. By shifting away from expensive halohydrin precursors and eliminating post-synthetic halogenation steps, this method offers a robust solution for manufacturing high-value intermediates. The core of this technology lies in the selective reactivity of dihaloalkanes, specifically leveraging the difference in leaving group ability between bromine and chlorine atoms to achieve regioselective N-alkylation without compromising the chiral center.
This novel approach represents a paradigm shift for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering complex chiral molecules. The process begins with readily available optically active alanine esters, reacting them directly with dihalopropanes such as 1-bromo-3-chloropropane. This strategic choice of starting materials bypasses the multi-step sequences previously required, thereby enhancing overall process mass intensity (PMI). For R&D teams focused on impurity profiles, the elimination of hydroxyl-to-halogen conversion steps removes a major source of potential genotoxic impurities and side products. Furthermore, the method ensures that the asymmetric carbon atom retains its spatial configuration throughout the reaction, delivering products with exceptional enantiomeric excess suitable for stringent regulatory environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-(halopropyl)amino acid derivatives relied heavily on the reaction of amino acids with halohydrins, such as bromopropanol. This conventional route suffers from inherent economic and technical disadvantages that hinder large-scale production. Firstly, halohydrins are significantly more expensive than their dihaloalkane counterparts due to more complex manufacturing processes. Secondly, the initial reaction yields an N-(hydroxypropyl) intermediate, necessitating a subsequent, often harsh, halogenation step to convert the hydroxyl group into a leaving group. This additional transformation not only increases raw material consumption but also introduces new impurities that require rigorous purification, typically involving column chromatography which is impractical for ton-scale manufacturing. Moreover, alternative routes starting from racemic mixtures require optical resolution, effectively discarding 50% of the material and doubling the cost basis for the active enantiomer.
The Novel Approach
The methodology disclosed in CN101778815B overcomes these hurdles by employing dihalopropanes where the two halogen atoms differ in atomic number and reactivity, such as 1-bromo-3-chloropropane. In this system, the bromine atom acts as the superior leaving group, facilitating selective nucleophilic attack by the alanine amine nitrogen, while the chlorine atom remains intact for downstream cyclization. This selectivity eliminates the need for a separate halogenation step entirely. The process operates under mild conditions, typically using inorganic bases like potassium carbonate in polar aprotic solvents such as acetonitrile. By avoiding the handling of unstable hydroxyl intermediates and expensive halohydrins, this approach achieves cost reduction in API manufacturing through simplified unit operations. The result is a streamlined two-step sequence: selective alkylation followed by amine protection, both of which can be executed with high efficiency and minimal waste generation.
Mechanistic Insights into Selective Nucleophilic Substitution
The success of this synthesis hinges on the precise control of nucleophilic substitution kinetics. The reaction mechanism involves an SN2 displacement where the lone pair of the nitrogen atom in the alanine ester attacks the carbon bonded to the more reactive halogen (bromine). The difference in bond dissociation energy and leaving group ability between the C-Br and C-Cl bonds is exploited to ensure that alkylation occurs exclusively at the bromine-terminated end of the propyl chain. This chemoselectivity is crucial because it preserves the terminal chlorine atom, which serves as the handle for future intramolecular cyclization to form proline analogs or other cyclic structures. The use of a base, such as potassium carbonate, serves to neutralize the acid generated during the formation of the ammonium salt, driving the equilibrium toward the free amine and facilitating the substitution. Crucially, the reaction conditions are optimized to prevent racemization at the alpha-carbon of the alanine moiety.
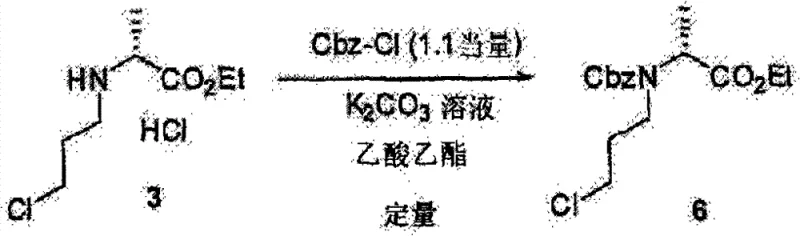
Impurity control is inherently built into this mechanistic design. By avoiding the use of halohydrins, the formation of ether byproducts or elimination products associated with hydroxyl activation is prevented. Furthermore, the patent data indicates that maintaining the reaction temperature between 20°C and 100°C, preferably around 50°C, strikes an optimal balance between reaction rate and stereochemical stability. The preservation of optical purity is paramount; experimental data confirms that starting with alanine esters of >99% ee yields final products with 100% ee. This suggests that the basic conditions employed are sufficiently mild to avoid enolization of the ester or epimerization of the chiral center. The subsequent protection step, whether using Boc anhydride or Cbz chloride, proceeds via standard acylation mechanisms that are well-understood and easily scalable, ensuring that the final high-purity amino acid derivatives meet the strict specifications required for pharmaceutical applications.
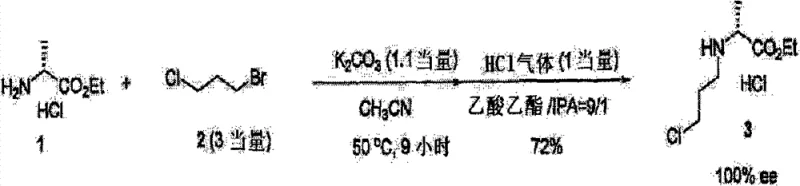
How to Synthesize (R)-N-(3-Chloropropyl)alanine Ethyl Ester Efficiently
The practical implementation of this technology involves a straightforward alkylation followed by a protection step, both of which are amenable to standard reactor setups. The process begins by suspending the alanine ester salt in a solvent like acetonitrile and adding an excess of the dihalopropane along with a base. The reaction mixture is heated to promote the substitution, monitored by GC to ensure complete consumption of the starting amine. Upon completion, the workup involves simple extraction and acidification to isolate the intermediate as a stable hydrochloride salt. This salt can then be directly subjected to protection conditions without the need for extensive purification, further enhancing the process efficiency. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible results.
- React D-alanine ethyl ester hydrochloride with 1-bromo-3-chloropropane (3 equivalents) and potassium carbonate (1.1 equivalents) in acetonitrile at 50°C for 9 hours.
- Isolate the intermediate hydrochloride salt by extracting with ethyl acetate, washing, and treating with HCl gas in ethyl acetate to precipitate the product.
- Introduce a protecting group (e.g., Boc or Cbz) by reacting the free amine or its salt with the corresponding anhydride or acid chloride in the presence of a base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the substantial optimization of the cost structure. By replacing expensive bromopropanol with commodity dihaloalkanes, the raw material cost per kilogram of the intermediate is significantly lowered. Additionally, the elimination of the halogenation step and the associated purification procedures reduces utility consumption, solvent usage, and labor hours. This streamlined workflow allows for faster batch turnover times, directly addressing the industry-wide challenge of reducing lead time for high-purity intermediates. The robustness of the chemistry also implies fewer failed batches and less variability, ensuring a consistent supply stream for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of high-cost specialty reagents with bulk chemicals. 1-bromo-3-chloropropane is produced on a much larger industrial scale than bromopropanol, leading to better pricing stability and availability. Furthermore, the removal of the hydroxyl-to-halogen conversion step eliminates the need for reagents like thionyl chloride or phosphorus tribromide, which carry their own safety and disposal costs. The high yields reported in the patent examples, such as 72% for the alkylation and up to quantitative yields for protection, maximize the throughput of the facility. This efficiency means that less capital is tied up in work-in-progress inventory, improving the overall cash flow of the manufacturing operation.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche starting materials that may have limited suppliers. By utilizing widely available dihaloalkanes and standard alanine esters, the supply chain becomes more resilient to market fluctuations. The simplicity of the synthesis also means that the process can be easily transferred between different manufacturing sites or scaled up from pilot to commercial production without significant re-engineering. This flexibility is critical for mitigating risks associated with single-source dependencies. Moreover, the stability of the intermediate hydrochloride salt allows for easier storage and transportation, providing a buffer against logistical disruptions and ensuring that production schedules for the final API are not compromised.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with green chemistry principles by reducing the number of synthetic steps and the volume of waste generated. The avoidance of column chromatography in favor of crystallization or extraction significantly reduces silica gel waste and solvent consumption. The use of acetonitrile, while requiring proper recovery systems, is a standard solvent in the industry with well-established recycling protocols. The high atom economy of the direct alkylation ensures that a greater proportion of the input materials end up in the final product rather than as byproducts. This reduced environmental footprint simplifies regulatory compliance and waste disposal management, making the commercial scale-up of complex intermediates more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, aiming to clarify the operational parameters and chemical behavior of the system. Understanding these nuances is essential for process engineers and quality assurance teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is 1-bromo-3-chloropropane preferred over bromopropanol for this synthesis?
A: Using 1-bromo-3-chloropropane eliminates the need for a separate halogenation step required when starting from bromopropanol. This reduces raw material costs and simplifies the purification process by avoiding column chromatography.
Q: Does the alkylation process affect the optical purity of the alanine derivative?
A: No, the method described in patent CN101778815B maintains the stereochemistry of the starting material. Starting with >99% ee alanine ester results in a final product with 100% ee, avoiding the need for optical resolution.
Q: What protecting groups are compatible with this synthetic route?
A: The process supports various standard protecting groups including tert-butoxycarbonyl (Boc), benzyloxycarbonyl (Cbz), and benzoyl (Bz), allowing flexibility for downstream cyclization reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(3-Chloropropyl)alanine Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team has extensively analyzed the pathway described in CN101778815B and possesses the expertise to execute this chemistry with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee 100% ee and GC methods to monitor residual halides and solvents. We are committed to delivering intermediates that meet the highest standards of quality and consistency.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific application needs. Whether you require custom synthesis of protected derivatives or bulk supply of the free amine salts, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our reliable manufacturing capabilities.
