Revolutionizing Cyclic Sulfate Production: A One-Step Commercial Scale-Up Strategy for Battery Electrolytes
The landscape of fine chemical manufacturing for energy storage materials is undergoing a significant transformation, driven by the urgent need for safer and more cost-effective synthesis routes. Patent CN111995615B discloses a groundbreaking preparation method for cyclic sulfates, a critical class of compounds serving as essential electrolyte additives in lithium-ion batteries and valuable intermediates in pharmaceutical synthesis. This innovation addresses the longstanding inefficiencies of traditional multi-step processes by introducing a streamlined one-step transesterification metathesis reaction. By utilizing dialkyl sulfate as a primary raw material reacting directly with dibasic esters or cyclic carbonates, the technology bypasses the hazardous and expensive steps associated with conventional oxidation methods. For R&D directors and procurement strategists, this represents a pivotal shift towards sustainable manufacturing that aligns with rigorous environmental standards while maintaining the high purity specifications required for electronic grade chemicals.
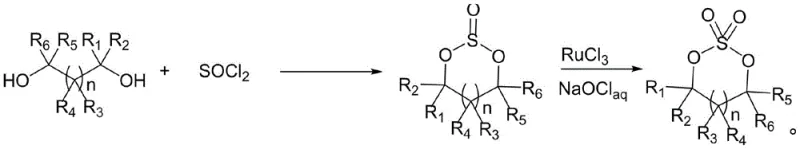
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cyclic sulfates has relied on a cumbersome two-step synthetic pathway that poses significant operational and environmental challenges. As illustrated in the prior art, the process initiates with the reaction of diol compounds with thionyl chloride to form sulfite intermediates, a step notorious for generating large volumes of corrosive hydrogen chloride gas that severely damages reactor integrity. The subsequent oxidation step requires the use of precious metal catalysts, specifically ruthenium trichloride, alongside sodium hypochlorite as a strong oxidant, leading to violently exothermic reactions that are difficult to control safely on a large scale. Furthermore, this oxidative approach generates substantial quantities of salty wastewater, creating a heavy burden on waste treatment facilities and escalating the overall cost of compliance with environmental regulations. The combination of expensive noble metal catalysts, which are difficult to recover and recycle, and the aggressive reaction conditions makes the conventional route economically unattractive for high-volume commercial production.
The Novel Approach
In stark contrast, the novel methodology presented in the patent utilizes a direct transesterification strategy that fundamentally simplifies the molecular construction of the cyclic sulfate ring. This innovative route employs dialkyl sulfate reacting with either a dibasic ester or a cyclic carbonate in a single vessel, effectively merging what were previously distinct synthetic stages into one cohesive operation. The reaction proceeds smoothly under moderate thermal conditions, typically between 50°C and 150°C, utilizing activated clay as a heterogeneous catalyst that is both inexpensive and easily separable from the reaction mixture. By avoiding the use of thionyl chloride and strong oxidants, this method eliminates the generation of corrosive gases and salty effluents, thereby drastically reducing the engineering controls required for safe operation. The result is a cleaner, safer, and more atom-economical process that delivers the target cyclic sulfate structures with high efficiency and minimal environmental footprint.
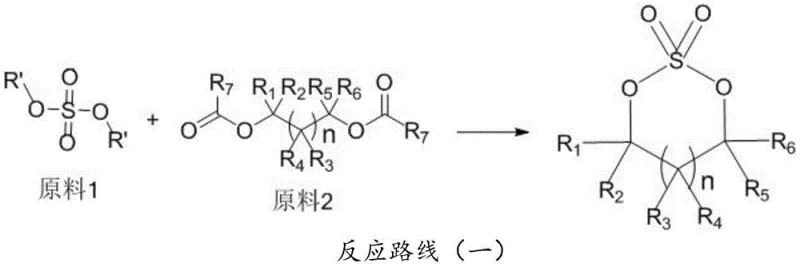
Mechanistic Insights into Activated Clay-Catalyzed Transesterification
The core of this technological advancement lies in the mechanistic efficiency of the transesterification metathesis reaction facilitated by activated clay. Unlike homogeneous acid catalysts that require complex neutralization and separation steps, the solid acid sites on the activated clay surface promote the nucleophilic attack of the ester or carbonate oxygen on the sulfur center of the dialkyl sulfate. This interaction facilitates the exchange of alkoxy groups and the subsequent cyclization to form the stable five or six-membered sulfate ring without the need for external oxidizing agents. The reaction equilibrium is driven forward by the continuous removal of low-boiling byproducts, such as methyl acetate or dimethyl carbonate, through vacuum distillation, ensuring high conversion rates even without stoichiometric excesses of reagents. This mechanism not only enhances the reaction kinetics but also simplifies the downstream processing by allowing the catalyst to remain in the solid phase throughout the transformation.
Furthermore, the selection of activated clay serves a dual function that is critical for achieving the stringent purity levels demanded by the battery industry. Beyond its catalytic activity, the porous structure of the clay acts as a potent adsorbent for linear transesterification byproducts and colored impurities that often plague organic syntheses. This in-situ purification capability means that the crude reaction mixture already possesses a significantly improved color profile and reduced impurity load before any formal crystallization steps are undertaken. By filtering off the spent clay, manufacturers can effectively remove both the catalyst and the adsorbed impurities in a single unit operation, streamlining the workflow. This integrated catalytic and adsorptive action ensures that the final recrystallization yields a product with GC purity exceeding 99.5% and excellent color stability, meeting the rigorous specifications for lithium battery electrolyte additives.
How to Synthesize Cyclic Sulfate Efficiently
The practical implementation of this synthesis route involves a straightforward protocol that balances reaction efficiency with ease of purification. The process begins by charging the reactor with the dialkyl sulfate and the cyclic carbonate or dibasic ester along with the activated clay catalyst under an inert atmosphere. Detailed standardized synthesis steps see the guide below.
- Mix dialkyl sulfate with cyclic carbonate or dibasic ester and activated clay catalyst in a reactor equipped for vacuum distillation.
- Heat the mixture to 50°C-150°C under vacuum (300-5000Pa) to drive the transesterification metathesis reaction and remove low-boiling byproducts.
- Filter the activated clay, remove solvents under reduced pressure, and recrystallize the residue using a mixed solvent system to obtain high-purity cyclic sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers profound strategic advantages that extend far beyond simple chemical transformation. The elimination of precious metal catalysts like ruthenium trichloride removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth and noble metals. Additionally, the removal of corrosive reagents such as thionyl chloride reduces the maintenance frequency of production equipment and extends the lifespan of reactors and distillation columns, leading to substantial capital expenditure savings over time. The absence of salty wastewater generation simplifies the environmental compliance landscape, allowing facilities to operate with lower waste treatment overheads and reduced regulatory scrutiny. These factors combine to create a robust manufacturing platform that ensures consistent supply continuity and cost stability for downstream battery manufacturers.
- Cost Reduction in Manufacturing: The transition away from expensive noble metal catalysts and hazardous oxidants fundamentally alters the cost structure of cyclic sulfate production. By utilizing activated clay, a commodity material, instead of ruthenium complexes, the direct material costs are significantly lowered while simultaneously eliminating the need for complex catalyst recovery systems. The one-step nature of the reaction also reduces energy consumption by removing the heating and cooling cycles associated with intermediate isolation and multi-step processing. Consequently, the overall cost of goods sold is optimized, providing a competitive pricing advantage in the market for battery electrolyte additives without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals like dialkyl sulfates and cyclic carbonates ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions. Unlike specialized oxidants or rare metal catalysts which may face supply bottlenecks, the raw materials for this process are produced at massive scales for other industries, guaranteeing consistent availability. The simplified process flow also reduces the lead time for production batches, enabling manufacturers to respond more agilely to fluctuations in market demand. This reliability is crucial for maintaining the uninterrupted production schedules of lithium-ion battery cell manufacturers who depend on just-in-time delivery of high-purity additives.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the transesterification process make it exceptionally well-suited for commercial scale-up from pilot plants to multi-ton annual production capacities. The mild reaction conditions and lack of hazardous gas evolution allow for the use of standard stainless steel equipment without the need for exotic corrosion-resistant alloys, facilitating easier expansion. Moreover, the green chemistry attributes of the process, specifically the avoidance of toxic byproducts and wastewater, align perfectly with increasingly stringent global environmental regulations. This ensures long-term operational viability and minimizes the risk of production shutdowns due to environmental non-compliance, securing the supply chain for the foreseeable future.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced cyclic sulfate synthesis technology. These insights are derived directly from the experimental data and process descriptions detailed in the patent literature to provide clarity on feasibility and performance. Understanding these aspects is vital for stakeholders evaluating the integration of this method into their existing manufacturing portfolios.
Q: What are the primary advantages of the new cyclic sulfate synthesis method over traditional routes?
A: The new method eliminates the need for corrosive thionyl chloride and expensive ruthenium catalysts, significantly reducing equipment corrosion risks and raw material costs while avoiding salty wastewater generation.
Q: How does the process ensure high purity suitable for lithium battery electrolytes?
A: The use of activated clay serves a dual purpose as both a catalyst and an adsorbent for linear impurities, followed by a specific recrystallization step that achieves GC purity exceeding 99.5%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the one-step transesterification process operates under manageable temperatures (50°C-150°C) and utilizes standard vacuum distillation equipment, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance electrolyte additives play in the next generation of energy storage solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify every batch against the highest industry standards. Our dedication to process excellence allows us to offer reliable cyclic sulfate supplies that empower our partners to achieve superior battery performance and longevity.
We invite you to collaborate with us to leverage these cutting-edge synthesis technologies for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product quality.
