Scalable Manufacturing of Optically Pure (-)-Clausenamide via Novel Asymmetric Catalysis
Scalable Manufacturing of Optically Pure (-)-Clausenamide via Novel Asymmetric Catalysis
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of bioactive natural products, particularly those with potent neuroprotective properties. Patent CN102249976B introduces a groundbreaking preparation method for optically pure (-)-clausenamide compounds, addressing critical bottlenecks in previous synthetic routes. This technology leverages a fructose-derived chiral ketone catalyst to drive the asymmetric epoxidation of trans-cinnamate esters, setting the stereochemical foundation for the entire synthesis. Unlike traditional methods that rely on inefficient chiral resolution, this approach ensures high enantiomeric excess from the outset, making it a superior choice for large-scale manufacturing. The subsequent steps, including transesterification, oxidation, cyclization, and reduction, are meticulously optimized to maximize yield and purity while minimizing environmental impact. For procurement and supply chain leaders, this patent represents a significant opportunity to secure a reliable supply of high-quality pharmaceutical intermediates.
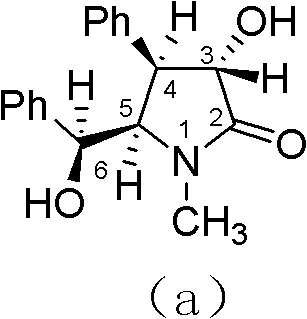
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure (-)-clausenamide has been plagued by significant technical and economic challenges that hinder industrial adoption. Conventional strategies often involve the synthesis of racemic mixtures followed by chiral resolution, a process that inherently caps the maximum theoretical yield at 50% and generates substantial waste. Furthermore, many existing routes rely heavily on preparative chromatography for purification, which is notoriously difficult to scale and incurs prohibitive costs in solvent consumption and stationary phase materials. The reliance on menthol-derived chiral auxiliaries or complex resolution agents also introduces additional steps and raw material costs, complicating the supply chain. These inefficiencies result in a final product that is not only expensive but also subject to batch-to-batch variability in optical purity, posing risks for regulatory compliance in drug development.
The Novel Approach
The methodology disclosed in CN102249976B fundamentally reimagines the synthetic landscape by employing a catalytic asymmetric strategy that bypasses the need for resolution entirely. By utilizing inexpensive fructose-derived chiral ketones as organocatalysts, the process achieves high levels of stereocontrol during the initial epoxidation step, effectively transferring chirality to the final product. This catalytic approach drastically reduces the amount of chiral material required compared to stoichiometric auxiliaries, leading to immediate cost savings. Moreover, the downstream processing has been engineered to favor crystallization over chromatography, with specific optimization of the cyclization step to achieve a cis-to-trans ratio exceeding 20:1. This high diastereoselectivity simplifies isolation procedures, allowing for the use of standard industrial unit operations like filtration and recrystallization, thereby enhancing the overall feasibility of commercial scale-up.
Mechanistic Insights into Fructose-Derived Chiral Ketone Catalysis
The cornerstone of this synthesis is the asymmetric epoxidation of trans-cinnamate esters, mediated by a fructose-derived chiral ketone in the presence of an oxidant such as Oxone. Mechanistically, the chiral ketone reacts with the oxidant to generate a reactive dioxirane intermediate in situ, which then transfers an oxygen atom to the alkene substrate with high facial selectivity. The rigid carbohydrate backbone of the catalyst imposes a specific steric environment that directs the approach of the oxidant, ensuring the formation of the desired (2S, 3R)-epoxy cinnamate with excellent enantiomeric excess. This step is critical as it establishes the stereocenters that dictate the configuration of the final (-)-clausenamide molecule. The use of phase transfer catalysts and additives like EDTA further stabilizes the reaction environment, preventing non-selective background oxidation and maintaining high catalytic turnover.
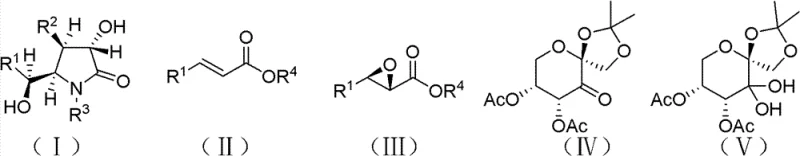
Following the establishment of chirality, the synthetic pathway proceeds through a series of functional group transformations designed to preserve stereochemical integrity. The transesterification step couples the chiral epoxy ester with an amino alcohol, forming the amide bond while retaining the epoxide ring. Subsequent oxidation of the secondary alcohol to a ketone using ruthenium trichloride prepares the molecule for the crucial cyclization event. Impurity control is rigorously managed throughout these steps; for instance, the selection of mild oxidants and controlled temperatures prevents epoxide ring opening or racemization. The final cyclization and reduction steps are tuned to favor the thermodynamically stable cis-isomer, ensuring that the impurity profile remains well within acceptable limits for pharmaceutical applications without the need for complex purification techniques.
How to Synthesize (-)-Clausenamide Efficiently
The synthesis of (-)-clausenamide via this patented route involves a streamlined five-step sequence that transitions smoothly from simple starting materials to the complex target molecule. The process begins with the catalytic asymmetric epoxidation, followed by amidation, oxidation, cyclization, and finally, stereoselective reduction. Each step has been optimized for operational simplicity, utilizing common solvents like acetonitrile, ethyl acetate, and methanol, which facilitates easy solvent recovery and recycling. The detailed standardized synthesis steps below outline the specific conditions, reagent ratios, and workup procedures required to achieve the high yields and purity reported in the patent examples. This protocol is designed to be adaptable for both pilot-scale and full commercial production, ensuring consistent quality.
- Perform asymmetric epoxidation of trans-cinnamate esters using a fructose-derived chiral ketone catalyst and an oxidant like Oxone to generate chiral epoxy intermediates.
- Conduct transesterification of the epoxy ester with N-methyl-beta-hydroxyphenethylamine under basic conditions to form the amide linkage.
- Oxidize the secondary alcohol moiety of the amide intermediate to a ketone using ruthenium trichloride and a stoichiometric oxidant.
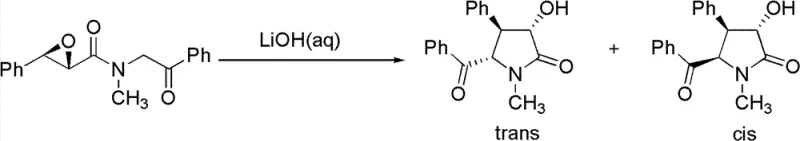
- Execute an intramolecular cyclization under basic aqueous conditions to form the pyrrolidinone ring with high cis-selectivity.
- Complete the synthesis by stereoselective reduction of the ketone group using sodium borohydride to yield the final (-)-clausenamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative advantages in terms of cost structure and supply reliability. By eliminating the need for chiral resolution and chromatographic purification, the process significantly reduces the consumption of expensive chiral reagents and vast quantities of organic solvents. The reliance on crystallization for purification not only lowers direct material costs but also shortens the production cycle time, allowing for faster throughput and improved asset utilization. Furthermore, the use of readily available starting materials like trans-cinnamate esters and fructose derivatives mitigates supply chain risks associated with specialized or scarce reagents. This robustness ensures a continuous and stable supply of high-purity intermediates, essential for maintaining uninterrupted drug manufacturing schedules.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps represents a major driver for cost reduction in pharmaceutical intermediate manufacturing. Traditional methods often require silica gel columns and large volumes of eluents, which are costly to purchase, dispose of, and recycle. By engineering the cyclization step to produce a high ratio of the desired cis-isomer, this process allows for purification via simple recrystallization, drastically cutting down on waste disposal fees and solvent procurement costs. Additionally, the catalytic nature of the chiral induction step means that expensive chiral materials are used in sub-stoichiometric amounts, further lowering the bill of materials. These efficiencies compound to deliver a substantially lower cost of goods sold (COGS) compared to legacy synthetic routes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and robust reaction conditions that are less sensitive to minor variations in raw material quality. The starting materials, such as trans-cinnamic acid derivatives and fructose, are produced on a massive global scale, ensuring that supply disruptions are unlikely to impact production timelines. The process avoids the use of highly unstable or hazardous reagents that might require special handling or storage, simplifying logistics and warehousing requirements. This stability allows for better inventory planning and reduces the need for safety stock, freeing up working capital. Consequently, partners can rely on a predictable and dependable supply of (-)-clausenamide intermediates to support their long-term development and commercialization goals.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns perfectly with modern green chemistry principles and regulatory expectations. The avoidance of heavy metal catalysts in the key chiral induction step and the minimization of solvent waste through crystallization-based purification reduce the environmental footprint of the manufacturing process. The reactions operate at moderate temperatures and pressures, making them suitable for standard stainless steel reactors found in most multipurpose chemical plants. This ease of scale-up means that production can be increased from kilograms to tons without requiring significant capital investment in new equipment. Furthermore, the reduced generation of hazardous waste simplifies compliance with increasingly stringent environmental regulations, lowering the risk of regulatory penalties and enhancing the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (-)-clausenamide using the methodology described in patent CN102249976B. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline. We encourage technical teams to review these points to gain a deeper appreciation of the process capabilities.
Q: How is optical purity achieved in this clausenamide synthesis?
A: Optical purity is established in the first step using a fructose-derived chiral ketone catalyst (Shi catalyst) for asymmetric epoxidation, achieving high enantiomeric excess without the need for chiral resolution.
Q: What is the diastereoselectivity of the cyclization step?
A: The optimized cyclization conditions yield a cis-to-trans ratio greater than 20:1, significantly minimizing the formation of the inactive trans-isomer and simplifying purification.
Q: Does this process require chromatographic purification?
A: No, the process is designed for industrial scalability, utilizing crystallization and extraction for purification, thereby eliminating the cost and waste associated with column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Clausenamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing high-quality intermediates for neuroprotective drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to delivering (-)-clausenamide with stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and crystallization protocols required by this patented process, guaranteeing a product that meets the exacting demands of global pharmaceutical regulators.
We invite you to collaborate with us to optimize your supply chain and reduce your overall development costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines. Let us be your partner in bringing this promising therapeutic candidate to market efficiently and reliably.
