Revolutionizing Thiopyran Synthesis: A Cost-Effective Lewis Acid Catalyzed Route for Pharmaceutical Intermediates
Revolutionizing Thiopyran Synthesis: A Cost-Effective Lewis Acid Catalyzed Route for Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient and sustainable pathways to access complex molecular scaffolds essential for modern drug discovery. Patent CN114380790B introduces a groundbreaking methodology for the synthesis of polysubstituted thiopyran derivatives, a class of six-membered sulfur-containing heterocycles known for their significant biological activity and presence in various natural products. This innovation addresses critical bottlenecks in traditional synthetic routes by utilizing a Lewis acid-promoted [4+2] cyclization strategy that operates under remarkably mild conditions. By leveraging readily available starting materials such as α-thiocarbonyl-N,S-ketene ketals and butynoates, this process eliminates the dependency on scarce and expensive noble metal catalysts, offering a robust alternative for the industrial production of high-value pharmaceutical intermediates. The strategic shift towards base metal catalysis not only aligns with green chemistry principles but also provides a tangible pathway for cost reduction in API manufacturing, making it an attractive option for large-scale commercial operations seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the thiopyran ring system has been a challenging endeavor for synthetic chemists, often relying on multi-step sequences that suffer from poor atom economy and low overall yields. Traditional approaches frequently necessitate the use of precious transition metal catalysts, such as palladium or gold complexes, which introduce significant cost burdens and complicate the purification process due to stringent regulatory limits on residual heavy metals in final drug substances. Furthermore, many existing methods require harsh reaction conditions, including high temperatures or strong bases, which can lead to the decomposition of sensitive functional groups and limit the scope of substrates that can be effectively utilized. These limitations create substantial barriers for procurement managers and supply chain heads who are tasked with securing reliable sources of complex intermediates without incurring exorbitant expenses or facing delays caused by difficult scale-up procedures. The reliance on specialized catalysts also poses a risk to supply continuity, as the availability of these noble metals can be subject to geopolitical fluctuations and market volatility.
The Novel Approach
In stark contrast to these conventional hurdles, the methodology disclosed in CN114380790B offers a streamlined, one-step solution that constructs the thiopyran core with exceptional efficiency and selectivity. By employing zinc chloride (ZnCl2) as a Lewis acid promoter, the reaction facilitates a direct [4+2] cycloaddition between the α-thiocarbonyl-N,S-ketene ketal and the electron-deficient alkyne, bypassing the need for pre-functionalized precursors or expensive catalytic systems. This approach not only simplifies the operational workflow but also significantly enhances the safety profile of the manufacturing process by avoiding toxic reagents and extreme conditions. The versatility of this method is further underscored by its compatibility with a wide range of solvents, with N,N-dimethylformamide (DMF) identified as the optimal medium for achieving superior yields. As illustrated in the general reaction scheme below, this transformation allows for the rapid assembly of diverse polysubstituted thiopyran derivatives, providing medicinal chemists with a powerful tool for exploring new chemical space.
![General reaction scheme showing the Lewis acid catalyzed [4+2] cyclization of alpha-thiocarbonyl-N,S-ketene ketal and butynoate to form polysubstituted thiopyran derivatives](/insights/img/polysubstituted-thiopyran-derivatives-lewis-acid-pharma-supplier-20260302201624-04.webp)
Mechanistic Insights into ZnCl2-Catalyzed [4+2] Cyclization
The success of this synthetic route lies in the precise activation of the alkyne component by the zinc species, which lowers the energy barrier for the subsequent nucleophilic attack by the sulfur-containing enamine system. Mechanistically, the Lewis acid coordinates with the ester carbonyls of the butynoate, increasing the electrophilicity of the triple bond and rendering it susceptible to attack by the electron-rich sulfur atom of the ketene ketal. This initial interaction triggers a cascade of bond-forming events that ultimately result in the closure of the six-membered thiopyran ring with high regioselectivity. The mild nature of the zinc catalyst ensures that the reaction proceeds smoothly without promoting unwanted side reactions such as polymerization or hydrolysis, which are common pitfalls in acid-mediated transformations. Understanding this mechanistic nuance is vital for R&D directors aiming to replicate these results, as it highlights the importance of maintaining anhydrous conditions and controlling the stoichiometry of the Lewis acid to prevent catalyst deactivation or substrate inhibition.
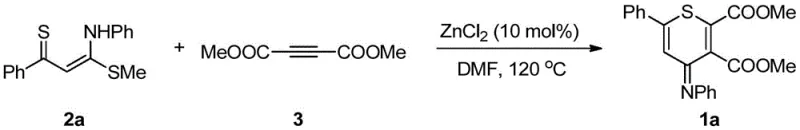
Furthermore, the impurity profile of the resulting thiopyran derivatives is notably clean, attributed to the high chemoselectivity of the zinc-promoted cyclization. Unlike radical-based or oxidative methods that can generate complex mixtures of byproducts, this ionic pathway favors the formation of the thermodynamic product, minimizing the presence of regioisomers or oligomeric impurities. The specific example shown below demonstrates the practical application of this mechanism, where the reaction of a phenyl-substituted ketene ketal with dimethyl butynedioate yields the corresponding thiopyran derivative in an impressive 81% isolated yield. Such high efficiency is indicative of a well-optimized process that balances reaction kinetics with thermodynamic stability, ensuring that the final product meets the rigorous purity specifications required for pharmaceutical applications. This level of control over the reaction outcome is a key differentiator for suppliers looking to establish themselves as leaders in the production of high-purity OLED material or API intermediates.
How to Synthesize Polysubstituted Thiopyran Derivatives Efficiently
Implementing this novel synthetic route requires careful attention to detail regarding reagent quality and reaction parameters to ensure reproducibility and optimal performance. The process begins with the precise weighing of the α-thiocarbonyl-N,S-ketene ketal and the dialkyl butynedioate, followed by the addition of the zinc chloride catalyst in a controlled inert atmosphere to prevent moisture interference. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in scaling this methodology from benchtop to pilot plant operations seamlessly.
- Charge a reaction vessel with alpha-thiocarbonyl-N,S-ketene ketal, dialkyl butynedioate, and 10 mol% Zinc Chloride (ZnCl2) under an inert nitrogen atmosphere.
- Add anhydrous N,N-dimethylformamide (DMF) as the solvent to achieve a molar concentration of 0.05-1.0M for the starting ketene ketal.
- Heat the reaction mixture to 120°C in an oil bath and stir for 12 hours to ensure complete conversion to the polysubstituted thiopyran derivative.
- Upon completion, cool the mixture to room temperature, remove volatiles under reduced pressure, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this zinc-catalyzed protocol offers profound advantages that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. The substitution of noble metals with abundant zinc salts represents a paradigm shift in cost structure, removing the volatility associated with precious metal pricing and reducing the capital expenditure required for catalyst recovery systems. This transition not only lowers the direct material costs but also simplifies the regulatory compliance landscape by eliminating the need for extensive heavy metal testing and remediation protocols that often delay product release. For supply chain heads, the robustness of this method translates into enhanced reliability, as the starting materials are commodity chemicals that can be sourced from multiple vendors, thereby mitigating the risk of single-source dependency. The ability to operate under relatively mild thermal conditions further contributes to energy efficiency, reducing the overall carbon footprint of the manufacturing process and aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or gold catalysts drastically reduces the raw material expenditure per kilogram of product, while the simplified workup procedure minimizes solvent consumption and waste disposal costs. By avoiding the need for specialized ligands or complex catalyst activation steps, the process becomes accessible to facilities with standard chemical processing equipment, lowering the barrier to entry for contract manufacturing organizations. This economic efficiency allows for more competitive pricing strategies in the global market for fine chemical intermediates, enabling companies to capture greater market share without sacrificing profit margins.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a consistent supply flow, reducing the likelihood of production stoppages due to reagent shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or mixing, making it highly suitable for continuous manufacturing setups that demand unwavering consistency. This stability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed delivery schedules and batch-to-batch reproducibility for their clinical and commercial programs.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes the number of unit operations required, streamlining the scale-up process from grams to tons without the need for intermediate isolation or purification steps. The use of zinc chloride, a relatively non-toxic Lewis acid, simplifies effluent treatment and reduces the environmental impact compared to processes generating heavy metal waste streams. This alignment with green chemistry principles not only improves the corporate sustainability profile but also facilitates faster regulatory approvals in jurisdictions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this thiopyran synthesis method, drawing directly from the experimental data and insights provided in the patent literature. These answers are designed to clarify potential ambiguities and provide actionable guidance for technical teams evaluating this technology for adoption. By understanding the nuances of catalyst selection, solvent effects, and substrate scope, organizations can make informed decisions about integrating this process into their existing manufacturing portfolios.
Q: Why is Zinc Chloride preferred over noble metal catalysts for this thiopyran synthesis?
A: Zinc Chloride serves as a cost-effective and relatively non-toxic Lewis acid promoter that efficiently activates the alkyne moiety for [4+2] cyclization. Unlike traditional methods requiring expensive palladium or ruthenium catalysts, ZnCl2 eliminates the need for rigorous heavy metal removal steps, significantly simplifying downstream purification and reducing overall production costs while maintaining high yields up to 90%.
Q: What are the optimal reaction conditions for maximizing yield in this protocol?
A: The patent data indicates that performing the reaction in N,N-dimethylformamide (DMF) at temperatures between 100-120°C provides the best results. Specifically, a reaction time of 5-12 hours under a nitrogen atmosphere with a catalyst loading of 10 mol% ZnCl2 ensures optimal conversion rates and minimizes side reactions associated with oxidative degradation.
Q: Can this synthetic method accommodate diverse functional groups on the thiopyran scaffold?
A: Yes, the method demonstrates excellent functional group tolerance, allowing for the incorporation of various aryl, heteroaryl, and alkyl substituents at the R1, R2, and R3 positions. This structural diversity is crucial for medicinal chemistry applications, enabling the rapid generation of libraries for SAR studies without compromising the integrity of sensitive functional groups like esters or amines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Thiopyran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Lewis acid-catalyzed technology in accelerating the development of next-generation therapeutics and functional materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity polysubstituted thiopyran derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our dedication to quality and consistency makes us the preferred partner for global pharmaceutical companies seeking a reliable source of complex heterocyclic intermediates.
We invite you to collaborate with us to explore how this innovative synthetic route can be tailored to your specific project requirements, offering a Customized Cost-Saving Analysis that highlights the economic benefits of switching to this zinc-catalyzed protocol. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain. Together, we can unlock new possibilities in drug discovery and material science through the power of advanced organic synthesis.
