Scalable Manufacturing of Chiral (S)-4-Hydroxypyrrolidone Intermediates via Natural L-Malic Acid
Introduction to Advanced Chiral Intermediate Manufacturing
The pharmaceutical industry's relentless pursuit of high-purity nootropic agents has placed significant emphasis on the efficient synthesis of chiral building blocks like (S)-4-hydroxypyrrolidone. Patent CN101239938B introduces a transformative methodology that leverages the inherent chirality of natural L-Malic acid to construct the pyrrolidone core with exceptional stereochemical fidelity. This approach represents a paradigm shift from traditional racemic syntheses followed by resolution, offering a direct route to the biologically active (S)-enantiomer which is critical for drugs such as Oxiracetam. By utilizing a renewable, naturally occurring starting material, the process not only aligns with green chemistry principles but also establishes a robust foundation for cost-effective manufacturing. The strategic integration of protection, cyclization, and selective reduction steps creates a streamlined pathway that minimizes waste and maximizes overall yield. For R&D directors and procurement specialists, understanding this technology is essential for securing a reliable supply chain for next-generation cognitive enhancers.
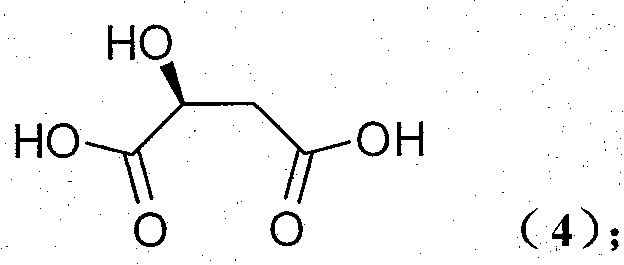
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-4-hydroxypyrrolidone derivatives has been plagued by inefficient multi-step sequences that rely on non-chiral starting materials. Prior art, such as the methods reported in Tetrahedron: Asymmetry (1992), often necessitates complex dehydroxylation reactions or asymmetric hydrogenation steps that require expensive transition metal catalysts and rigorous purification protocols. These conventional routes frequently suffer from low overall yields due to the accumulation of losses at each synthetic stage, particularly during the difficult separation of enantiomers. Furthermore, the use of hazardous reagents and the generation of significant chemical waste pose substantial environmental and regulatory challenges for large-scale production facilities. The economic burden of these inefficiencies is passed down the supply chain, resulting in higher costs for the final active pharmaceutical ingredients (APIs). Consequently, there is an urgent industrial need for a synthesis strategy that bypasses these bottlenecks by utilizing a chiral pool approach.
The Novel Approach
The methodology disclosed in CN101239938B elegantly circumvents these historical hurdles by initiating the synthesis with L-Malic acid, a commercially abundant and inexpensive chiral feedstock. This novel route condenses the synthetic sequence into four distinct, high-yielding operations: hydroxyl protection, thermal cyclization with an amine, chemoselective reduction, and final deprotection. By locking the stereochemistry early in the sequence via the natural chiral center of L-Malic acid, the process guarantees the optical purity of the final product without the need for downstream resolution. The cyclization step is particularly innovative, allowing for the direct formation of the pyrrolidone ring under relatively mild thermal conditions (50 to 200°C). This reduction in step count not only accelerates the production timeline but also drastically reduces the consumption of solvents and reagents, translating directly into improved process mass intensity (PMI) metrics that are crucial for modern sustainable manufacturing.
Mechanistic Insights into Selective Imide Reduction
The cornerstone of this synthetic strategy lies in the precise chemoselective reduction of the cyclic imide intermediate (Formula 2) to the corresponding lactam (Formula 3). This transformation is chemically challenging because imides possess two electrophilic carbonyl groups, and non-selective reduction can lead to diols or over-reduced species that compromise product purity. The patent describes a sophisticated protocol where a metal borohydride, such as sodium borohydride or potassium borohydride, is employed in the presence of a Lewis acid or acyl chloride modulator. This modulation activates one carbonyl group preferentially, directing the hydride attack to a specific position while leaving the other carbonyl intact to form the desired lactam ring. The reaction is conducted within a controlled temperature window of -20 to 20°C, which is critical for suppressing side reactions and maintaining high regioselectivity. Solvent selection plays a pivotal role, with ethers, alcohols, or chlorinated hydrocarbons providing the necessary medium to stabilize the transition state. This level of control ensures that the intermediate retains the integrity of the chiral center established in the earlier steps.
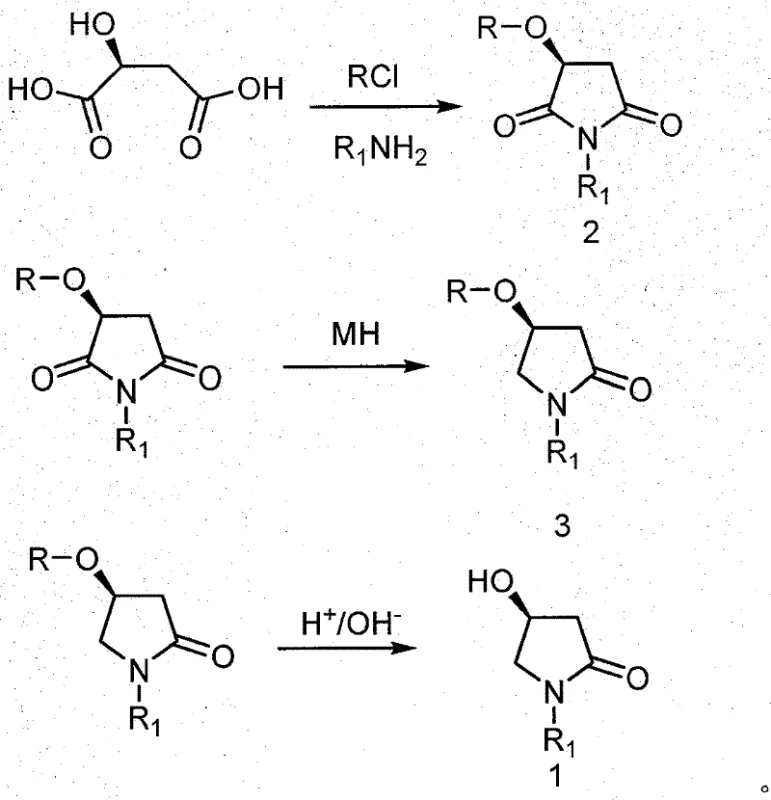
Furthermore, the mechanism inherently supports high impurity control, which is a primary concern for R&D directors overseeing API registration. The use of L-Malic acid ensures that the stereochemical configuration is fixed from the outset, meaning that diastereomeric impurities arising from racemization are virtually eliminated. The selective reduction step, when optimized with the correct acid/acyl chloride ratio, minimizes the formation of over-reduced byproducts, simplifying the subsequent workup and purification processes. The final deprotection step using mineral acids or alkalis is a robust hydrolysis that cleanly removes the protecting group without affecting the newly formed lactam or the chiral center. This mechanistic clarity allows for the establishment of tight in-process controls (IPCs), ensuring that every batch meets stringent specifications for enantiomeric excess (ee) and chemical purity. Such predictability is invaluable when scaling from laboratory benchtop to multi-ton commercial reactors.
How to Synthesize (S)-4-Hydroxypyrrolidone Efficiently
Implementing this synthesis requires a disciplined approach to reaction conditions and reagent quality to fully realize the benefits outlined in the patent. The process begins with the activation of L-Malic acid, followed by a thermal cyclization that demands precise temperature management to ensure complete ring closure without degradation. The subsequent reduction phase is the most sensitive step, requiring careful addition of the borohydride reagent to manage exotherms and maintain the narrow temperature range required for selectivity. Detailed standard operating procedures (SOPs) regarding solvent drying, reagent stoichiometry, and quenching protocols are essential for reproducible results. The following guide outlines the critical operational phases derived from the patent examples, serving as a framework for process engineers to adapt the chemistry to their specific manufacturing infrastructure.
- Protect the hydroxyl group of L-Malic acid using acyl chloride to form the protected intermediate.
- Cyclize the protected acid with an amine at elevated temperatures (50-200°C) to form the cyclic imide intermediate.
- Perform selective reduction of the imide using metal borohydride with acid/acyl chloride modulation at -20 to 20°C to yield the lactam.
- Deprotect the hydroxyl group using mineral acid or alkali to obtain the final (S)-4-hydroxypyrrolidone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this L-Malic acid-based route offers compelling economic and logistical advantages over legacy technologies. The primary driver of value is the substitution of expensive, synthetic chiral catalysts or resolution agents with L-Malic acid, a commodity chemical produced on a massive global scale with stable pricing. This shift fundamentally alters the cost structure of the intermediate, removing the volatility associated with precious metal catalysts and specialized chiral ligands. Additionally, the reduction in the total number of synthetic steps directly correlates to lower operational expenditures (OPEX), as fewer unit operations mean reduced labor, energy, and equipment occupancy time. The simplified workflow also enhances supply chain resilience by reducing the number of potential failure points and minimizing the inventory of hazardous intermediates that need to be stored and transported between stages.
- Cost Reduction in Manufacturing: The elimination of complex asymmetric catalysis and resolution steps leads to substantial cost savings in raw materials and processing. By avoiding the use of expensive transition metals and chiral auxiliaries, the direct material cost is significantly lowered, while the simplified purification requirements reduce solvent consumption and waste disposal fees. This lean manufacturing approach allows for a more competitive pricing structure for the final API, providing a strategic advantage in cost-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing L-Malic acid from established bio-fermentation suppliers ensures a consistent and reliable feedstock availability, mitigating the risk of supply disruptions common with specialty fine chemicals. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures or ultra-high pressures, means that the process can be executed in a wider range of manufacturing facilities, increasing the flexibility of the supply network. This accessibility reduces lead times and allows for more agile responses to fluctuations in market demand for nootropic pharmaceuticals.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing common solvents like alcohols and ethers that are easily recovered and recycled, thereby supporting sustainability goals. The absence of heavy metal catalysts simplifies the regulatory burden regarding residual metal limits in the final drug substance, accelerating the path to market approval. Furthermore, the high atom economy of the cyclization and reduction steps minimizes waste generation, aligning with increasingly strict environmental regulations and reducing the carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures within the patent documentation and are intended to clarify the feasibility and benefits for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into your existing production portfolio.
Q: What is the primary advantage of using L-Malic acid for this synthesis?
A: Using natural L-Malic acid leverages the chiral pool strategy, eliminating the need for expensive asymmetric catalysis or resolution steps, thereby significantly reducing raw material costs and simplifying the supply chain.
Q: How does the selective reduction step ensure high purity?
A: The process utilizes a modulated metal borohydride system with Lewis acids or acyl chlorides at controlled low temperatures (-20 to 20°C), which ensures chemoselective reduction of only one carbonyl group in the imide ring, minimizing byproduct formation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the simplicity of operation, the use of common solvents like alcohols and ethers, and the avoidance of extreme cryogenic conditions, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-Hydroxypyrrolidone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of advanced nootropic therapies. Our technical team has extensively analyzed the L-Malic acid route described in CN101239938B and possesses the expertise to optimize this chemistry for commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of every batch. We are committed to delivering (S)-4-hydroxypyrrolidone derivatives that meet the highest global pharmacopeial standards.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can improve your margins. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and economical supply of this vital pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
