Pioneering Palladium-Catalyzed Route to High-Purity Trifluoromethyl Biheterocyclic Compounds: Scalable Manufacturing Solutions for Global Pharma Partners
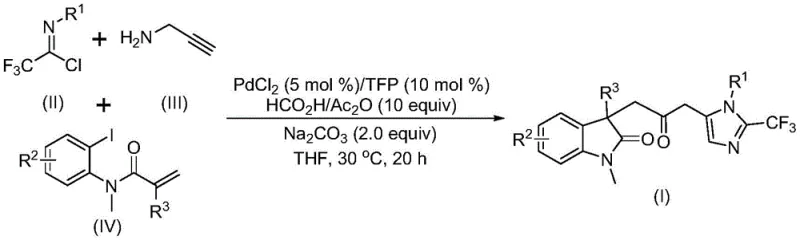
Patent CN115353511A introduces a groundbreaking multi-component synthesis method for carbonyl-bridged biheterocyclic compounds, representing a significant advancement in pharmaceutical intermediate manufacturing that directly addresses critical industry challenges. This innovative approach eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility through a carefully designed palladium-catalyzed cascade reaction sequence. The methodology leverages readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide that can be efficiently transformed into diverse trifluoromethyl-containing biheterocyclic structures under mild reaction conditions at 30°C for 12–20 hours. This patent demonstrates exceptional potential for industrial scale-up with successful gram-scale reactions confirming practical viability for commercial production environments without requiring specialized high-pressure equipment typically associated with conventional carbonylation processes. The elimination of hazardous gases and simplified operational procedures represent substantial improvements over traditional synthetic routes, offering pharmaceutical manufacturers enhanced safety profiles alongside reduced regulatory compliance burdens throughout their production workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds have been plagued by significant limitations that hinder industrial adoption and scalability across pharmaceutical manufacturing operations worldwide. Conventional methods frequently require toxic carbon monoxide gas under high-pressure conditions creating serious safety hazards that necessitate specialized containment equipment substantially increasing capital expenditure for production facilities while complicating regulatory compliance procedures. Direct coupling reactions between heterocycle substrates typically suffer from poor regioselectivity and low yields due to competing side reactions that generate complex impurity profiles requiring extensive purification processes which significantly reduce overall process efficiency and increase production costs per kilogram of final product. Oxidation-based cyclization strategies often produce unwanted byproducts that complicate downstream processing while transition metal-catalyzed methods previously employed demonstrate limited substrate scope where many functional groups prove incompatible with reaction conditions restricting structural diversity of accessible compounds essential for modern drug discovery pipelines requiring tailored molecular architectures.
The Novel Approach
The patented methodology presented in CN115353511A overcomes these longstanding challenges through an elegant palladium-catalyzed cascade reaction operating under mild conditions without hazardous carbon monoxide gas requirements while maintaining exceptional reaction efficiency across diverse substrate combinations. By utilizing formic acid and acetic anhydride as safe carbon monoxide surrogates this innovative approach eliminates significant safety concerns associated with traditional carbonylation methods while preserving high-yield transformation capabilities essential for commercial viability in pharmaceutical intermediate production environments. The process demonstrates remarkable substrate versatility accommodating wide functional group tolerance through careful molecular design enabling synthesis of diverse trifluoromethyl-containing biheterocyclic structures with excellent yields across multiple structural variants as evidenced by experimental data supporting scalability from laboratory to gram-scale reactions confirming industrial readiness. Operating at moderate temperature conditions in common organic solvents like THF this method offers practical advantages for manufacturing implementation requiring minimal specialized equipment while the streamlined one-pot procedure significantly reduces processing steps compared to conventional multi-step syntheses resulting in improved atom economy reduced environmental footprint and enhanced operational efficiency across production facilities.
Mechanistic Insights into Palladium-Catalyzed Carbonyl-Bridged Biheterocyclic Formation
The reaction mechanism involves sophisticated palladium-mediated transformations beginning with zero-valent palladium insertion into carbon-iodine bonds followed by intramolecular Heck reactions generating key divalent alkyl palladium intermediates essential for subsequent cascade transformations. This critical intermediate undergoes controlled carbonylation facilitated by carbon monoxide released from formic acid/acetic anhydride systems forming acyl palladium species that serve as crucial electrophiles driving subsequent molecular transformations toward final product formation. Simultaneously base-promoted condensation between trifluoroethylimidoyl chloride and propargylamine yields trifluoroacetamidine intermediates through intermolecular carbon-nitrogen bond formation which subsequently isomerize to generate nucleophilic partners required for final cyclization steps completing the complex molecular architecture assembly process.
Impurity control is achieved through multiple mechanisms inherent to the reaction design including mild temperature conditions preventing thermal decomposition pathways while carefully optimized reactant stoichiometry minimizes unreacted starting materials and dimerization byproducts that could compromise final product quality profiles required for pharmaceutical applications. Sodium carbonate base promotes clean conversion without causing excessive hydrolysis or other side reactions while specific palladium chloride/trifuryl phosphine ligand combinations create catalytic systems with excellent chemoselectivity directing reactions toward desired cascade pathways while suppressing alternative routes leading to impurities. The one-pot nature eliminates intermediate isolation steps where impurities could accumulate resulting in products meeting stringent pharmaceutical purity specifications without requiring extensive purification beyond standard column chromatography techniques commonly employed in manufacturing environments.
How to Synthesize Trifluoromethyl Biheterocyclic Compounds Efficiently
This patented methodology represents a significant advancement in synthesizing complex biheterocyclic pharmaceutical intermediates through innovative use of safe carbon monoxide surrogates and streamlined reaction design eliminating hazardous operational requirements while maintaining high efficiency suitable for commercial manufacturing environments where safety compliance is paramount concern across global operations. By leveraging readily available starting materials and standard laboratory equipment this approach offers pharmaceutical manufacturers practical pathways to produce high-value intermediates with minimal capital investment requirements while ensuring consistent product quality meeting stringent regulatory standards required throughout drug development pipelines from discovery through commercial production phases.
- Combine trifluoroethylimidoyl chloride (II), propargylamine (III), iodinated precursor (IV), palladium chloride catalyst (5 mol%), trifuryl phosphine ligand (10 mol%), sodium carbonate base (2 equiv), formic acid/acetic anhydride mixture (10 equiv) in THF solvent under nitrogen atmosphere.
- Maintain precise temperature control at 30°C with continuous stirring for 16 hours to ensure complete conversion while preventing thermal decomposition pathways that could compromise product quality.
- Execute post-reaction processing through filtration over silica gel followed by standard column chromatography purification to isolate high-purity carbonyl-bridged biheterocyclic product (I) meeting pharmaceutical grade specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route addresses critical pain points in pharmaceutical intermediate supply chains by offering safer more efficient alternatives to conventional manufacturing methods through elimination of toxic carbon monoxide gas requirements significantly reducing regulatory compliance burdens alongside safety management costs while enabling implementation in standard manufacturing facilities without specialized high-pressure equipment investments required by traditional approaches.
- Cost Reduction in Manufacturing: Elimination of expensive high-pressure reactor systems reduces capital expenditure substantially while cost-effective palladium chloride catalyst usage combined with avoidance of extensive purification steps due to high reaction selectivity contributes significantly to operational cost savings across production facilities without requiring specialized infrastructure investments typically associated with conventional carbonylation processes.
- Enhanced Supply Chain Reliability: Reliance on widely available starting materials with established global supply chains ensures consistent raw material availability regardless of regional market fluctuations or geopolitical factors affecting more specialized reagents while inherent process robustness provides flexibility to adapt potential raw material shortages without significant revalidation efforts maintaining reliable production schedules essential for just-in-time manufacturing environments.
- Scalability and Environmental Compliance: Demonstrated scalability from laboratory to gram-scale reactions confirms readiness for commercial implementation without requiring significant process re-engineering typically needed when transitioning from bench to plant scale while elimination of toxic gases reduces environmental impact aligning with increasingly stringent regulatory frameworks supporting corporate sustainability initiatives through simplified waste management protocols.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement R&D and supply chain professionals regarding implementation benefits of this patented synthetic methodology derived directly from technical specifications experimental data presented in patent CN115353511A providing evidence-based insights into practical application across diverse manufacturing environments serving global pharmaceutical clients.
Q: How does this method eliminate safety concerns associated with traditional carbonylation processes?
A: The patented process replaces hazardous carbon monoxide gas with a safe formic acid/acetic anhydride surrogate system that generates CO in situ under controlled conditions, eliminating high-pressure handling requirements while maintaining efficient carbonylation without compromising reaction efficiency or product quality.
Q: What substrate scope does this methodology accommodate for pharmaceutical intermediate production?
A: The process demonstrates broad functional group tolerance across multiple structural variants including aryl substitutions (methyl, methoxy, halogen, nitro groups) on R¹/R² positions, enabling customization of molecular structures while maintaining consistent yields across diverse therapeutic requirements.
Q: How does this approach improve supply chain reliability compared to conventional methods?
A: By utilizing commercially available starting materials with established global supply chains and eliminating specialized equipment requirements, this methodology reduces vulnerability to raw material shortages while providing flexibility to adapt to potential availability issues without significant process modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Biheterocyclic Compound Supplier
Our patented methodology represents significant advancement in synthesizing complex biheterocyclic pharmaceutical intermediates offering manufacturers safer more efficient pathways essential for modern drug development pipelines requiring high-purity building blocks meeting stringent regulatory standards worldwide NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs and rigorous quality control protocols ensuring consistent product quality meeting global pharmacopeial requirements across all manufacturing scales.
We invite you to contact our technical procurement team requesting specific COA data route feasibility assessments evaluating how this innovative synthetic approach optimizes manufacturing economics ensuring consistent supply chain performance Contact us today to receive your Customized Cost-Saving Analysis tailored specifically to your production requirements.
