Scalable Green Synthesis of 3-Amino-2-Indolones via Metal-Free C-H Amination
Scalable Green Synthesis of 3-Amino-2-Indolones via Metal-Free C-H Amination
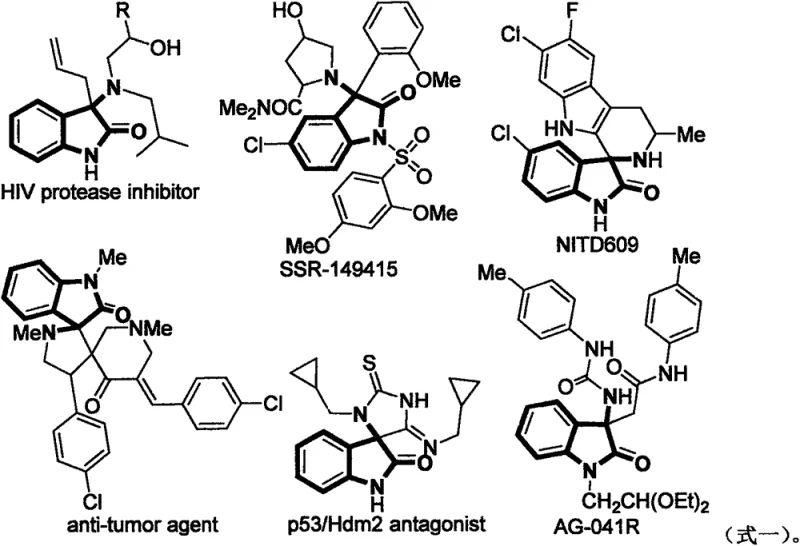
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign pathways to access complex heterocyclic scaffolds that serve as the backbone for numerous therapeutic agents. Patent CN108003086B introduces a transformative approach to synthesizing 3-amino-2-indolone compounds, a privileged structure found in a wide array of bioactive molecules ranging from HIV protease inhibitors to anti-tumor agents. This technology leverages a metal-free I2-TBHP catalytic system to achieve direct C(sp3)-H amination under remarkably mild conditions. By utilizing molecular iodine and tert-butyl hydroperoxide, the process circumvents the need for costly transition metals and harsh acidic environments, representing a significant leap forward in green chemistry manufacturing. The versatility of this method allows for the efficient construction of diverse derivatives, addressing the critical need for reliable pharmaceutical intermediate suppliers who can deliver high-purity materials with a reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-amino-2-indolone core has relied on synthetic routes that pose significant challenges for industrial scalability and cost efficiency. Traditional methodologies often involve intramolecular α-arylation of amides or nucleophilic additions to imines, which frequently require stoichiometric amounts of strong acids such as perchloric acid or the use of expensive precious metal catalysts like rhodium complexes. These conventional pathways not only inflate the raw material costs but also introduce severe safety hazards and waste disposal issues associated with handling corrosive reagents and toxic heavy metals. Furthermore, the necessity for rigorous exclusion of moisture and oxygen in many metal-catalyzed systems demands specialized equipment and inert gas atmospheres, thereby increasing the capital expenditure and operational complexity for manufacturing facilities. The presence of metal residues in the final product also necessitates additional purification steps to meet stringent regulatory standards for pharmaceutical intermediates, further eroding profit margins and extending lead times.
The Novel Approach
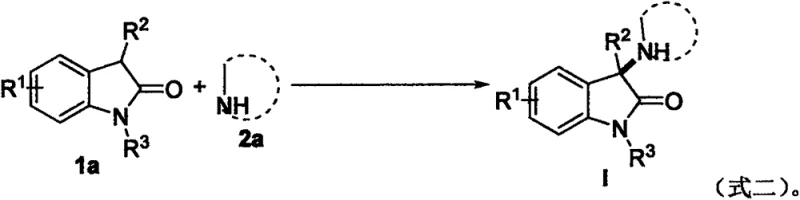
In stark contrast to these legacy methods, the novel protocol described in the patent utilizes a simple yet highly effective I2-TBHP system to drive the amination reaction with exceptional efficiency. This metal-free strategy operates at a moderate temperature of 60°C and uniquely employs ambient air as the reaction atmosphere, eliminating the need for expensive inert gas protection. The use of inexpensive molecular iodine as a catalyst and commercially available TBHP as an oxidant drastically reduces the cost of goods sold while simplifying the supply chain logistics. The reaction proceeds through a radical mechanism that tolerates a wide range of functional groups, allowing for the direct functionalization of 2-indolone derivatives with various amines without the need for pre-functionalized substrates. This streamlined approach not only enhances the overall atom economy but also facilitates a much cleaner reaction profile, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into I2-TBHP Catalyzed C-H Amination
The core of this technological breakthrough lies in the synergistic interaction between molecular iodine and tert-butyl hydroperoxide, which generates reactive radical species capable of activating the inert C(sp3)-H bond at the 3-position of the oxindole ring. The proposed mechanism involves the initial generation of tert-butoxyl radicals from TBHP, which subsequently abstract a hydrogen atom from the substrate to form a carbon-centered radical. This reactive intermediate is then trapped by the amine nucleophile in the presence of the iodine catalyst, leading to the formation of the C-N bond with high regioselectivity. Unlike transition metal catalysis which often suffers from issues related to catalyst deactivation or ligand dissociation, this organocatalytic system maintains robust activity throughout the reaction course. The absence of metal coordination spheres allows for greater flexibility in substrate scope, accommodating sterically hindered amines and electronically diverse indolone derivatives that might otherwise poison a metal catalyst.
From an impurity control perspective, the metal-free nature of this catalytic cycle offers distinct advantages for ensuring product purity and safety. Traditional metal-catalyzed reactions often generate complex mixtures of metal-ligand byproducts or suffer from homocoupling side reactions that are difficult to separate. In this I2-TBHP system, the primary byproducts are tert-butanol and water, which are easily removed during the standard aqueous workup and concentration steps. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups, thereby minimizing the formation of degradation impurities. Moreover, the use of air as the oxidant source avoids the accumulation of hazardous peroxide residues that can occur with stoichiometric oxidants. This clean reaction profile translates directly into simplified downstream processing, reducing the burden on quality control laboratories and ensuring that the final active pharmaceutical ingredients meet the rigorous specifications required by global regulatory agencies.
How to Synthesize 3-Amino-2-Indolone Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory glassware and commonly available reagents. The process begins by charging a reaction vessel with the 2-indolone derivative, the desired amine, catalytic iodine, and TBHP in a suitable solvent such as dichloroethane. The mixture is then heated to 60°C and stirred under an open air atmosphere, with reaction progress monitored by standard analytical techniques like TLC or GC. Upon completion, typically achieved within 4 hours, the solvent is removed under reduced pressure, and the crude product is purified via column chromatography to yield the target 3-amino-2-indolone compound in high purity. For detailed standardized operating procedures and specific molar ratios optimized for different substrates, please refer to the guide below.
- Charge a Schlenk flask with 2-indolone derivative, amine, Iodine catalyst, TBHP oxidant, and dichloroethane solvent.
- Stir the reaction mixture at 60°C under an air atmosphere for approximately 4 hours until completion.
- Concentrate the reaction mixture under reduced pressure and purify the residue via column chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology presents a compelling value proposition centered on cost stability and operational resilience. By eliminating the dependency on volatile precious metal markets for catalysts like rhodium or palladium, manufacturers can secure a more predictable cost structure for their raw materials. The ability to run the reaction under air atmosphere removes the logistical bottleneck of sourcing and storing large volumes of inert gases, thereby enhancing the continuity of supply. Furthermore, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, contributing to substantial cost savings in consumables. This process efficiency allows for faster batch turnover times, enabling suppliers to respond more agilely to fluctuating market demands without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive molecular iodine results in a drastic reduction in catalyst costs, which is a significant component of the overall production expense. Additionally, the avoidance of strong acids and complex ligands reduces the cost of reagents and the associated safety measures required for their handling. The simplified purification process means less solvent usage and lower waste disposal fees, further driving down the total cost of ownership for the manufacturing process. These cumulative savings can be passed down the supply chain, offering competitive pricing for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like iodine and TBHP ensures that the supply chain is not vulnerable to the geopolitical or mining-related disruptions that often affect precious metal availability. The robustness of the reaction under air atmosphere means that production is less susceptible to interruptions caused by utility failures in inert gas lines. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of large-scale pharmaceutical clients. The broad substrate scope also allows for flexible sourcing of starting materials, reducing the risk of single-source supplier dependencies.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly stringent environmental regulations, minimizing the generation of hazardous heavy metal waste. The mild reaction conditions and use of air as an oxidant make the process inherently safer and easier to scale from kilogram to multi-ton production without significant engineering hurdles. This scalability ensures that suppliers can ramp up production capacity quickly to support clinical trials or commercial launches. The reduced environmental footprint also enhances the corporate sustainability profile of the manufacturing entity, a key factor for modern procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific pipeline candidates.
Q: What are the advantages of the I2-TBHP system over traditional Rhodium catalysts?
A: The I2-TBHP system eliminates the need for expensive precious metal catalysts like Rhodium and toxic strong acids, significantly reducing raw material costs and simplifying downstream purification processes by removing heavy metal residues.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (60°C) and uses air as the reaction atmosphere instead of inert gases, which drastically lowers operational complexity and equipment costs for commercial scale-up.
Q: What is the substrate scope for this amination reaction?
A: The method demonstrates broad adaptability, successfully reacting various 2-indolone derivatives substituted with alkyl, halogen, or aryl groups with different amines including morpholine, indoline, and anilines to achieve high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-2-Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity 3-amino-2-indolone intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical infrastructure. By leveraging the metal-free I2-TBHP technology, we can offer our partners a sustainable and cost-effective supply solution that aligns with their long-term strategic goals.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Contact us today to request a Customized Cost-Saving Analysis and obtain specific COA data and route feasibility assessments for your target molecules. Let us collaborate to accelerate your drug development timeline while optimizing your manufacturing budget through superior chemical engineering.
