Advanced Pd-Catalyzed Route to High-Purity Quinolinone Derivatives for Commercial Pharmaceutical Manufacturing Scale-Up
The recently granted Chinese patent CN113045489B introduces a groundbreaking synthetic methodology for producing structurally diverse 3-arylquinoline-2(1H)-one derivatives through an innovative palladium-catalyzed aminocarbonylation process that represents a significant advancement in heterocyclic chemistry. This technology leverages benzisoxazole compounds as dual-function reagents serving simultaneously as nitrogen sources and formyl donors within a single reaction vessel under mild conditions at precisely controlled temperatures. The methodology addresses longstanding challenges in quinolinone synthesis by eliminating the need for multiple synthetic steps and hazardous reagents typically required in conventional approaches such as Vilsmeier-Haack or Friedlander reactions while demonstrating exceptional versatility across a broad spectrum of functional groups including halogens, alkyl groups, and electron-withdrawing substituents. Furthermore, the process maintains high reaction efficiency without requiring specialized equipment or extreme pressure conditions, establishing a new paradigm for manufacturing these pharmacologically important intermediates with direct implications for drug discovery pipelines in the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinolinone derivatives such as Vilsmeier-Haack formylation or Knorr cyclization suffer from multiple critical limitations including harsh reaction conditions requiring strong acids or high temperatures above 150°C that often lead to decomposition of sensitive functional groups. These methods typically involve multi-step sequences with intermediate isolations that significantly reduce overall yields while generating substantial waste streams requiring complex purification procedures. Furthermore, conventional approaches frequently employ expensive or hazardous reagents such as phosphoryl chloride or toxic metal catalysts that necessitate extensive post-reaction treatment to remove trace metal residues below pharmacopeial limits. The narrow functional group tolerance of existing methodologies also restricts structural diversity in final products, limiting their applicability in medicinal chemistry programs where diverse substitution patterns are essential for structure-activity relationship studies. Additionally, scale-up challenges arise from exothermic reactions that become increasingly difficult to control at larger volumes, creating significant barriers to commercial manufacturing.
The Novel Approach
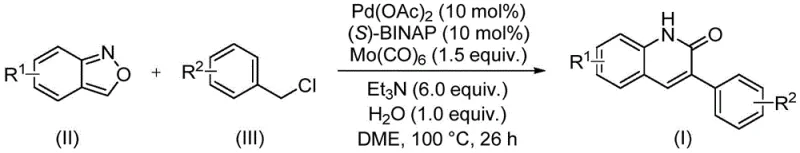
The patented methodology overcomes these limitations through an elegant palladium-catalyzed aminocarbonylation process that utilizes benzisoxazole as a dual-purpose reagent functioning simultaneously as both nitrogen source and formyl donor within a single transformation step. This innovative approach operates under remarkably mild conditions at precisely controlled temperatures of 90–110°C without requiring specialized pressure equipment or hazardous reagents typically associated with traditional methods. The reaction demonstrates exceptional functional group tolerance across diverse substituents including halogens, alkyl groups, alkoxy moieties, and electron-withdrawing groups such as cyano or trifluoromethyl while maintaining high efficiency across all tested substrates. Crucially, the process eliminates multiple intermediate isolation steps through its convergent design, significantly reducing processing time and waste generation while improving overall atom economy. The use of commercially available catalysts including palladium acetate and (S)-BINAP ligand ensures accessibility while the straightforward aqueous workup and chromatographic purification protocol minimizes residual metal content to levels acceptable for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The reaction mechanism proceeds through a well-defined catalytic cycle initiated by oxidative addition of benzyl chloride to the palladium(0) species generated in situ from palladium acetate and (S)-BINAP ligand under reducing conditions provided by molybdenum carbonyl. This key step forms an arylpalladium intermediate that subsequently undergoes nucleophilic attack by the nitrogen atom of benzisoxazole followed by ring-opening to generate an imine species that serves as the formyl equivalent. The catalytic cycle continues with carbon monoxide insertion facilitated by molybdenum carbonyl as a CO source, forming an acylpalladium intermediate that undergoes intramolecular cyclization to construct the quinolinone core structure. The stereochemical control imparted by the chiral (S)-BINAP ligand ensures selective formation of the desired regioisomer while suppressing potential side reactions such as homocoupling or hydrodehalogenation that commonly plague traditional methods. This mechanistic pathway represents a significant departure from conventional approaches by integrating both nitrogen incorporation and carbonyl group installation within a single catalytic sequence.
Impurity control is inherently achieved through multiple design features including the selective nature of the palladium-catalyzed transformation that minimizes side products from competing reaction pathways. The aqueous reaction medium facilitates easy separation of polar impurities during workup while the chromatographic purification step effectively removes residual catalysts and unreacted starting materials without requiring additional specialized techniques. The absence of transition metal contaminants beyond trace levels is ensured through careful optimization of catalyst loading at precisely controlled stoichiometric ratios that prevent metal accumulation during scale-up operations. Furthermore, the mild reaction conditions prevent thermal degradation pathways that typically generate colored impurities in conventional high-temperature syntheses, resulting in products meeting stringent pharmaceutical purity specifications without additional polishing steps.
How to Synthesize Quinolinone Derivatives Efficiently
This patented methodology provides a streamlined approach to synthesizing structurally diverse quinolinone derivatives through a carefully optimized catalytic system that integrates multiple synthetic transformations into a single operation. The process leverages commercially available starting materials including benzisoxazole compounds and substituted benzyl chlorides that can be sourced from multiple global suppliers without supply chain constraints. Detailed standardized synthesis steps are provided below to ensure consistent production quality across different manufacturing scales while maintaining high product purity standards required for pharmaceutical applications.
- Combine benzisoxazole substrate, benzyl chloride compound, palladium acetate catalyst (0.05 mol%), (S)-BINAP ligand (0.05 mol%), molybdenum carbonyl (1.5 equiv.), triethylamine (6.0 equiv.), and water (1.0 equiv.) in ethylene glycol dimethyl ether under nitrogen atmosphere.
- Heat the homogeneous mixture to precisely 100°C with vigorous stirring and maintain reaction temperature for exactly 26 hours to ensure complete conversion while preventing thermal degradation.
- Execute post-processing via filtration through silica gel followed by column chromatography purification using standard elution protocols to isolate high-purity quinolinone products with minimal residual metal content.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming complex multi-step syntheses into a single streamlined operation that significantly reduces raw material complexity while enhancing process reliability. The elimination of hazardous reagents commonly used in traditional quinolinone production not only improves workplace safety but also reduces regulatory compliance burdens associated with handling controlled substances during manufacturing operations.
- Cost Reduction in Manufacturing: The strategic use of benzisoxazole as dual nitrogen and formyl source eliminates separate reagent procurement costs while simplifying reactor setup requirements; elimination of transition metal removal steps reduces downstream processing expenses; utilization of commercially available catalysts at optimized loadings minimizes raw material expenditure without compromising product quality; simplified purification protocols decrease solvent consumption and waste disposal costs across production scales.
- Enhanced Supply Chain Reliability: Sourcing flexibility is achieved through multiple global suppliers for all starting materials including readily available benzisoxazole derivatives; reduced dependency on specialized equipment enables rapid technology transfer between manufacturing sites; consistent product quality across batches minimizes rejection risks during quality control inspections; robust process design accommodates minor variations in raw material quality without requiring significant process adjustments.
- Scalability and Environmental Compliance: The aqueous-based reaction system generates minimal hazardous waste streams compared to traditional methods; straightforward scale-up protocol maintains consistent yields from laboratory to commercial production volumes; elimination of high-pressure requirements reduces capital investment needs; simplified purification process decreases energy consumption per unit mass produced while meeting stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation of this patented methodology based on extensive experimental validation data from the original patent disclosure and subsequent process optimization studies conducted under GMP-compliant conditions.
Q: How does this method overcome limitations of conventional quinolinone synthesis?
A: The novel approach eliminates multi-step sequences and hazardous reagents required in traditional Vilsmeier-Haack or Knorr methods by utilizing benzisoxazole as dual nitrogen and formyl source under mild palladium catalysis at ambient pressure.
Q: What impurity control mechanisms are inherent in this synthetic route?
A: The high functional group tolerance minimizes side reactions while aqueous workup and chromatographic purification ensure stringent removal of metal residues and byproducts without requiring specialized equipment.
Q: How does this process support scalable manufacturing for pharmaceutical supply chains?
A: The use of commercially available starting materials and straightforward purification protocol enables seamless transition from laboratory-scale to multi-ton production volumes without specialized infrastructure requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinone Derivative Supplier
Our company leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver high-purity quinolinone derivatives meeting stringent purity specifications required by global regulatory authorities. With rigorous QC labs equipped for comprehensive analytical testing including advanced chromatography systems and spectroscopic validation methods, we ensure consistent product quality across all manufacturing scales while maintaining full compliance with ICH guidelines throughout the production lifecycle.
We invite you to request our Customized Cost-Saving Analysis tailored to your specific manufacturing requirements through our technical procurement team who can provide detailed information including specific COA data and route feasibility assessments for your target molecules.
