Advanced Mitsunobu Strategy for High-Purity Deuterated Methyl Triclosan Production
Advanced Mitsunobu Strategy for High-Purity Deuterated Methyl Triclosan Production
The landscape of analytical chemistry and environmental monitoring demands increasingly precise standards for quantifying persistent organic pollutants. Patent CN112898132B introduces a groundbreaking synthetic methodology for producing stable isotope deuterium-labeled methyl triclosan, a critical reference material for Isotope Dilution Mass Spectrometry (IDMS). This innovation addresses the long-standing challenges associated with the toxicity and instability of traditional labeling reagents. By leveraging a modified Mitsunobu reaction protocol, the process utilizes deuterated methanol as the sole source of the stable isotope, reacting it with triclosan under mild redox coupling conditions. This approach not only streamlines the synthetic pathway into a single efficient step but also guarantees exceptional isotopic stability, ensuring that the deuterium label remains intact throughout the reaction and subsequent purification phases. For global supply chains reliant on high-fidelity analytical standards, this patent represents a pivotal shift towards safer, more sustainable, and economically viable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methylated triclosan derivatives has relied heavily on the use of dimethyl sulfate as the methylating agent under strongly alkaline conditions. While chemically effective for non-labeled compounds, this traditional route presents severe drawbacks when applied to stable isotope labeling. Dimethyl sulfate is a known carcinogen and poses significant health risks to operators, necessitating expensive containment infrastructure and rigorous waste treatment protocols that drive up operational expenditures. Furthermore, when attempting to introduce a deuterium label using deuterated dimethyl sulfate, the cost of the raw material becomes prohibitive due to its complex synthesis and scarcity. Existing literature also describes routes requiring high-temperature conditions, which increase energy consumption and pose a risk of isotopic scrambling or label loss, thereby compromising the purity required for precise mass spectrometric quantification. These factors collectively create a bottleneck in the reliable supply of high-quality deuterated standards for environmental analysis.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112898132B employs a Mitsunobu reaction mechanism, fundamentally altering the safety and efficiency profile of the synthesis. This novel approach utilizes deuterated methanol (CD3OD), a reagent that is not only commercially accessible and affordable but also exhibits significantly lower toxicity compared to sulfate esters. The reaction proceeds under mild conditions, typically maintained between 0°C and 5°C, which drastically reduces energy requirements and minimizes the thermal stress on the sensitive ether linkage. 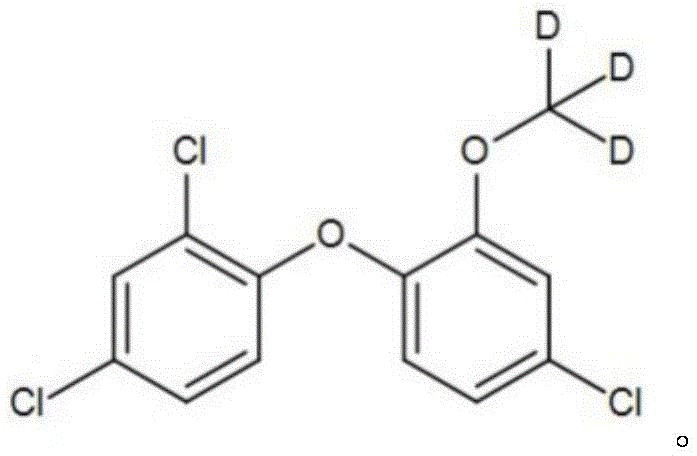 As illustrated in the structural representation, the deuterium atoms are securely positioned on the methyl group attached to the phenolic oxygen. This specific architecture is achieved through a concerted nucleophilic substitution facilitated by the redox couple, ensuring that the isotopic label is introduced with high regioselectivity and without the formation of complex by-products that are difficult to separate. The result is a streamlined process that delivers the target molecule with superior chemical and isotopic purity.
As illustrated in the structural representation, the deuterium atoms are securely positioned on the methyl group attached to the phenolic oxygen. This specific architecture is achieved through a concerted nucleophilic substitution facilitated by the redox couple, ensuring that the isotopic label is introduced with high regioselectivity and without the formation of complex by-products that are difficult to separate. The result is a streamlined process that delivers the target molecule with superior chemical and isotopic purity.
Mechanistic Insights into Mitsunobu-Mediated Deuteration
The core of this synthetic breakthrough lies in the elegant application of the Mitsunobu reaction, a powerful tool for converting alcohols into various functional groups via nucleophilic substitution. In this specific transformation, triclosan acts as the nucleophile, while deuterated methanol serves as the electrophilic partner activated by the phosphine-azo reagent system. The mechanism initiates with the formation of a betaine intermediate between the phosphine reagent, such as triphenylphosphine, and the azo reagent, typically diisopropyl azodicarboxylate (DIAD) or diethyl azodicarboxylate (DEAD). This activation step generates a highly reactive oxyphosphonium species from the deuterated methanol, rendering the methyl carbon susceptible to nucleophilic attack by the phenolic oxygen of triclosan. The reaction is conducted in anhydrous solvents like tetrahydrofuran or dichloromethane to prevent hydrolysis of the active intermediates, ensuring maximum conversion efficiency.
Crucially, the control of impurities is inherent to the mechanistic design of this low-temperature protocol. By maintaining the reaction temperature strictly within the 0°C to 5°C range during the dropwise addition of the azo reagent, the formation of side products is kinetically suppressed. This thermal control prevents potential elimination reactions or over-alkylation that could degrade the isotopic integrity of the final product. Following the reaction period of 1 to 2 hours, the by-products, primarily triphenylphosphine oxide and the hydrazine derivative, are easily removed during the workup phase. The target deuterated methyl triclosan is then isolated via column chromatography, a purification step that is highly effective due to the polarity differences between the product and the phosphine oxide waste. This robust mechanism ensures that the final material achieves both chemical purity and stable isotope abundance exceeding 99%, meeting the stringent requirements for IDMS applications.
How to Synthesize Deuterated Methyl Triclosan Efficiently
The practical execution of this synthesis requires precise adherence to the stoichiometric ratios and thermal parameters defined in the patent examples to ensure reproducibility and high yield. The process begins with the dissolution of the starting materials in an inert atmosphere to exclude moisture, followed by the controlled addition of the activating agents. Operators must monitor the exotherm carefully during the addition phase to maintain the critical low-temperature window. Detailed standardized operating procedures regarding solvent selection, reagent grade, and purification metrics are essential for scaling this chemistry from the benchtop to pilot plant operations. The following guide outlines the critical operational steps derived directly from the patented embodiments.
- Dissolve triclosan, deuterated methanol (CD3OD), and a phosphine reagent (e.g., triphenylphosphine) in an anhydrous solvent like tetrahydrofuran at 0-5°C.
- Dropwise add an azo reagent solution (e.g., DIAD or DEAD) at a controlled rate of 1-2 drops per second while maintaining the temperature between 0-5°C.
- Stir the reaction mixture for 1-2 hours, remove the solvent under reduced pressure, and purify the crude product via column chromatography to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Mitsunobu-based synthesis route offers profound strategic advantages beyond mere technical feasibility. The shift away from hazardous alkylating agents like dimethyl sulfate translates directly into a safer working environment and a substantial reduction in regulatory compliance burdens associated with handling carcinogenic substances. This safety improvement simplifies logistics and storage requirements, allowing for more flexible inventory management and reduced insurance premiums. Furthermore, the use of deuterated methanol as the isotopic source leverages a commodity chemical that is widely available in the global market, mitigating the supply chain risks associated with sourcing exotic, custom-synthesized labeled reagents. This accessibility ensures a consistent and reliable flow of raw materials, which is critical for maintaining uninterrupted production schedules for high-value analytical standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and toxic reagents, coupled with a simplified downstream processing workflow. By avoiding the need for specialized containment equipment required for dimethyl sulfate, capital expenditure for facility upgrades is significantly minimized. Additionally, the high yield and selectivity of the Mitsunobu reaction reduce the volume of waste generated per kilogram of product, leading to lower waste disposal costs and improved overall atom economy. The ability to achieve high purity through standard chromatographic techniques rather than complex distillation or recrystallization sequences further lowers the operational costs associated with energy and labor.
- Enhanced Supply Chain Reliability: The reliance on common laboratory reagents such as triphenylphosphine and DIAD ensures that the supply chain is resilient against disruptions. Unlike proprietary catalysts or rare isotopic precursors that may have long lead times, the key inputs for this synthesis are stocked by multiple global suppliers. This diversification of the supplier base reduces the risk of single-source dependency and allows procurement teams to negotiate more favorable terms. The robustness of the reaction conditions also means that production can be sustained across different geographic locations without the need for highly specialized infrastructure, enhancing the agility of the supply network.
- Scalability and Environmental Compliance: From an environmental perspective, this green chemistry approach aligns perfectly with modern sustainability goals and strict regulatory frameworks. The absence of heavy metal catalysts and the use of low-toxicity solvents facilitate easier compliance with environmental discharge limits. The process is inherently scalable, as the exothermic nature of the Mitsunobu reaction can be effectively managed in larger reactors through controlled dosing strategies. This scalability ensures that manufacturers can rapidly respond to surges in demand for deuterated standards without compromising on quality or safety, providing a competitive edge in the fast-paced analytical reagents market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterated methyl triclosan. These insights are derived from the specific experimental data and advantageous effects reported in the patent literature, providing clarity on the feasibility and benefits of this advanced synthetic route. Understanding these details is crucial for stakeholders evaluating the integration of this material into their quality control workflows.
Q: Why is the Mitsunobu reaction preferred over dimethyl sulfate for deuterium labeling?
A: The Mitsunobu reaction utilizes deuterated methanol, which is significantly less toxic and safer to handle than dimethyl sulfate. It also avoids the harsh alkaline conditions and high energy consumption associated with traditional methylation methods, ensuring the stable isotope label does not detach during the process.
Q: What represents the critical quality attribute for this deuterated standard?
A: The critical quality attributes are chemical purity and isotopic abundance, both of which must exceed 99%. This ensures the material is suitable for use as an internal standard in Isotope Dilution Mass Spectrometry (IDMS) for accurate quantitative analysis of environmental pollutants.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the process operates at mild temperatures (0-5°C) and uses readily available reagents like triphenylphosphine and DIAD. The simple workup involving solvent removal and column chromatography makes it highly amenable to scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Triclosan-D3 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity isotopic standards in ensuring the accuracy of environmental and pharmaceutical analysis. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated Mitsunobu chemistry described in patent CN112898132B can be translated into robust industrial processes. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex deuterium labeling projects positions us as a strategic partner for organizations requiring reliable access to specialized fine chemical intermediates.
We invite you to collaborate with us to optimize your supply chain for deuterated reference materials. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your research and development goals with unmatched reliability and quality.
