Revolutionizing Asymmetric Synthesis: Scalable Production of High-Purity Naphthalene-Pyrrole Phosphine Catalysts for Pharmaceutical Manufacturing
This groundbreaking patent CN114029086B introduces an innovative axial chiral naphthalene-pyrrole phosphine catalyst that represents a significant advancement in asymmetric catalysis technology for the fine chemical industry. The novel catalyst system demonstrates exceptional enantioselectivity while operating under remarkably mild reaction conditions, addressing long-standing challenges in the production of chiral intermediates for pharmaceutical applications. Unlike conventional chiral phosphine catalysts that predominantly rely on carbon-centered chirality, this invention pioneers a unique axial chirality approach based on a naphthalene-pyrrole scaffold, offering unprecedented control over stereochemical outcomes in complex transformations. The patent details a streamlined synthetic pathway that achieves high optical purity and excellent yields, making it particularly valuable for manufacturers seeking to produce high-value chiral compounds at commercial scale. This development is especially timely given the increasing demand for enantiomerically pure pharmaceutical intermediates in the global market, where precise stereochemical control directly impacts drug efficacy and safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral phosphine catalysts have long been constrained by their reliance on carbon-centered chirality, which often leads to limited structural diversity and suboptimal performance in challenging asymmetric transformations. Many existing systems require harsh reaction conditions, including elevated temperatures, strong acids or bases, and extended reaction times, which significantly increase production costs and complicate scale-up processes for industrial applications. Furthermore, conventional catalysts frequently suffer from moderate enantioselectivity, necessitating additional purification steps that further erode process efficiency and economic viability. The scarcity of effective axial chiral phosphine catalysts has been particularly problematic, with most prior art limited to biaryl-based systems that lack the structural flexibility needed for diverse substrate scope. These limitations have created substantial barriers to the efficient production of complex chiral molecules required in modern pharmaceutical synthesis, forcing manufacturers to compromise between yield, purity, and cost-effectiveness.
The Novel Approach
The patented axial chiral naphthalene-pyrrole phosphine catalyst overcomes these limitations through an ingenious molecular design that leverages axial chirality rather than traditional carbon-centered stereocenters. This innovative approach enables the catalyst to operate under exceptionally mild conditions (0-40°C), eliminating the need for energy-intensive heating or cooling systems that drive up operational costs in commercial manufacturing environments. The synthetic pathway described in the patent achieves consistently high enantioselectivity (93% ee or higher across multiple examples) while maintaining excellent yields (71-98%), demonstrating remarkable robustness across diverse substrate combinations. Crucially, the catalyst's modular structure allows for easy structural modification through variations in R groups, enabling fine-tuning for specific reaction requirements without fundamental redesign of the catalytic system. This flexibility represents a significant advancement over conventional approaches, providing manufacturers with a versatile tool for producing a wide range of chiral intermediates with minimal process development effort.
Mechanistic Insights into Axial Chiral Naphthalene-Pyrrole Phosphine Catalysis
The catalytic mechanism of this novel system centers on the unique spatial arrangement of the naphthalene-pyrrole scaffold, which creates a well-defined chiral pocket that precisely controls substrate orientation during asymmetric transformations. The axial chirality arises from restricted rotation around the naphthalene-pyrrole bond, establishing a stable stereogenic axis that directs the approach of reactants with exceptional fidelity. This structural feature enables the catalyst to differentiate between prochiral faces of substrates through a combination of steric and electronic interactions, resulting in highly enantioselective outcomes across various reaction types. The modular nature of the catalyst design allows for systematic optimization of the chiral environment by modifying substituents at key positions (R1-R6), providing researchers with a powerful tool for reaction-specific tuning without compromising the fundamental catalytic architecture.
Impurity control in this catalytic system is achieved through the precise stereodifferentiation enabled by the rigid chiral framework, which minimizes undesired side reactions that typically lead to impurity formation in asymmetric processes. The mild reaction conditions (20°C for key steps) further contribute to impurity reduction by preventing thermal degradation pathways that often complicate traditional catalytic systems operating at elevated temperatures. The patent demonstrates consistent production of products with high optical purity (93% ee or higher) across multiple examples, indicating exceptional selectivity that translates directly to reduced purification requirements in manufacturing settings. This level of impurity control is particularly valuable for pharmaceutical applications where strict regulatory requirements demand extremely high purity standards for intermediate compounds.
How to Synthesize Axial Chiral Naphthalene-Pyrrole Phosphine Catalyst Efficiently
This patented synthesis route represents a significant advancement in the production of complex chiral catalysts, offering a streamlined pathway that maintains high stereoselectivity while operating under commercially viable conditions. The process begins with readily available starting materials and employs a carefully designed sequence of transformations that preserve the critical axial chirality throughout the synthesis. Key innovations include the use of mild reaction conditions (0-40°C) and commercially accessible reagents that eliminate the need for specialized equipment or hazardous materials typically required in traditional chiral catalyst synthesis. The detailed standardized synthesis steps below provide a comprehensive guide for manufacturing teams looking to implement this technology in their production facilities.
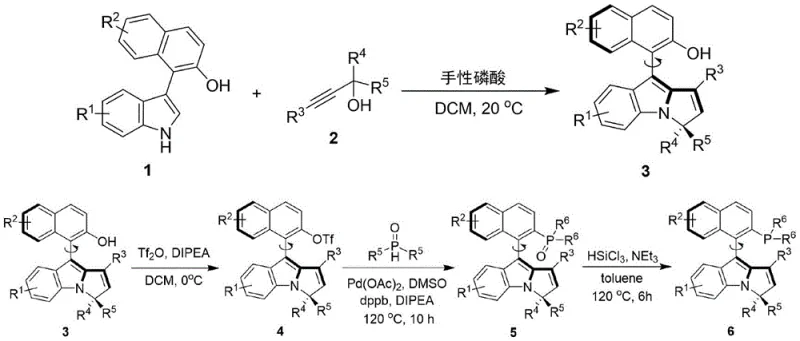
- Combine formula 1 compound with formula 2 compound using chiral phosphoric acid catalyst in dichloromethane at 20°C to obtain formula 3 compound with high enantioselectivity
- Convert formula 3 compound to formula 6 through multi-step synthesis involving trifluoromethanesulfonic anhydride activation followed by palladium-catalyzed coupling reactions
- Purify intermediate compounds using silica gel column chromatography with optimized solvent systems to maintain high optical purity throughout the synthesis
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this novel catalytic system delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional asymmetric synthesis methods. The simplified synthetic pathway reduces dependency on specialized raw materials while maintaining exceptional product quality, creating significant opportunities for cost optimization without compromising performance metrics essential for pharmaceutical applications. This technology enables manufacturers to achieve greater process reliability through its consistent performance across multiple substrate types, reducing batch-to-batch variability that often disrupts supply chain continuity in fine chemical production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in key steps significantly reduces raw material costs while avoiding expensive metal removal processes required in traditional catalytic systems. The mild reaction conditions (20°C) substantially lower energy consumption compared to conventional high-temperature processes, contributing to overall operational cost savings without requiring capital investment in specialized equipment.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and simplified purification requirements creates greater supply chain resilience by reducing dependency on specialized or geographically constrained raw material sources. The consistent high yields (71-98%) across diverse substrate combinations minimize production variability, ensuring reliable output volumes that support just-in-time manufacturing models preferred by major pharmaceutical clients.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production (as evidenced by multiple successful examples) ensures seamless technology transfer without significant process re-engineering. The reduced use of hazardous reagents and lower energy requirements contribute to improved environmental metrics, aligning with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented catalytic technology in industrial settings. Each response is grounded in the specific experimental data and process parameters documented in patent CN114029086B, providing factual guidance for decision-makers evaluating this technology for their manufacturing operations.
Q: What makes this axial chiral naphthalene-pyrrole phosphine catalyst superior to conventional chiral phosphine catalysts?
A: Unlike conventional carbon-centered chiral phosphines, this catalyst utilizes axial chirality which provides enhanced stereochemical control and broader substrate scope while operating under milder conditions (0-40°C) with consistently high enantioselectivity (93% ee or higher).
Q: How does the mild reaction condition benefit large-scale manufacturing of chiral intermediates?
A: The mild reaction conditions (20°C) significantly reduce energy consumption compared to traditional high-temperature processes while eliminating thermal degradation pathways that typically complicate purification and increase impurity profiles in commercial production.
Q: What types of asymmetric reactions can this catalyst effectively catalyze?
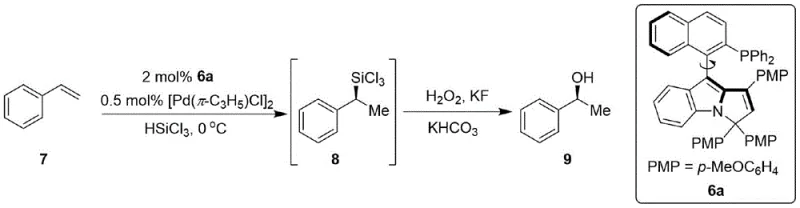
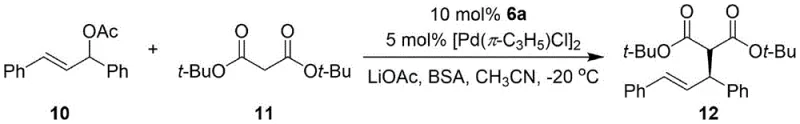
A: This catalyst demonstrates excellent performance in asymmetric hydrosilylation oxidation reactions (e.g., styrene conversion) and asymmetric coupling reactions (e.g., between allyl acetate and di-tert-butyl malonate), both critical transformations in pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Naphthalene-Pyrrole Phosphine Catalyst Supplier
Our company stands at the forefront of advanced catalytic technology development, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. This patented axial chiral naphthalene-pyrrole phosphine catalyst represents just one example of our commitment to delivering innovative solutions that address the most challenging aspects of asymmetric synthesis for pharmaceutical manufacturing. Our technical team has successfully implemented similar complex catalytic systems across multiple therapeutic areas, ensuring we can provide tailored support for your specific production requirements while meeting the highest quality standards demanded by global regulatory authorities.
We invite you to initiate a strategic partnership by requesting our Customized Cost-Saving Analysis, which will demonstrate how this technology can be integrated into your existing manufacturing processes to achieve significant operational improvements. Contact our technical procurement team today to receive specific COA data and route feasibility assessments for your target molecules, enabling you to make informed decisions about implementing this breakthrough catalytic technology in your production pipeline.
